AAV Vector Design for Batten Disease
Recombinant adeno-associated virus (rAAV) vectors are excellent tools in gene therapy for the treatment of neurological diseases due to their ability to efficiently transduce neurons and provide long-term expression. Creative Biolabs is an excellent service provider of AAV vectors, providing a wide range of AAV vector design services to meet your basic research and clinical development needs.
Introduction of Batten Disease
Batten disease (also called neuronal ceroid lipofuscinoses [NCLs]) are a group of fatal, inherited lysosomal storage disorders mostly affecting the central nervous system of children. This family of diseases is caused by mutations in approximately 13 known ceroid-lipofuscinosis neuronal (CLN)-related genes, designated CLN1-CLN8 and CLN10-CLN14. Despite their genetic diversity, NCLs are defined by similar pathological and clinical features including progressive neuronal loss, retinal degeneration, seizures, and psychomotor decline, which ultimately lead to premature death. The age of disease onset and disease progression vary considerably depending on the genetic variant. In general, Batten disease is the most prevalent cause of neurodegenerative disease in children, with an incidence of 2-4/100,000 live births. There are no effective treatments other than approved enzyme replacement therapy for CLN2 disease. In recent years, gene therapy has made great progress in NCLs and has become a very attractive strategy for the treatment of NCLs.
Gene Therapy Strategies for Batten Disease
1. Gene Replacement Therapy
The most advanced approach involves delivering a functional copy of the mutated CLN gene using viral vectors such as AAV.
- Restores lysosomal enzyme activity
- Reduces intracellular waste accumulation
- Slows or halts neurodegeneration
This approach has shown promising results in preclinical and clinical studies targeting CLN1, CLN2, CLN3, and CLN6.
2. CNS-Targeted AAV Delivery
Efficient delivery to the brain is critical. We optimize:
- AAV serotype selection (e.g., AAV9 for CNS tropism)
- Intrathecal, intracerebroventricular, or intravenous delivery routes
- Neuron-specific promoter design
Preclinical studies demonstrate that AAV-mediated gene therapy can significantly improve neurological outcomes in Batten disease models.
3. Antisense and RNA Modulation Approaches
For certain mutations, RNA-level interventions can restore protein function:
- Splice correction
- Premature stop codon bypass
- Gene expression modulation
These approaches complement gene replacement strategies.
4. Emerging Gene Editing Strategies
We support exploratory development of:
- CRISPR/Cas-based correction
- Base editing for CLN mutations
- Long-term genomic repair strategies
These technologies aim to provide permanent correction of disease-causing mutations.
Key Challenges in Batten Disease Gene Therapy - And How We Solve Them
| Challenges | Creative Biolabs Solutions |
|---|---|
| Blood-brain barrier (BBB) | Intrathecal (IT), intracerebroventricular (ICV), or intracranial injection protocols with AAV9, AAVrh.10, and engineered BBB-penetrant capsids. |
| Large transgene size (e.g., CLN3, CLN5 >4.5 kb) | Dual AAV (trans-splicing or overlapping) and hybrid nanoparticle strategies. |
| Pre-existing immunity to AAV | Neutralizing antibody screening and capsid serotype switching; optional immunomodulation co-treatment. |
| Lack of translatable animal models | Access to 8+ NCL mouse models and naturally occurring ovine/canine NCL models. |
| Long-term expression in post-mitotic neurons | Optimized promoters and validated AAV episomal persistence for >12 months. |
AAV-based Gene Therapy for Batten Disease
In the past few decades, AAV has been developed as a promising non-pathogenic gene therapy vector for the treatment of neurodegenerative diseases in the CNS. The development of different serotypes and genetic targets for different cells could assist in the curing of neurodegenerative diseases from various causes. Much progress has been made in the development of gene therapy for the NCLs, particularly the emergence of more potent AAV serotypes that are capable of crossing the blood-brain barrier and may result in significant clinical advances in the near future.
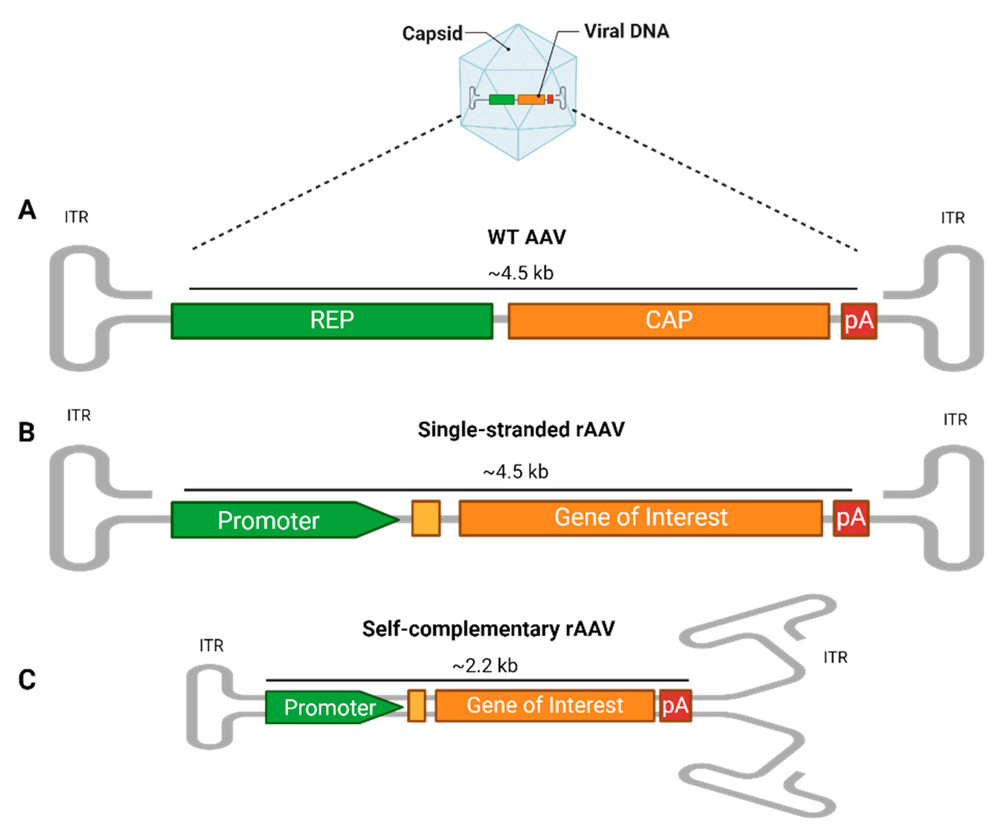 Figure 1. AAV vector structure.1
Figure 1. AAV vector structure.1
Recently, AAV (such as AAV1/2) vectors expressing the CLN2 cDNA and self-complementary AAV serotype 9 (scAAV9) expressing CLN3, CLN5 and CLN6 gene have been developed to deliver genes to the brain of children. In addition, AAVrh.10 vector expressing CLN2 and CLN3 is also used in gene therapy for NCLs. These rAAV vectors have been shown to slow or prevent the progression of disease in the CNS, supporting the great potential of AAV-mediated gene therapy approaches in the treatment of NCLs.
Our Custom Services for AAV-based Gene Therapy
The rational design of AAV vectors is critical to achieving effective gene therapy for NCLs. Creative Biolabs has extensive experience in AAV vector design and can provide customized AAV vector design services based on customer's specific needs. In the gene therapy for NCLs, we provide tissue/cell-specific targeting AAV service, AAV vector design for gene expression, titration service of AAV vector, AAV vector purification service and self-complementary AAV vector service to achieve efficient delivery of therapeutic genes by rAAV vectors. At Creative Biolabs, our rAAV vectors exhibit the following advantages:
- Do not elicit significant immune responses in vivo
- Broad tropism - tissue specificity with different AAV serotypes
- Do not integrate into the host genome
- Can achieve stable and efficient expression of the target gene
Scalable, High-Purity AAV Packaging and Production
Transitioning from a promising plasmid construct to a high-titer, ultra-pure viral vector is a highly complex biomanufacturing challenge. At Creative Biolabs, our viral vector core is equipped with state-of-the-art facilities capable of supporting your project from research-grade micro-scale production up to large-scale, pre-clinical/GLP-like manufacturing.
Advanced Cell Line & Transfection Technologies
We utilize optimized, suspension-adapted HEK293T cell lines cultured in serum-free media to produce AAVs. Our proprietary triple-plasmid transient transfection protocols ensure high packaging efficiency, minimizing the generation of empty viral capsids that can trigger unwanted immune responses without delivering therapeutic benefit.
Stringent Downstream Purification
CNS-directed gene therapies require the highest possible purity to prevent severe neuroinflammatory responses following direct brain or intrathecal administration. Our downstream purification processes are uncompromising:
- Ultracentrifugation: Iodixanol or Cesium Chloride (CsCl) gradient ultracentrifugation for robust separation of full from empty capsids.
- Chromatography: Advanced Affinity Chromatography and Ion Exchange Chromatography (IEX) to achieve ultra-high purity profiles, eliminating host cell proteins (HCP), host cell DNA (HCD), and endotoxins.
In Vitro & Ex Vivo Validation Models
Before moving to expensive animal models, we validate your customized AAV vectors using advanced cellular assays:
- Primary Neuronal Cultures: Transduction of primary cortical or hippocampal neurons to verify neuron-specific promoter activity and assess cellular toxicity.
- Patient-Derived iPSC Models: We offer services to generate Induced Pluripotent Stem Cells (iPSCs) from patients with specific CLN mutations, differentiate them into neuronal lineages, and utilize these exact disease models to test your AAV's ability to clear lipofuscin accumulation, restore lysosomal enzyme activity, and rescue cellular phenotypes.
In Vivo Efficacy Studies in Batten Disease Models
We support robust in vivo proof-of-concept studies utilizing established murine models of Batten disease (e.g., Cln1-/-, Cln2-/-, Cln3-/-, Cln6- deficient mouse models). Our specialized neuroscience preclinical team executes complex study designs, including:
- Diverse Administration Routes: Expertise in stereotaxic Intraparenchymal injection, Intracerebroventricular (ICV), Intrathecal (IT), Intracisternal (ICM), and systemic Intravenous (IV) administrations tailored to mimic potential clinical delivery routes.
- Biodistribution & Transgene Expression Profiling: Utilizing highly sensitive ddPCR, RT-qPCR, and immunohistochemistry (IHC) to map vector genomes and protein expression comprehensively across the brain, spinal cord, and peripheral organs.
- Neuropathological Rescue Assessment: Advanced histological analysis to quantify the reduction of autofluorescent storage material (ceroid/lipofuscin), mitigation of astrogliosis (GFAP) and microgliosis (Iba1), and prevention of neuronal loss in critical brain regions (e.g., cortex, thalamus, cerebellum).
- Behavioral & Motor Function Assays: Long-term longitudinal studies evaluating the prevention of disease progression through rotarod, open field tests, and specific cognitive behavioral assessments.
Why Batten Disease Researchers Choose Creative Biolabs
- Rapid turnaround – Fast vector cloning and short experimental timelines for pilot efficacy studies.
- Rare disease focus – Dedicated team for NCLs with 20+ completed gene therapy projects for CLN2, CLN3, CLN6, CLN7.
- High-quality data – All in vivo studies follow ARRIVE guidelines; histology and biochemical readouts are validated and auditable.
- Transparent pricing – Fixed-price packages for feasibility, vector production, and GLP toxicology.
Frequently Asked Questions (FAQ)
Q: Which AAV serotype is best for my Batten disease project?
A: The "best" serotype depends entirely on your specific CLN target and intended route of administration. For global systemic delivery targeting the CNS in early-life models, AAV9 is highly effective. For direct CNS administration (ICV/IT), AAVrh.10 and AAV9 both show exceptional distribution. Our experts provide data-driven recommendations during your initial consultation.
Q: Why should I choose scAAV over ssAAV for Batten disease?
A: Many CLN genes are small enough to fit into a self-complementary AAV (scAAV) format (capacity < 2.2 kb). scAAVs do not require second-strand DNA synthesis upon entering the nucleus, leading to much faster and significantly stronger gene expression. For rapidly progressing neurodegenerative diseases like NCLs, this rapid onset of action can be critical for preserving neuronal tissue.
Q: Can you help us design an AAV that avoids liver toxicity?
A: Yes. We address off-target expression (such as hepatotoxicity, which has been observed in some high-dose systemic AAV applications) by utilizing CNS-restricted, neuron-specific promoters (like Synapsin I) and incorporating microRNA target sequences (e.g., miR-122 binding sites) into the 3' UTR of the transgene cassette to actively suppress expression in the liver.
Q: Can you help with patient-derived iPSC models for efficacy screening?
A: Absolutely. We differentiate patient iPSCs into cortical neurons and evaluate vector-mediated enzyme restoration, reduction of lipofuscin, and electrophysiological rescue using multi-electrode arrays.
Q: How do you handle large genes like CLN3 (>4.5 kb) for AAV packaging?
A: We apply dual AAV vector strategies (trans-splicing or overlapping) to reconstitute full-length protein. Alternatively, we offer lentiviral or non-viral nanoparticles for larger transgenes. Recent data from our CLN3 program show successful expression using hybrid AAV/DNA nanoparticles.
Get in Touch – Accelerate Your Batten Disease Program
Whether you are validating a novel CLN gene target, struggling with poor transduction efficiency in your current vectors, or require an experienced CRO to conduct rigorous in vivo efficacy studies, Creative Biolabs is ready to accelerate your program. Our experienced technical experts provide custom services for each step of the AAV vectors design process. For more details, please contact us and we will be happy to assist you.
Reference
- Zwi-Dantsis L, Mohamed S, Massaro G, et al. Adeno-associated virus vectors: principles, practices, and prospects in gene therapy. Viruses, 2025, 17(2): 239. https://doi.org/10.3390/v17020239 Distributed under Open Access license CC BY 4.0, without modification.
