- Home
- Applications
- Mesothelin-expressing Tumor
ADC Development Services for Mesothelin-expressing Tumor Research
Over the past years, antibody-drug conjugate (ADC) as a promising and effective therapeutic agent has been developed widely for a variety of cancer therapies. ADCs possess unique advantages such as the specificity and potent antitumor activity compared with other traditional therapeutic options.
Creative Biolabs is a global leader in the field of antibody development and bio-conjugation, and we offer custom ADCs development services against mesothelin-expressing tumours. With extensive experience in linker-payload synthesis, antibody production and conjugation technology, Creative Biolabs has constructed mature system for ADCs design and construction. Our experienced scientists also provide an extensive range of ADCs related products for ADCs preparation.
Introduction of Mesothelin-expressing Tumours
Mesothelin (MSLN) is a glycosylphosphatidylinositol (GPI)-anchored glycoprotein and is a type of tumour differentiation antigen. Normally, it exists in the mesothelial cells lining the pleura, peritoneum and pericardium. However, the abnormal expression of the protein can cause many cancers including malignant mesothelioma (MM), pancreatic, ovarian and lung adenocarcinoma. Mesothelin is highly expressed in all epithelial malignant mesothelioma. In pancreatic cancer (PC), mesothelin is expressed in all pancreatic adenocarcinomas but absent in normal pancreas, islet cell tumors of the pancreas and in chronic pancreatitis. Mesothelin is overexpressed in most serious ovarian cancers. During lung cancer, mesothelin is present in about half the patients with lung adenocarcinoma. Therefore, mesothelin is a useful marker during the targeted immunotherapy for mesothelin-expressing tumours due to its limited expression in normal tissues and high expression in cancers.
Current Therapeutic Strategies for Mesothelin-expressing Tumours
Mesothelin is highly expressed in many tumours and it has become an attractive target for a variety of therapeutic options. These targeted immunotherapies include the monoclonal antibodies, ADCs, anti-MSLN immunogenic toxin, CAR T-cell therapy and so on.
- Amatuximab (MORAb-009 or anti-MSLN monoclonal antibody) is a chimeric, humanized IgG1/k MAb that targets the cell surface mesothelin. It can potentially inhibit tumor growth by hampering MSLN binding to the extracellular substrate and also by antibody-dependent cellular cytotoxicity (ADCC). Amatuximab is investigated in the Phase II trial for the treatment of pleural mesothelioma and PC.
- SS1P is a recombinant anti-MSLN immunogenic toxin composed of a variable fragment of a murine anti-MSLN antibody and the pseudomonas exotoxin 38 (PE38) portion. SS1P is used to treat the patients with mesothelioma, lung cancer, or PC in the Phase II clinical trial currently. LMB-100 (RG7787) is another recombinant immunogenic toxin targeting MSLN, which consists of a humanized Fab fragment and an engineered PE24. Two initial Phase I–II studies demonstrated that LMB-100 has effective antitumor activity in patients with malignant mesothelioma and pancreatic adenocarcinoma.
- Chimeric antigen receptor (CAR) T-cell therapy is a novel therapy and is displaying promise for mesothelin-expressing tumors. The phase I clinical trial showed that a CAR T-cell therapy targeting the mesothelin has significant antitumor activity and no obvious toxicity in patients with the malignant pleural disease from mesothelioma.
- ADCs as a new therapy strategy has been developed for the treatment of the mesothelin-expressing tumours. BAY 94-9343 (anetumab ravtansine) is an ADC against the MSLN composed of an anti-MSLN antibody and a tubulin polymerase inhibitor DM4. It is assessed in the clinical phase II trials currently for the treatment of patients with mesothelin-positive tumors. DMOT4039A is an ADC consisting of the humanized IgG1 anti-MSLN mAb h7D9.v3 (αMSLN) linked to the antimitotic agent monomethyl auristatin E through a protease-labile linker. It is evaluated in clinical phase I for the therapy of mesothelin-expressing ovarian and pancreatic cancers. BMS-986148 is another ADC targeting the MSLN conjugated with the potent alkylating agent duocarmycin (MED2460). Preclinical and clinical trials showed that this ADC has powerful anti-tumor activity in patients with mesothelin-expressing tumours including the MM, NSCLC, OC, PC, and gastric cancer.
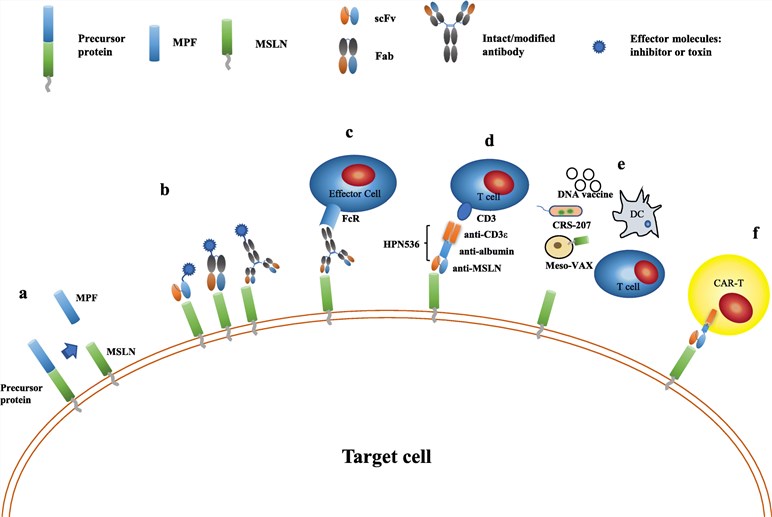 Fig.1 MSLN-targeted therapy strategies.1,2
Fig.1 MSLN-targeted therapy strategies.1,2
Target of ADC Development for Mesothelin-expressing Tumors:
Creative Biolabs has many years of experience in the fields of antibody production and bio-conjugation, we now offer a full range of ADCs development services against the mesothelin-expressing tumours. Creative Biolabs is confident in speeding your novel ADCs drug discovery project and achieving ideal development goals with the most competitive price. For more information, please feel free to contact us.
Reference
- Lv, Jiang, and Peng Li. "Mesothelin as a biomarker for targeted therapy." Biomarker Research 7 (2019): 1-18.
- Distributed under Open Access License CC BY 4.0, without modification.
For Research Use Only. NOT FOR CLINICAL USE.

Online Inquiry
Welcome! For price inquiries, please feel free to contact us through the form on the left side. We will get back to you as soon as possible.
