All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
Target Background
Mesothelin, also known as MSLN, is an attractive target for cancer immunotherapy, especially for adoptive T-cell therapy. MSLN is highly expressed in a broad spectrum of solid tumors including mesothelioma, lung cancer, pancreatic cancer, breast cancer, and ovarian cancer and so on while with the exception of normal mesothelia cells. Despite the nonessential function of MSLN in normal tissues, numerous preclinical and clinical studies have shown that aberrant MSLN expression plays an active role in tumor malignant transformation and aggressiveness by promoting cancer cell proliferation, fostering local migration/invasion and metastasis. Misexpression of MSLN also enhances resistance to apoptosis induced by cytotoxic agents, which is in accordance with the good responses of MSLN-targeted clinical immunotherapies. Both the molecular mechanism and the expression discrepancy of MSLN between normal tissues and tumors make it a CAR-T cell target of great potential.
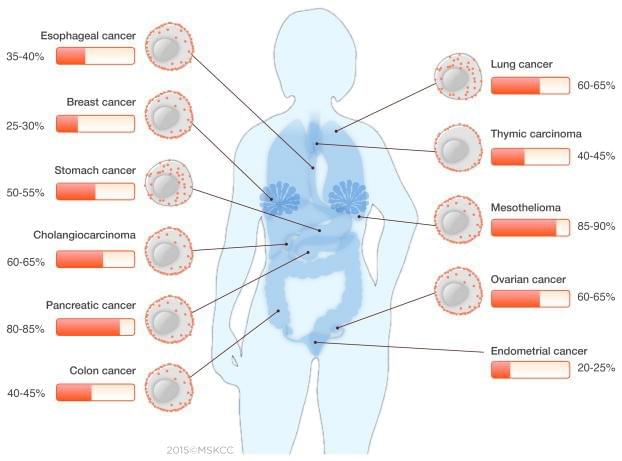
Frequency and distribution pattern of the mesothelin protein in solid malignancies
Cancer Discov. 2016 Feb; 6(2):133-46.
Anti-MSLN CAR-T Cell Therapy
Multiple Phase I clinical trials are currently being conducted to determine the safety and the maximum tolerating dose of MSLN CAR-T cell therapy. Accordingly, different strategies to address the safety concern about the risk of on-target/off-tumor toxicity are also under investigation. For instance, transfection of T cells with mRNA instead of lentivirus that encodes the MSLN CAR results in transient CAR expression, which improves CAR-T safety. And also, the utilization of a suicide gene provides the chance to eliminate T cells in the situation where adverse effects of CAR-T cells emerge. Besides, numerous approaches involving in promoting CAR-T cell infiltration, augmenting the CAR-T cells` functional persistence and enhancing CAR-T cells to combat with inhibitory signals are currently under evaluation to break the obstacles posed to MSLN CAR-T cells by solid-tumor microenvironment.
Animal Models for in vivo Study of anti-MSLN CAR-T Cell Therapy
For the preclinical in vivo studies of MSLN CAR-T cell therapy, Creative Biolabs provides versatile animal models for mesothelioma, ovarian cancer, and lung cancer either in subcutaneous or orthotopic pattern. In addition, Creative Biolabs also assists customers in creating clinically relevant orthotopic mouse models including but not limited to the following categories.
Xenograft models for malignant pleural mesothelioma with the capacity of extensive lymphangiogenesis and mediastinal lymph node metastasis mimicking human pleural mesothelioma by MSTO-211H cell transfer to NOD/SCID gamma mice. Transduced mesothelin CAR-T cells will be adoptively transferred into the thoracic cavity of mice by direct intrapleural injection or by systemic tail vein injection into the tumor-bearing mice, followed by subsequent disease monitoring.
Allograft models for pleural mesothelioma by AB12/EL4 cell transfer to SCID/beige or BALB/c mice.
Ovarian cancer mouse models either by subcutaneously or intraperitoneally inoculating A1847 cells into NSG mice. For the subcutaneous model, when the tumor burden reaches ~150–250 mm3, mice will be injected intratumorally with T cells, followed by subsequent disease monitoring. For the intraperitoneal model, two weeks after peritoneal inoculation, tumor bearing mice will then receive intravenously administered CAR-T cells, followed by subsequent disease monitoring.
Lung adenocarcinoma mouse models by orthotopically injection of H1299 cells into the left lung of NSC mice or by intravenously injection with A549 cells to form metastatic models.
In vivo Assay Parameters and Techniques
At Creative Biolabs, we offer the most exquisite and comprehensive service platform for preclinical MSLN CAT-T cell therapy research.
Efficacy Test
Tumor remission monitored by tumor volume recording or bioluminescence imaging and survival curve tracking.
Viability and Bio-distribution Studies
Durability, GLP-compliant bio-distribution studies
Toxicity Evaluation
Pilot tolerability (MTD, The route of administration, Dose regimen/response/onset)
Clinical observation (body weight, feed consumption, ophthalmologic and clinical pathology)
Cytokine storm surveillance (fever, hypertension, prolonged cytopenia)
Complete necropsy, organ weight
Histopathology
Tumorigenicity study
GLP-Compliant Preclinical Test
All our experiments are performed by well-trained and experienced technicians in a GLP-compliant and IACUC-regulated facility.
To continually keep pace with technology advances, Creative Biolabs intends to establish more reliable and relevant animal models in order to meet the rigorous requirements of preclinical studies for IND application. Beyond that, we welcome any customized service and are pleased to design studies together with our customers. Please feel free to contact our customer service for more information.
References
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION