Antibody-modified Adenovirus Vector Construction Service
Adenovirus (Ad) vectors are the most commonly used system for gene therapy applications, due in part to their ability to infect a wide range of cell types and tissues. However, to achieve successful gene therapy, the appropriate genes must be delivered to and expressed in target cells, without harming non-target cells. During the past years, many efforts have been made by scientists to develop technologies and strategies to alter the Ad surface, thereby achieving cell-specific targeting. Antibodies and antibody-derived moieties are potentially useful agents that can be attached to the Ad surface to provide target-specific gene transfer due to their unparalleled affinity and specificity of binding to a wide range of target cell surface markers.
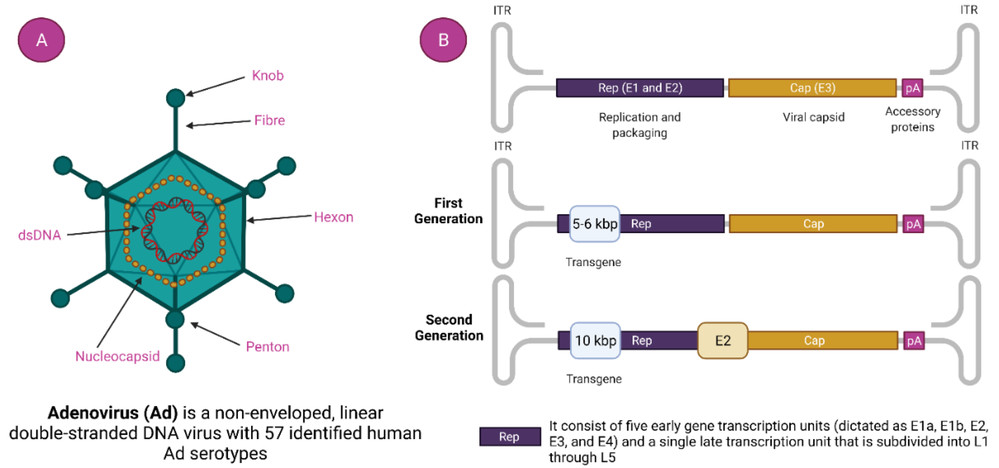 Figure 1. Adenovirus and adenovirus-based vector. (A). Adenovirus Structure and (B). Generation of Adenoviral Vectors.1
Figure 1. Adenovirus and adenovirus-based vector. (A). Adenovirus Structure and (B). Generation of Adenoviral Vectors.1
At Creative Biolabs, with solid expertise and optimized technology, we are ready to offer unparalleled Ad vector design and construction services to our global clients. Here, we introduce our antibody-modified Ad vector construction services to facilitate retargeting Ad infection specificity.
Value of Antibody-Modification Adenoviral Vectors Solutions
Antibody-modified adenoviral vectors address these pain points at the source by:
- Redirecting vector tropism to target cells via antibody-antigen recognition, eliminating CAR receptor dependence.
- Masking immunodominant epitopes on the viral capsid, reducing recognition by pre-existing NAbs.
- Enhancing binding and internalization in refractory cell types through high-affinity antibody-ligand interactions.
- Reducing immunogenicity and off-target toxicity, enabling safe repeat dosing.
This transformative approach significantly improves the clinical translatability of adenoviral vectors, increasing therapeutic index, efficacy, and patient eligibility.
Strategies for Antibody-modified Ad Vector Construction
The strategies for the construction of antibody-modified Ad vectors are divided into two types: covalent or noncovalent attachment of targeting antibodies to the capsid and genetic modification of capsid proteins. Genetic modification involves the cloning of the targeting antibody fragment directly into the virion coat proteins. For instance, Poulin et al. (2010) reported genetic fusion of a single-chain antibody (scFv) or single-domain antibody (sdAb) to capsid protein IX (pIX). The results showed that sdAb can be displayed on the pIX to achieve enhanced virus infection of cells expressing the targeted receptor. Besides, Kaliberov et al. (2014) genetically incorporated an anti-carcinoembryonic antigen (CEA) sdAb into a de-knobbed Ad5 fiber-fibritin chimera and demonstrated selective targeting to the cognate epitope expressed on the membrane surface of target cells.
On the other hand, covalent and noncovalent methods involve the addition of targeting antibodies after the virus has been purified, through the use of bispecific antibodies (one binding the Ad virion and the other binding the desired cellular ligand) or antibody-receptor ligand complexes. For instance, Korn et al. (2004) generated two recombinant bispecific antibody molecules for the retargeting of adenoviral vectors to CEA-expressing tumor cells. Besides, Glasgow et al. (2009) reported the formation of an scFv-targeted Ad vector via highly specific association of the Ad virion and a targeting scFv employing synthetic leucine zipper-like dimerization domains. The results showed that zipper-tagged virions and scFv provided target-specific gene transfer. Additionally, studies have also reported that an IgG Fc-binding motif can be genetically incorporated into the capsid to bind monoclonal antibody for selective gene delivery.
Core Technical Principles of Antibody-Modified Adenoviral Vectors
Core Modification Concept
Antibody-modified adenoviral vectors are engineered by conjugating antibodies or antibody fragments (e.g., scFv, Fab, nanobodies) to key capsid proteins via two primary strategies: genetic fusion modification or chemical conjugation modification. This engineering rewires the vector's tropism, enabling precise targeting to cells expressing the antigen of interest while abrogating native CAR binding.
Key Modification Sites (With Schematic Diagram)
The adenoviral capsid comprises three major proteins, each offering unique modification opportunities (as shown in Figure 1):
- Fiber Protein: The most widely used modification site. The fiber's knob domain mediates native CAR binding; inserting antibodies/fragments here disrupts CAR interaction and redirects tropism. This site is ideal for targeting due to its extracellular exposure and role in initial cell attachment.
- Hexon Protein: The most abundant capsid protein (240 copies per virion) with hypervariable regions (HVRs) that are major targets for NAbs. Modifying HVRs with antibodies masks immunogenic epitopes while enabling targeted binding, dual-functionalizing the vector for reduced immunity and enhanced targeting.
- Penton Base Protein: Facilitates cell internalization via integrin binding (RGD motif). Antibody conjugation here enhances endocytosis in target cells, complementing tropism redirection from fiber modifications.
Our Custom Antibody-Modified Adenovirus Vector Construction Services
Personalized Strategy Design
Our process begins with a collaborative consultation to ensure your project's success.
- Target & Antibody Selection: We work with you to identify the optimal target antigen for your disease model (oncology, hepatology, vaccinology, etc.). We advise on the best antibody format—single-chain variable fragment (scFv), antigen-binding fragment (Fab), or single-domain antibody (nanobody/VHH)—based on your target, stability needs, and production capabilities.
- Modification Strategy Customization: Based on your application (e.g., systemic in vivo delivery vs. ex vivo cell transduction), we help you select the optimal modification site (Fiber, Hexon, Penton) and the most suitable technical path (Genetic Fusion or Chemical Conjugation).
- Adenovirus Backbone Selection: We offer a wide range of adenovirus backbones, including E1/E3-deleted (replication-deficient), conditionally replicating oncolytic Ads, and high-capacity "gutless" Ads (HCAdV) for large or multiple genes.
- Feasibility Assessment: Our team provides a free, no-obligation technical assessment of your project, outlining potential challenges and delivering a detailed design report with a clear project timeline.
Recombinant Adenoviral Vector Construction
Our vector construction process ensures high fidelity and functionality:
Adenoviral Genome Modification: Utilize optimized tools for precise engineering:
- Homologous recombination in E. coli (AdEasy system) for efficient genome assembly.
- CRISPR/Cas9-mediated insertion for site-specific modification of capsid genes.
- Codon optimization of antibody genes for enhanced expression in packaging cells.
Shuttle Vector and Packaging Plasmid Construction:
- Cloning of fusion constructs into shuttle vectors (pAdTrack-CMV, pShuttle-CMV).
- Preparation of packaging plasmids (pAdEasy-1, pHelper) for replication-defective vectors.
- Sequencing validation (Sanger/NGS) of all constructs to ensure no mutations or frame shifts.
Mutant Screening and Optimization:
- Transient transfection of 293A cells to assess capsid protein expression and vector assembly.
- Screening for stable recombinant clones with optimal antibody display and viral titers.
- Iterative optimization of linker sequences or modification sites to improve vector stability.
High-Titer Virus Packaging and Purification
| Service Stage | Specific Content |
|---|---|
| Cell Line Selection |
- Standard: 293A/293T cells (stable E1A/E1B expression, high adenovirus yield) - Custom: Alternative cell lines (e.g., PER.C6) for serotype-specific packaging or reduced immunogenicity |
| Virus Amplification & Titer Enhancement |
- Optimized transfection/amplification protocols - Scale-up from T-25 flasks to 10-layer cell factories/bioreactors - Titer optimization via process refinement |
| Multi-Level Purification |
- Research-grade: Cesium chloride (CsCl) density gradient ultracentrifugation - Preclinical-grade: Anion-exchange chromatography (AEX) + Size-exclusion chromatography (SEC) |
| Purity & Titer Characterization |
- Titer determination: TCID50 (50% Tissue Culture Infectious Dose), Plaque assay - Purity analysis: A260/A280 ratio (1.2-1.4), SDS-PAGE, Western blot - Endotoxin testing: LAL (Limulus Amebocyte Lysate) assay - Empty/full capsid ratio: HPLC or electron microscopy |
Comprehensive Functional Validation and Analysis
| Validation Type | Test Items | Test Methods/Indicators |
|---|---|---|
| In Vitro Validation |
1. Transduction efficiency detection (target vs. non-target cells) 2. Targeting specificity validation 3. Antibody conjugation efficiency detection 4. In vitro vector activity analysis |
1. Flow cytometry/fluorescence microscopy (GFP/Luciferase expression quantification) 2. Transduction ratio (target cells/non-target cells) 3. Western blot/ELISA (antibody display quantification) 4. - Oncolytic vectors: CCK-8/MTT cell viability assay - Gene therapy vectors: qPCR/Western blot (transgene expression) - Vaccine vectors: MHC-I/II staining (antigen presentation in DC cells) |
| In Vivo Validation |
1. Tissue distribution detection 2. Targeting specificity validation 3. In vivo transduction efficiency & bioactivity analysis |
1. Bioluminescence imaging (BLI)/qPCR (vector accumulation in target/off-target tissues) 2. Immunohistochemistry (IHC) (vector localization in target cells) 3. - Therapeutic efficacy: Tumor growth inhibition (xenograft models)/disease phenotype correction (genetic disease models) - Vaccine efficacy: ELISA (antibody titer)/ELISPOT (T cell response) |
| Safety Validation |
1. In vitro cytotoxicity 2. In vivo acute toxicity 3. Immunogenicity testing 4. Vector genome integrity detection |
1. LDH release assay (normal cell damage assessment) 2. Body weight monitoring, clinical signs observation, organ histopathology (mice/rats) 3. Neutralizing antibody (NAb) titer assay, cytokine profiling (IL-6, TNF-α) 4. PCR/NGS (no unintended genome rearrangements) |
| Stability Validation |
1. Storage stability testing 2. Freeze-thaw stability testing 3. Formulation optimization |
1. Titer/activity testing after storage at -80°C/-20°C/4°C/RT for 1/3/6/12 months 2. Titer/activity assessment after 3-5 freeze-thaw cycles 3. Recommend optimal excipients (e.g., sucrose, HEPES) for long-term storage |
Customized Delivery Package
You receive a complete, ready-to-use package tailored to your project:
- Recombinant adenoviral vector plasmids (with full sequencing reports).
- High-titer purified virus (volume: 0.5-10 mL, depending on your needs) in research-grade/preclinical-grade/GMP-grade formulation.
-
Comprehensive characterization reports:
- Vector construction and sequencing data.
- Purification and titer analysis (TCID50, A260/A280, endotoxin).
- Functional validation (in vitro/in vivo activity, targeting specificity).
- Safety and stability test results.
-
Detailed documentation:
- Experimental protocols (vector construction, packaging, purification).
- Standard operating procedures (SOPs) for virus handling and use.
- Material Safety Data Sheet (MSDS).
-
Post-delivery technical support:
- Guidance on virus thawing, dilution, and administration.
- Troubleshooting for transduction or animal experiments.
- Recommendations for vector amplification or scale-up.
Why Creative Biolabs?
| Feature | Our Advantage |
|---|---|
| Extensive capsid engineering expertise | Many years' experience in adenovirus vector engineering |
| Multiple targeting strategies | Fiber / Hexon / Penton modification platforms |
| Antibody engineering integration | scFv, Fab, VHH antibody optimization |
| High-titer production | Up to 10¹³ IU/mL |
| Full validation pipeline | In vitro and in vivo functional testing |
Core Application Areas
Our antibody-modified adenoviral vectors enable breakthroughs in multiple therapeutic areas:
Cancer Gene Therapy
- Targeted Oncolytic Adenoviruses: Constructed to selectively replicate in and kill cancer cells (e.g., HER2-positive breast cancer, EGFR-positive lung cancer, AFP-positive hepatocellular carcinoma). Antibody modification enhances tumor penetration and reduces systemic toxicity.
- Therapeutic Gene Delivery: Targeted delivery of tumor suppressor genes (p53), suicide genes (HSV-tk), or immunomodulatory genes (IL-12, PD-L1 inhibitor) to cancer cells, improving efficacy while sparing healthy tissues.
Liver Disease Targeted Therapy
- For hepatitis B/C, cirrhosis, and hepatocellular carcinoma, we target hepatocyte-specific antigens (e.g., ASGPR, CD81) to deliver therapeutic genes (e.g., antiviral proteins, regenerative factors) or oncolytic vectors. This minimizes off-target effects on the liver parenchyma.
Targeted Vaccine Development
- Antibody-modified vectors deliver vaccine antigens (e.g., viral antigens, tumor neoantigens) to professional antigen-presenting cells (APCs) such as dendritic cells (DCs) via CD11c or DEC-205 targeting. This enhances antigen presentation, leading to 2-3-fold higher antibody titers and stronger cellular immune responses compared to unmodified vaccines.
Rare Disease Gene Therapy
- For monogenic rare diseases (e.g., cystic fibrosis, muscular dystrophy, hemophilia), we target disease-relevant cell types (e.g., airway epithelial cells, muscle cells, hepatocytes) to deliver functional copies of defective genes. Antibody modification ensures efficient transduction of rare or refractory target cells.
Frequently Asked Questions
Q: Will antibody-modified adenoviral vectors maintain consistent transduction efficiency across different batches?
A: Consistency is a core focus of our quality control system. We ensure batch-to-batch reproducibility through:
- Standardized manufacturing protocols (SOPs) for vector construction, packaging, and purification.
- Rigorous in vitro transduction efficiency testing (e.g., flow cytometry for GFP expression) for each batch, with a target coefficient of variation (CV) <15%.
- Batch matching of key parameters (antibody conjugation efficiency, viral titer, capsid integrity) against reference standards.
For preclinical vectors, we also conduct parallel testing across batches using validated cell lines and animal models to confirm consistent bioactivity.
Q: How do you select the optimal antibody type (scFv/Fab/nanobody) for different application scenarios?
A: Antibody selection is tailored to your specific use case, based on three key factors:
| Application Scenario | Recommended Antibody Type | Rationale |
|---|---|---|
| Tumor gene therapy (systemic delivery) | Nanobody/scFv | Small size (~15-27 kDa) enables better tumor penetration; low immunogenicity. |
| Vaccine development (DC cell targeting) | Fab/scFv | Balances affinity and stability; Fab's bivalent binding enhances DC cell engagement. |
| Liver disease therapy (hepatocyte targeting) | Fab/nanobody | Nanobodies bind ASGPR with high specificity; Fab offers better serum stability. |
| Repeat dosing requirements | Nanobody | Minimal immunogenicity reduces anti-antibody responses during multiple administrations. |
Q: Do you provide large-scale production of antibody-modified adenoviral vectors for clinical trials?
A: Yes. We offer scalable production capabilities to meet clinical trial demands:
- Preclinical scale: 10-100 mL (10¹⁰-10¹² IU total) for IND-enabling studies.
Q: What methods are used to confirm successful antibody conjugation to the viral capsid, beyond Western blot/ELISA?
A: We offer multiple orthogonal validation methods to verify antibody-capsid conjugation:
- Flow Cytometry-Based Binding Assay: Incubate modified vectors with target antigen-expressing cells; detect bound vectors using anti-antibody fluorochromes to confirm functional conjugation.
- Mass Spectrometry (MS): Quantify the molar ratio of antibody to capsid protein (e.g., Fiber/Hexon) by LC-MS/MS, ensuring consistent conjugation efficiency (~2-5 antibodies per capsid).
- Transmission Electron Microscopy (TEM): Visualize antibody conjugation via immunogold labeling (antibody-specific gold particles), confirming spatial distribution on the capsid surface.
- Surface Plasmon Resonance (SPR): Measure binding affinity between modified vectors and soluble target antigens, validating that antibody conjugation does not compromise antigen recognition.
Partner with Us
The future of gene therapy lies in precision. By partnering with us for your antibody-modified adenovirus vector needs, you are choosing a path beyond the limitations of conventional viral vectors. We empower your research with the tools to achieve unprecedented targeting specificity, evade immune barriers, and unlock the full therapeutic potential of your gene-based medicine. Contact us today to start designing your precision-targeted vector.
Reference
- Chavda V P, Bezbaruah R, Valu D, et al. Adenoviral vector-based vaccine platform for COVID-19: current status. Vaccines, 2023, 11(2): 432. https://doi.org/10.3390/vaccines11020432 Distributed under Open Access license https://creativecommons.org/licenses/by/4.0/deed, without modification.
