Creative Biolabs is one of the world’s most trusted experts for bispecific antibody (BsAbs) development and production. Based on our vast experience gained from developing hundreds of antibodies, we offer high quality Antibody-antibody conjugates with high affinity and low immunogenicity for both academic and clinical purposes.
Although genetic engineering is the prevalent methodology for generating BsAbs, there are still a significant number of researches that are performed with chemically crosslinked Abs. By using different starting Abs, crosslinking reagents and conjugation strategies, three formats of antibody-antibody conjugates are generated, IgG-IgG, F(ab’)2 and F(ab’)3.
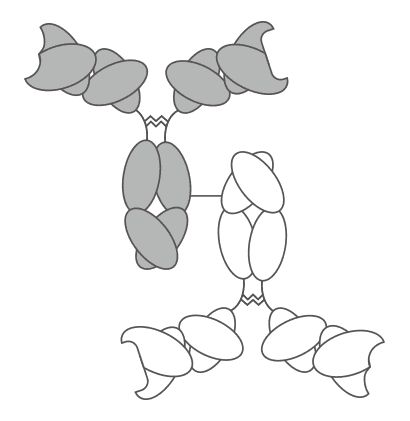
Fig. 1 Diagram of IgG-IgG BsAb.
IgG-IgG BsAbs
IgG-IgG BsAbs, one format of BsAb conjugates, are generated by conjugating two different whole antibodies together to get a 300kDa bispecific molecule with bivalent binding capability for each antigen. The process of IgG-IgG conjugation, which is the crosslinking of intact IgGs, is a common and direct approach for synthesizing BsAb. The generation of this type BsAb usually relies on amino-reactive reagents which target lysine side-chains, and the crosslinkers used to conjugate IgG-IgG are hetero-bifunctional in general. The most extensively used amino-reactive crosslinkers include SPDP (succinimidyl-3(2-pyridylthiol) propionate) and Traut’s reagent (2-iminothiolane)/Sulpho-SMCC(sulpho-[succinimidyl-4-(N-maleimidomethyl)-4-cyclohexane-1-carboxylate]). In the two strategies, different antibodies are incubated separately with same or different reagents. Subsequently, co-incubation of these two modified antibodies can lead to generating the BsAbs linked by a reducible disulfide bond or a non-reducible thioether bond respectively. However, in the strategy that the antibodies conjugate using SPDP, an additional step is needed, which is adding DTT into a low pH reaction system during the last incubation step so as to cleave the crosslinker’s internal disulfide and get the sulfhydryl activated protein.
F(ab’)2 and F(ab’)3 BsAbs
Except that F(ab’)2 can be generated by recombination, the generation of F(ab’)2 and F(ab’)3 BsAbs usually relies on homo-bifunctional reagents, such as o-PDM and DTNB. The fab fragment used in conjugations are generated from recombinant expression or enzymatic digestion of IgGs. One parental Fab is selected for alkylation with o-PDM and the remaining cysteine residue links to a o-PDM molecule containing a nonreacted maleimide, which will further react with the other reduced parental Fab. If the two parental antibodies are equal amounts, a significant amount of F(ab’)2 will be generated. While if the alkylated Fab is present in excess, F(ab’)3 will be the major product.
BsAbs Perspective
In the future, BsAbs may promote treatment options against autoimmune diseases, inflammatory diseases, and cancer. However, large-scale generation and purity are long-term pursuits. The desired platform should contain the whole development process from discovery and preclinical researches to clinical material generation, to enable quick discovery of potent lead BsAbs and purification of clinical-grade BsAbs in a short time.
With our well-established IgG-IgG BsAbs generation platform, the experienced scientists here at Creative Biolabs are dedicated to help you develop unique IgG-IgG BsAbs. We also provide other various services regarding BsAbs development. Please feel free to contact us for more information and a detailed quote.
Our products and services are for research use only, and not for use in diagnostic or therapeutic procedures.
Welcome! For price inquiries, we will get back to you as soon as possible.
To order, please email
INQUIRY





