Protein aggregation is a primary concern in the development of recombinant antibody therapeutics in the biotechnology industry. Aggregates may have unpredictable consequences on drug dose and biological activity of a therapeutic antibody, for example, triggering a severe, or even life threatening immune response in patients. Therefore, antibody therapeutics must be characterized for aggregation as part of the quality assurance process to obtain regulatory approval.
Creative biolabs offers a full range of analytic techniques for BsAbs aggregation, including Dynamic Light Scattering (DLS), Electrospray Differential Mobility Analysis (ES-DMA), Sedimentation Velocity Analytical Ultracentrifugation (SV-AUC), and Field Flow Fractionation (FFF).
Dynamic Light Scattering (DLS)
DLS, also known as Photon Correlation Spectroscopy (PCS) or Quasi-elastic Light Scattering (QELS), is commonly used to measure aggregates in macromolecular solutions, or to measure the sizes of proteins, nucleic acids, and complexes. It is also a useful tool to determine the particle size distribution of protein therapeutic formulations and other similar bioformulations. In a DLS test, the sample is illuminated by a laser beam and the fluctuations of the scattered light are determined at a known scattering angle θ by a fast photon detector.
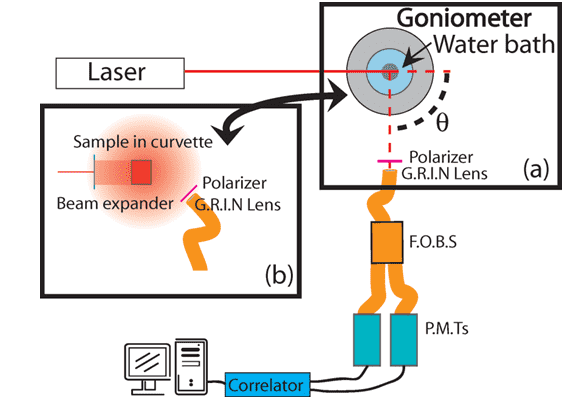
Fig. 1 Schematic of light scattering apparatus showing (a) goniometer for DLS and (b) the DWS setup.1
Electrospray Differential Mobility Analysis (ES-DMA)
ES-DMA, also named as Gas-phase Electrophoretic Mobility Molecular Analysis (GEMMA), is a powerful technique to measure low-order soluble aggregates of proteins in solution. ES-DMA allows to determine large aggregates up to 250 nm in size and small aggregates down to 3 nm with monomeric resolution. Thus, it allows characterization of the initial states of aggregation, especially the dimers, trimers, and tetramers. With a differential mobility analyzer, aqueous solutions of the antibody protein are electrosprayed and the different aerosolized species are separated during the ES-DMA. By this way, complete size distributions of protein species present from 3 to 250 nm can be achieved with the built current, comprising distinct peaks for IgG from monomers to pentamers.
Sedimentation Velocity Analytical Ultracentrifugation (SV-AUC)
SV-AUC is a quantitative technique applied to determine protein size distributions and sedimentation coefficients. Compared to other fractionation-dependent approaches that are more perturbing, SV-AUC leads to minimal disturbance of self-association equilibria, since it extracts size and conformation information directly from a sample in solution. Another advantage of SV-AUC is that experiments can usually be conducted in the product formulation buffer.
Field Flow Fractionation (FFF)
FFF is a promising technique used to measure protein aggregates and particles, according to its broad dynamic range and matrix-free separation mechanism. This technique can be commonly applied to achieve good monomer-oligomer separation and quantification for diverse protein types. Besides, it is a usable choice for an orthogonal approach for AUC.
With our well-established BsAbs analysis platform, the experienced scientists at Creative Biolabs are dedicated to helping you develop BsAbs. Our high quality aggregation measurement services will greatly contribute to the successes of your projects. Creative Biolabs also provides other various services regarding BsAbs development. Please feel free to contact us for more information and a detailed quote.
Reference
1. Mansel, Bradley W., et al. "A practical review of microrheological techniques." Rheology–New Concepts, Applications and Methods (2013). Distributed under open access license CC BY 3.0, without modification.
Our products and services are for research use only, and not for use in diagnostic or therapeutic procedures.
Welcome! For price inquiries, we will get back to you as soon as possible.
To order, please email
INQUIRY





