- Home
- UTC Development
- Fragment-Drug Conjugate Development
Fragment-Drug Conjugate Development Services
As data emerges highlighting the limitations of traditional immunoglobulin (Ig) G-based antibody-drug conjugates (ADCs), the importance of developing alternative formats for targeted payload delivery is becoming clear. Besides the expertise in traditional ADC design and preparation, Creative Biolabs is also dedicated to the areas of fragment-drug conjugates (FDCs) development utilizing various antibody fragments and scaffold proteins, appropriate linker-payload complexes and different conjugation approaches. With more than 10 years of experience in bio-conjugation chemistry and antibody engineering, the science team at Creative Biolabs provides worldwide customers with comprehensive services from fragment expression to the production of fully conjugated FDCs.
Limitations of Current ADC Technologies
Though ADCs can be excellent therapeutics, significant drawbacks have prevented them from reaching their full potential. Currently marketed ADCs, and the majority of those under development, focus on the IgG scaffold (∼150 kDa). The large size of IgG poses multiple problems such as high systemic accumulation, poor tumor penetration, and slow clearance profiles. Additionally, the presence of the immunogenic Fc region can cause Fc-mediated side effects and decrease clearance rates via salvage recycling by the neonatal Fc receptor pathway. The large size of IgG also presents issues during attachment of cytotoxic payloads.
Exploring Alternative Scaffolds
It is widely known that the targeting capacity of antibodies is achieved through relatively small variable loop structures (VH) present on the ends of the antigen binding fragments (Fab). Exploiting this, engineered scaffolds such as antigen binding fragments (Fab), single-chain variable fragments (scFv), disulphide stabilized scFv (ds-scFv), diabodies and VHH have been generated to improve stability and targeting efficiency. Besides, non-Ig binding proteins have begun to gain attention. They are generally small (<100 amino acids) and based on highly-structured scaffolds isolated from natural proteins. By mutating key residues within the binding sites of these scaffolds, it is possible to alter the affinity and specificity towards different targets. Depending on the targeting component used, antibody fragment-drug conjugates development services and scaffold-drug conjugates development services are available for you.
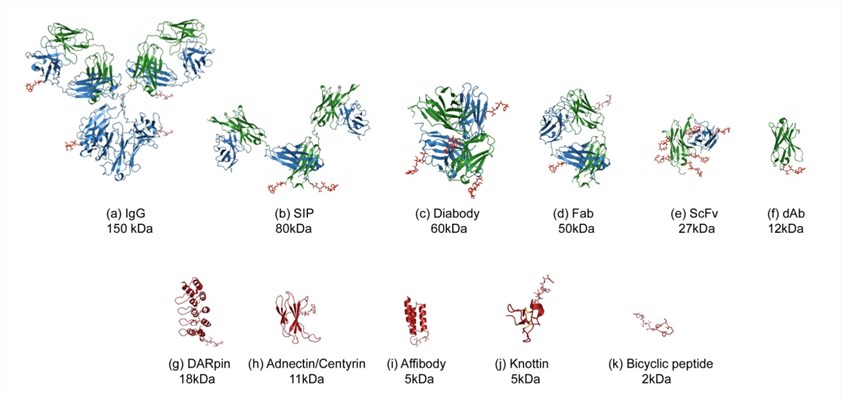 Fig.1 Illustration of the various formats for antibody fragment or alternative scaffold-drug conjugates.1,3
Fig.1 Illustration of the various formats for antibody fragment or alternative scaffold-drug conjugates.1,3
Much like the ADC field, different payload-linker (cleavable, non-cleavable) combinations, and various disease-target (internalizable vs non-internalizable) within the FDC community have been deeply studied. Currently, the most toxic payloads employed with the above scaffolds are pyrrolobenzodiazepines, auristatin (MMAE and MMAF) or maytansinoid (DM1) based. A suitable payload drug will be selected and coupled with an appropriate linker with desired releasing mechanism through our DrugLnk platform to assemble the warhead for an FDC, which is subsequently conjugated with the desired targeting component. Certain modification or engineering of the selected module is also available. In vitro analysis and in vivo analysis will be quite helpful for clients to evaluate the FDC2. It is reported that an acid-cleavable linker strategy was used for Fab-drug conjugation. Functional disulfide bridging of the single interchain disulfide bond of a trastuzumab Fab fragment yields a homogeneous FDC bearing a thiomaleamic acid linker. This linker is stable at physiological pH and temperature, but quantitatively cleaves at lysosomal pH to release the drug payload.
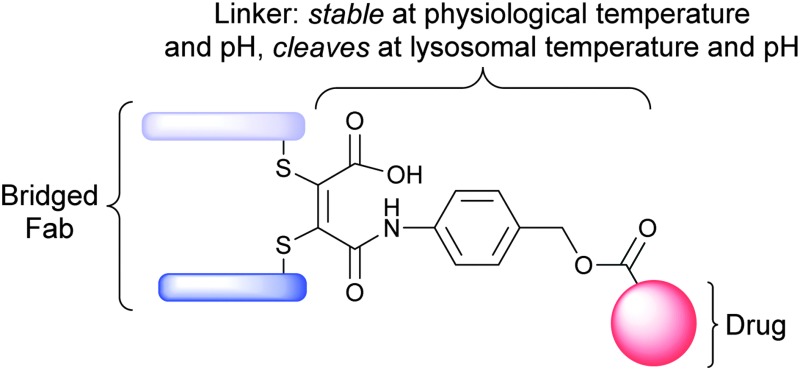 Fig.2 Functional disulfide bridging of the single interchain disulfide bond of a Fab fragment yields a homogeneous FDC.2,3
Fig.2 Functional disulfide bridging of the single interchain disulfide bond of a Fab fragment yields a homogeneous FDC.2,3
Optimized FDC Platform Features
- The use of de novo protein binders such as computationally designed ligands based on structural details of the target
- Prevention of unwanted molecular interactions that inhibit drug activity
- Applicable to most antibody fragment formats and synthetic scaffold protein
- Improved drug-conjugate manufacturing, solubility, and formulation
- Improved pharmacokinetics/dynamics over the whole IgG
- Maximization of drug potency by facilitating higher drug-to-antibody ratio (DAR) for targeted delivery of many active drug molecules
- Small format to enable fast and even tumor penetration ensuring more rapid efficacy
- Small format and lack of Fc to facilitate fast clearance from normal tissue and circulation
- Using a highly potent, clinically-validated cytotoxic payload, FDCs are better tolerated and less toxic than ADCs
- FDCs are relatively easy to discover and can be bioengineered for multiple drug-molecule conjugations, leading to higher loadings
Creative Biolabs has been involved in the development of ADCs for many years and has put a great deal of innovation in FDC generation as well as characterization. With our revolutionary and advanced technology platform, Creative Biolabs has adopted fragments to generate drug conjugates, which have uniquely high payload loadings and improved tumor penetration ability. From the fragments preparation to the acquisition of finally FDC conjugates, we offer one-stop services to meet your R&D timeline and budget. For more information, please do not hesitate to contact us.
References
- Deonarain, Mahendra P., et al. "Small-format drug conjugates: a viable alternative to ADCs for solid tumours?." Antibodies 7.2 (2018): 16.
- Castañeda, Lourdes, et al. "Acid-cleavable thiomaleamic acid linker for homogeneous antibody–drug conjugation." Chemical Communications 49.74 (2013): 8187-8189.
- Distributed under Open Access License CC BY 4.0, without modification.
For Research Use Only. NOT FOR CLINICAL USE.

Online Inquiry
Welcome! For price inquiries, please feel free to contact us through the form on the left side. We will get back to you as soon as possible.
