- Home
- Applications
- Cervical Cancer
ADC Development Services for Cervical Cancer Research
Cervical cancer is the fourth most common malignancy in women with a high incidence and mortality in the world. Traditional therapeutic option such as the chemotherapy is the only notable recent advance in the treatment of recurrent and metastatic cervical cancer. However, the median overall survival (OS) of patients with recurrent cervical cancer had not vastly improved with single agent or combination chemotherapy. Thus, it is needed to design novel treatment strategies to improve the outcome.
Creative Biolabs is a biotechnology company specializing in providing ADCs services and related products. We offer a comprehensive set of ADCs design and construction services targeting the cervical cancer. Our goal is to provide you with the most affordable, high-quality services or the related products to ensure your satisfaction in a timely and professional manner.
Introduction of Cervical Cancer
Cervical cancer is a type of cancer occurring in the cervix, which is the fourth most common cancer in women with a high rate of deaths. The main cause of the cancer is the abnormal growth of cells in the cervix and other parts of the body. The primary types of cervical cancer are squamous cell carcinoma and adenocarcinoma. Commonly, the signs and symptoms of the advanced cervical cancer include the vaginal bleeding after intercourse, between periods or after menopause, watery, bloody vaginal discharge, pelvic pain. Currently, the confirmed cause of cervical cancer is unclear, but it's certain that HPV plays a role. Other risk factors include having many sexual partners, early sexual activity, other sexually transmitted infections (STIs), a weakened immune system, smoking and exposure to miscarriage prevention drug. To date, treatment methods for cervical cancer include the surgery to remove the cancer, the cervix or uterus, radiation therapy, chemotherapy, targeted therapy and immunotherapy.
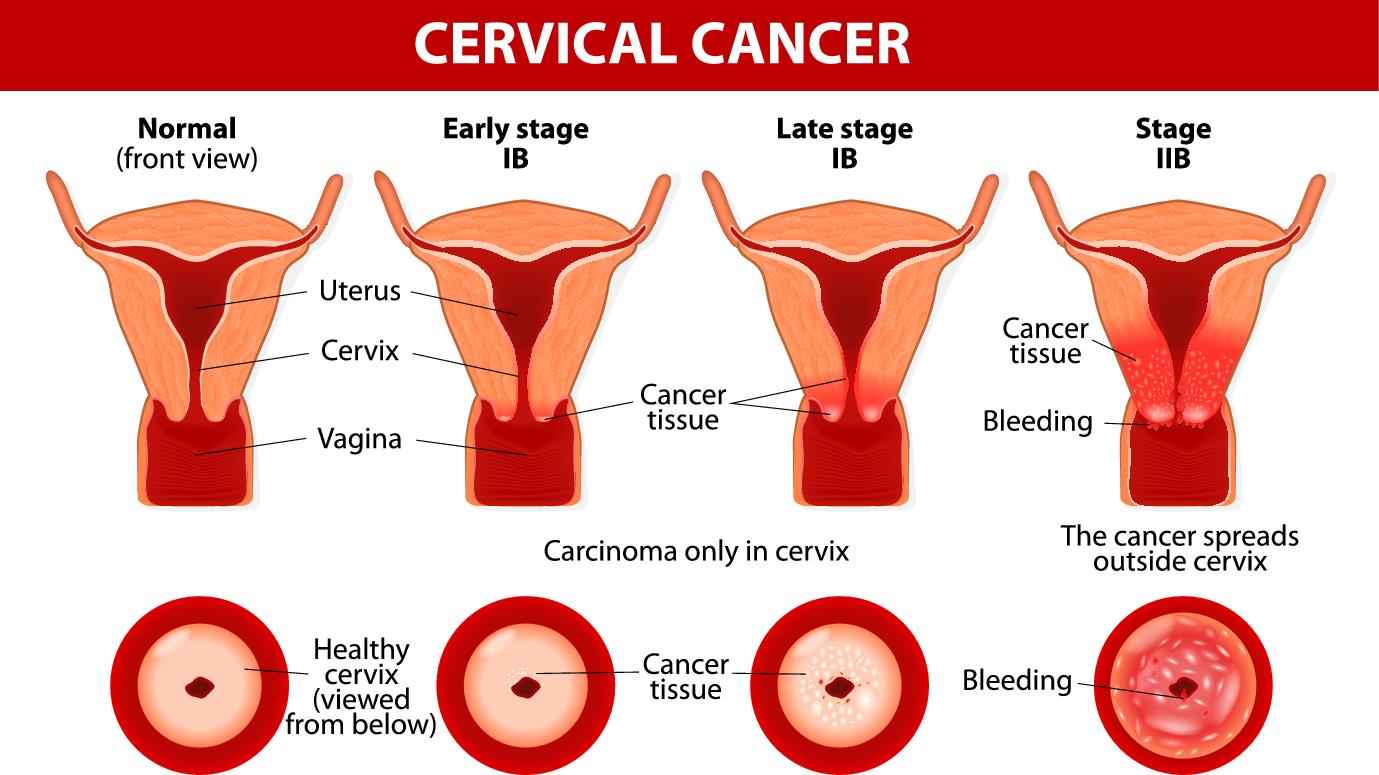 Fig.1 Cervical cancer.
Fig.1 Cervical cancer.
Current Therapeutic Strategies for Cervical Cancer
The standard treatments for various stages of cervical cancer are chemotherapy and surgery currently. However, the outcomes of these conventional therapeutic options remain poor for some recurrent cervical cancer. Some novel targeted treatment approaches have been developed such as the immune checkpoint inhibitors, therapeutic vaccines, antibody-drug conjugates, and others.
- VEGF/VEGFR-directed therapy represents one of the targeted therapies in metastatic and/or recurrent cervical cancer, such as the Bevacizumab and Brivanib. They have shown significant benefits in advanced cervical cancer treatment.
- Immune checkpoint inhibitors are a type of immune-directed therapy for cervical cancer. For example, a fully human antibody against PD-1 is applied as a second-line treatment of persistent or recurrent cervical cancer. Another immune checkpoint inhibitor is being investigated in the phase II trial.
- Cervical cancer therapeutic vaccines focus on eliminating HPV-infected cells by stimulating cytotoxic T cells targeting the viral/tumour antigens. For example, the axalimogene filolisbac (ADXS11-001), a live, attenuated Listeria monocytogenes bacterial vaccine is under assessment for treatment of HPV-associated cervical cancer. Poly (ADP-ribose) polymerase (PARP) inhibitor such as the veliparib is also investigated in women with recurrent or persistent cervical cancer.
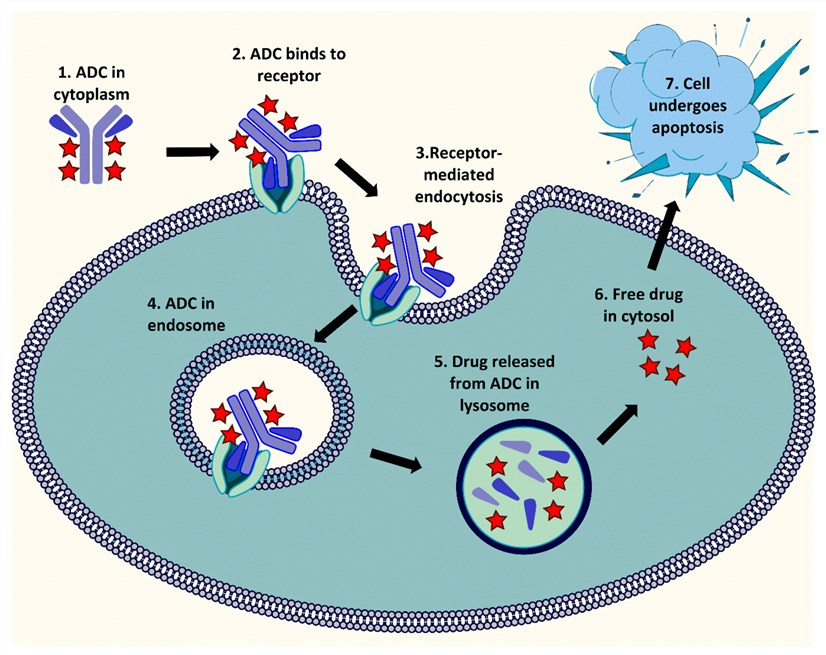 Fig.2 General mechanism of action of ADCs.1,2
Fig.2 General mechanism of action of ADCs.1,2
What Can We Do for You?
Creative Biolabs is a biotechnology company specializing in custom ADCs development and provides ADC preparation services against the cervical cancer. Targets of our ADC development for cervical cancer include, but not limited to:
We have sophisticated equipments, advanced technologies and highly experienced staffs, we can assist in all aspects of ADC development, which include antibody production, drug synthesis, as well as the bioconjugation. For more detail information, please feel free to contact us.
Reference
- Chen, Hao, et al. "Tubulin inhibitor-based antibody-drug conjugates for cancer therapy." Molecules 22.8 (2017): 1281.
- Distributed under Open Access License CC BY 4.0, without modification.
For Research Use Only. NOT FOR CLINICAL USE.

Online Inquiry
Welcome! For price inquiries, please feel free to contact us through the form on the left side. We will get back to you as soon as possible.
