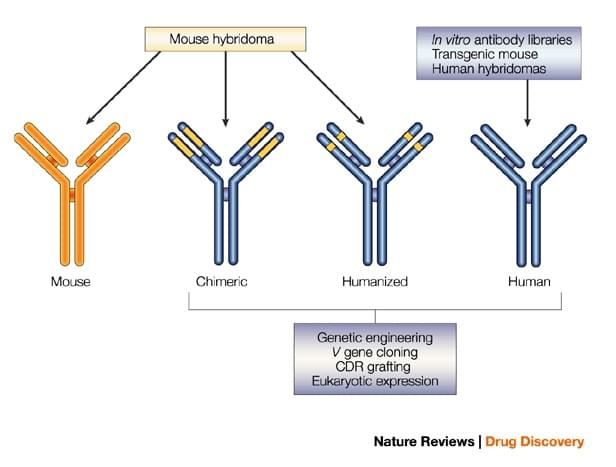
We are a recognized service provider in converting small gene-engineered scFv or Fab human/mouse antibodies derived from phage display antibody library screening or mouse/rat hybridoma cell lines into full-size recombinant human or mouse IgGs with Fc fragments of various functions, e.g. ADCC, or non-ADCC. We have established a solid platform for industrial scale production of recombinant IgG in mammalian cells.
Of note, to mimic the behavior of a recombinant human IgG antibody (targeting a human protein) in humans the best, specificity-matching recombinant mouse IgG antibodies derived from phage display mouse scFv/Fab libraries (targeting the mouse protein) are frequently produced and used in animal models.
We use a modified FRT/Flp-In recombinase system to produce human IgG in CHO cells for stable expression of humanized/recombinant human IgG. The same approach is used in stable expression of recombinant mouse IgG. We have pre-selected CHO cell lines that contain a single stably integrated FRT site at a transcriptionally active genomic locus and we have proved that they can produce high level expressers of recombinant human IgGs (targeting various antigens). Unfortunately, even starting from these pre-selected cell lines, significant amount of time and effort are required to create a stable cell line that can express recombinant human (mouse) IgG at a cost-effective level, e.g. 800mg/L.
In the end of production, the recombinant antibody will be three-step purified (protein A/G followed by two polishing steps, antibody monomer >99%, endotoxin level <0.5 EU/mg antibody, protein A residual < 10 PPM, which is equivalent to antibody for human use). Here, we do not disclose our process technology. We also do not provide the stable cell lines. However, the cell lines will be archived for a period of 2 years free of charge to client in case future production is needed.
• Construction of chimeric antibody expression vectors
- The gene of a scFv or Fab antibody obtained from phage display technology or amplified from a mouse/rat hybridoma cell line is cloned into our proprietary mammalian expression vectors that contain the constant regions of human or mouse IgG light chain and heavy chain.
• Transient transfection of CHO cells
- Transient expression using CHO or 293 cells will be performed to confirm the expression of the recombinant antibody. For small scale antibody production, transient expression is more inexpensive in comparison with stable expression.
• Creating stable CHO cell lines
- Stably transfected CHO cell line are screened to select high expressers. The production cell lines are adapted into protein-free culture medium for bio-manufacturing. The gene-engineered CHO cells we have allow establishment of CHO expressers with extremely high yield, which will dramatically reduce the cost in production of full-size recombinant IgG. We once achieved a cell line with a yield of 4.5g/L.
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.