As a global leader of recombinant antibody discovery and manufacturing, Creative Biolabs provides a range of purity measurement services for bispecific antibodies (BsAbs) and other therapeutic antibodies. With our comprehensive analysis, you can have an overall understanding of the biochemical features of your BsAbs. We are dedicated to offering you the first-class purity measurement services to meet your goal of BsAb development in a timely and cost-effective manner.
Our purity measurement services include but are not limited to the following approaches:
-
SDS-PAGE Analysis
-
Liquid Chromatography-Mass Spectrometry (LC-MS) Analysis
-
Partial-filling Affinity Capillary Electrophoresis (PFACE)
SDS-PAGE Analysis
Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) is a commonly used technique for antibody-purity analysis. In this method, a polypeptide chain combines with SDS proportionally to its relative molecular mass. Due to the special property of SDS, it denatures proteins by disrupting their noncovalent bonds, thus simplifying the molecular structures of those proteins. Besides, proteins are consistently coated by negatively charged SDS, leading to electric current-driven separation toward an anode. Due to a SDS–protein constant-weight binding ratio of 1:1.4, the intrinsic polypeptide charge can be ignored. Therefore, the final separation of proteins completely depends on the differences between the relative molecular masses of their denatured polypeptides.
Liquid Chromatography-Mass Spectrometry (LC-MS) Analysis
Liquid chromatography (LC) is a useful technique to separate a sample into its individual parts. Based on the interactions of the sample with the mobile and stationary phases, the separation is achieved. Mass spectrometry (MS) is an analytical technique that is able to quantify known materials, to identify unknown compounds within a sample, as well as to characterize the structure and chemical properties of different molecules. LC-MS, referred to use LC as a separation system and MS as a detection system, eventually achieves the spectrum. With the LC and MS being coupled, we are able to perform multi-stage MS to speculate the structure of the compound, thereby providing more accurate qualitative and quantitative analyses.
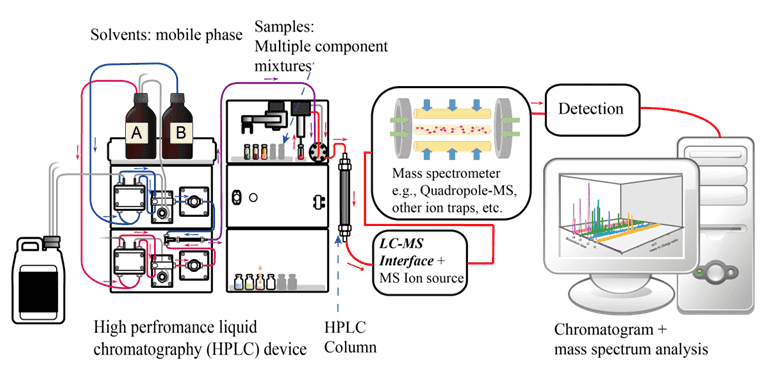 Distributed under CC0 1.0, from Wiki.
Distributed under CC0 1.0, from Wiki.
Partial-filling Affinity Capillary Electrophoresis (PFACE)
Affinity Capillary Electrophoresis (ACE) is a potent analytical technique that is used to study various bimolecular noncovalent interactions and to measure the binding and dissociation constants of formed complexes. This strategy takes advantage of the resolving power of CE to distinguish between the free and bound forms of a receptor as a function of the concentration of the free ligand. In Partial-filling ACE (PFACE), small plugs of sample are injected into the capillary column; equilibrium is set up between the receptor and the ligand during electrophoresis. Then, binding constants are achieved by Scatchard analysis based on the changes in the migration time of the receptor/ligand following the changes in the concentration of the ligand/receptor. Recently, PFACE is used for the purity analysis of BsAbs, exhibiting an outstanding performance compared to other approaches.
With our well-established BsAbs analysis platform, the experienced scientists at Creative Biolabs are dedicated to helping you develop BsAbs. Our high quality analysis services will greatly contribute to the successes of your projects. Creative Biolabs also provides various other services related to BsAbs development. Please feel free to contact us for more information and a detailed quote.
Our products and services are for research use only, and not for use in diagnostic or therapeutic procedures.
Welcome! For price inquiries, we will get back to you as soon as possible.
To order, please email
INQUIRY





