ADC Development Services Targeting NaPi2B
Non-small cell lung cancer (NSCLC) is the most common cause of cancer death. The development of more potent drugs and linkers with enhanced stability has greatly improved the prospects for antibody-drug conjugate (ADC). Creative Biolabs has been an experienced service provider in the field of antibody development and antibody-drug conjugation. During the past decade, we have constructed multiple technology platforms for ADCs generation and manufacturing. At present, we provide a full range of ADCs development services against the NaPi2B biomarker and we believe our featured services and products will facilitate your novel drug discovery project.
Introduction of NaPi2B
NaPi2B, SLC34A2 gene product, is a multi-transmembrane, type II sodium-dependent phosphate transporter, which is highly expressed in NSCLC, ovarian cancers, and papillary thyroid cancer. As a member of the SLC34 solute carrier protein family, it is responsible for transcellular inorganic phosphate absorption and maintenance of phosphate homeostasis. Besides, NaPi2B has been reported to be associated with cell differentiation and tumorigenesis. Normal lung cells, in the absence of injury, are not highly proliferative and therefore less susceptible to antimitotic agents such as monomethyl auristatin E (MMAE), which largely relies upon cell division to elicit cytotoxic effects. This consideration, together with the retention of high levels of NaPi2B on cancer cells make it a promising target for ADC therapeutics.
Anti-NaPi2B ADC in NSCLC
It has reported that anti-NaPi2B ADC was effective in mouse NSCLC tumor xenograft models and well-tolerated in rats and cynomolgus monkeys at levels in excess of therapeutic doses. For example, anti-NaPi2B-vc-MMAE is an ADC composed of a humanized IgG1 anti-NaPi2B monoclonal antibody (mAb) and MMAE, linked through a protease sensitive valine-citrulline peptide. This ADC exhibited an acceptable safety profile with a dose-limiting toxicity unrelated to normal tissue target expression. This ADC targeting NaPi2B provides an effective new therapy for the treatment of NSCLC and is currently undergoing clinical developments.
Anti-NaPi2B ADC in Ovarian Cancer
NaPi2B protein is also expressed at an immunohistochemistry (IHC) level of 2+/3+ in ~90% of ovarian cancer. Lifastuzumab vedotin (DNIB0600A) is a humanized anti-NaPi2B mAb conjugated to MMAE. The mAb is linked to the cytotoxic agent via a cleavable maleimidocaproyl-valyl-citrullinyl-p-aminobenzyloxycarbonyl (mc-val-cit-PABC) type linker on an average of 3-4 cysteinyl. A phase I study of DNIB0600A in patients with NSCLC and ovarian cancer demonstrated encouraging safety profile and evidence of its antitumor activity. The current phase II study of DNIB0600A was undertaken in platinum-resistant' ovarian cancer (PROC) patients.
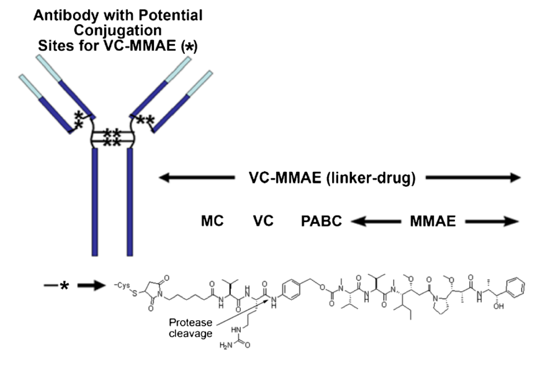 Fig.1 Chemical structure of MMAE-ADCs with MC-VC-PABC linker.1,2
Fig.1 Chemical structure of MMAE-ADCs with MC-VC-PABC linker.1,2
ADC Development Services Targeting NaPi2B
Anti-NaPi2B ADC has demonstrated significant efficacy in non-clinical NaPi2B-expressing xenograft models and exhibited acceptable safety profiles in animal studies, suggesting that anti-NaPi2B ADC may be a promising therapeutic for the treatment of NSCLC and Ovarian Cancer. Creative Biolabs is competent to provide more combinations of mAb, linker, and drug. We also harbor a large collection of pre-assembled DrugLnk Products which are of high quality (>95% purity) and chemically, and can be used for ready-to-go antibody conjugation to speed up ADC development processes. In addition, DrugLnk™ Custom Synthesis is also available for customers here. With toxin targeting defined intracellular targets and linker with various releasing mechanisms, Creative Biolabs will design and synthesis the most appropriate linker-payload to help you develop your own ADC. We also possess featured ADC Antibody Screening platform and perfect Antibody Design and Conjugation platform to ensure the high quality of the project. Once an ADC is prepared, a comprehensive set of ADC in vitro Analysis and ADC in vivo Analysis are avaliable for you. If you are interested in our ADC development services targeting NaPi2B, please feel to contact us for more information.
References
- Lu, Dan, et al. "Semi-mechanistic multiple-analyte pharmacokinetic model for an antibody-drug-conjugate in cynomolgus monkeys." Pharmaceutical Research 32 (2015): 1907-1919.
- Distributed under Open Access License CC BY 4.0, without modification.
For Research Use Only. NOT FOR CLINICAL USE.

Online Inquiry
Welcome! For price inquiries, please feel free to contact us through the form on the left side. We will get back to you as soon as possible.
