Lentiviral Vector Development for X-linked Chronic Granulomatous Disease
Creative Biolabs' highly efficient and rigorous team of scientists is ready to provide professional technical support for your research. By continuously improving our research and development capabilities, we have established the world's leading lentiviral vector technology platform, which can be used to develop specific vectors based on the treatment needs of different diseases. The gene therapy of X-linked chronic granulomatous disease (CGD) has always been one of the areas we valued. No matter what difficulties you have encountered in the relevant research work, you can contact us to get your own customized service.
Introduction of X-linked Chronic Granulomatous Disease
GCD is a rare genetically heterogeneous primary immunodeficiency disease caused by defects in the nicotinamide adenine dinucleotide phosphate (NADPH) oxidase complex. This disease can cause the patient's phagocytic cells do not normally produce superoxide and mediate the killing of intracellular microorganisms. And patient is more susceptible to chronic inflammation and bacterial or fungal infection.
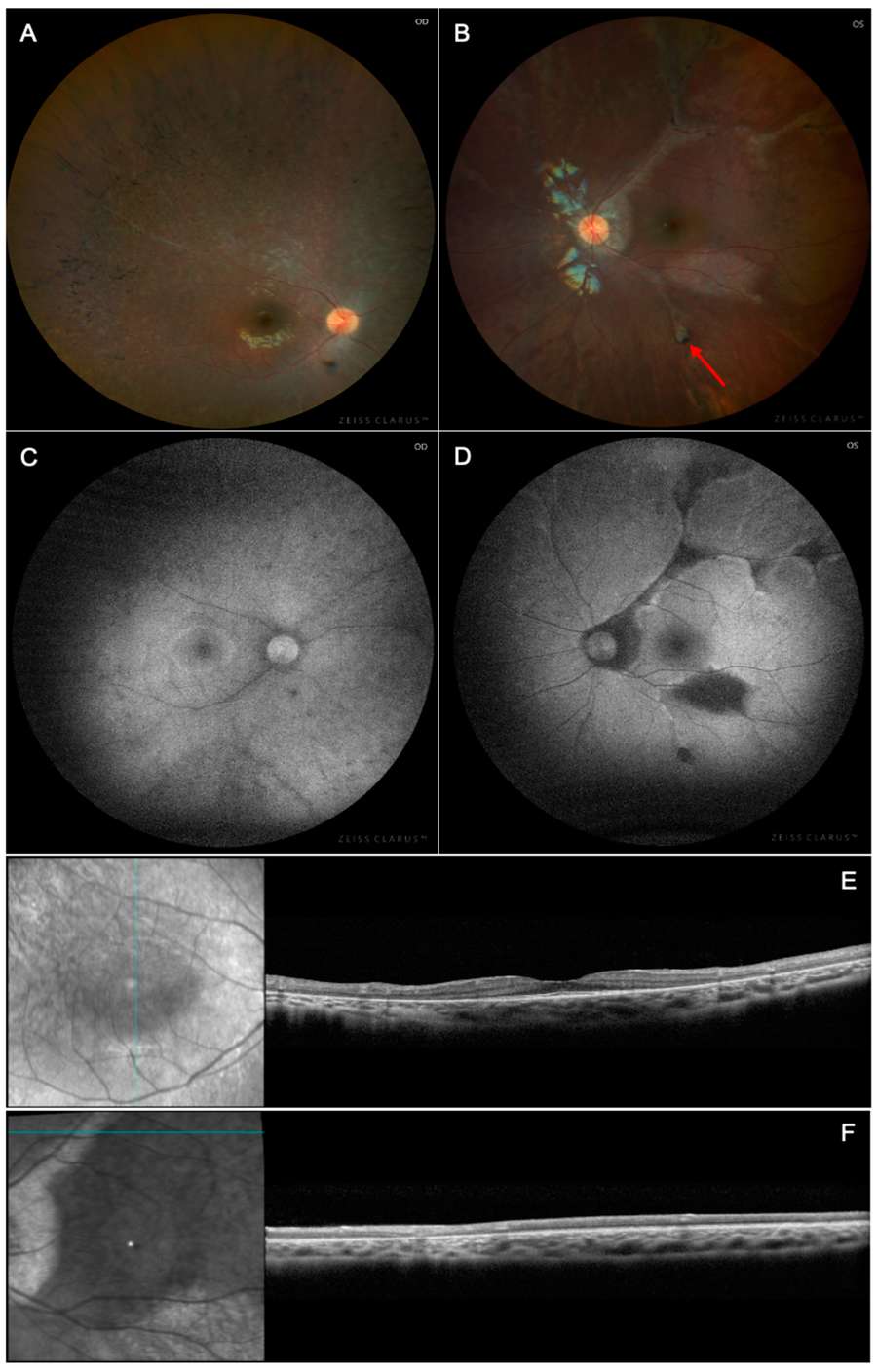 Figure 1. Multimodal imaging of X-linked chronic granulomatous disease chorioretinitis.1
Figure 1. Multimodal imaging of X-linked chronic granulomatous disease chorioretinitis.1
Among all the mutations that cause CGD, the most common is X-linked CGD caused by mutations in the CYBB gene, which accounts for more than 70% of the overall diseases. This gene encodes the key transmembrane protein gp91-phox in the NADPH oxidase complex. To date, there are more than 700 different CYBB gene mutations recorded in the database. The treatment method of CGD is mainly based on antibiotics and antifungal drugs. Bone marrow transplantation (BMT) can cure CGD, however, this method has higher requirements for transplant recipients and donors, and this specific process of treatment is relatively complicated, moreover, the costs and risks make it cannot be easily universalized. Therefore, the development of gene therapy technology will make it the first choice for the treatment of CGD and other related gene defects in the future.
Gene Therapy for X-linked Chronic Granulomatous Disease
In-depth research in molecular biology has led us to a more comprehensive understanding of the CYBB gene encoding the gp91-phox subunit, making it possible to cure X-linked CGD by gene therapy. The specific treatment program is to extract some blood-making cells from the patient using a technique called apheresis, and then uses a special virus-retrovirus to transfect the normal CYBB gene into the cells, so that these blood-making cells can produce normal gp91-phox subunit. In this process, treated patients require a small dose of busulfan chemotherapy, but the risk is much lower than traditional allogeneic BMT treatment. Finally, when we get the genetically repaired cells, we can re-implant them into the patient.
Lentiviral Vector Development for Gene Therapy
The emergence of lentiviral vector technology and its successful application in the field of gene therapy is a breakthrough in human life and health. The ability of this vector to infect undifferentiated or terminally differentiated cells greatly expands the target of disease that can be used for gene therapy. In addition, lentiviral vectors increase the efficiency of gene transfer to target cells, reduce the difficulty of the transfection process, and reduce the need for in vitro cell manipulation, making the gene therapy safer and more efficient. Therefore, the development and optimization of lentiviral vectors will become a technical means that needs to be paid more attention for gene therapy in the future.
Our Advanced Lentiviral Vector Technology Platform
Traditional retroviral vectors used in early CGD trials were often associated with insertional mutagenesis. Our next-generation Self-Inactivating (SIN) Lentiviral Vectors are engineered to maximize safety while ensuring robust expression in the myeloid lineage.
Key Features of Our Platform:
- Safety-First Design: We utilize 3rd and 4th generation SIN-LVV systems that eliminate the risk of replication-competent lentivirus (RCL) and minimize transactivation of neighboring oncogenes.
- Lineage-Specific Promoters: To prevent ectopic expression and exhaustion of stem cells, we offer customized myeloid-specific promoters (e.g., Cathepsin G, c-fes, or chimeric promoters) that restrict CYBB expression to mature neutrophils and monocytes.
- High Functional Titers: Our optimized production protocols ensure ultra-high titers, essential for efficient transduction of "hard-to-transduce" primary CD34+ cells.
- Codon Optimization: We apply proprietary algorithms to optimize the CYBB transgene sequence, enhancing mRNA stability and protein translation efficiency in human cells.
Our Comprehensive X-CGD Lentiviral Vector Service
We provide a seamless continuum of services, from initial vector design to GMP manufacturing and regulatory support. Our team of virologists, molecular biologists, and process scientists will work collaboratively with you at every stage.
Custom Vector Design and Optimization Service: A Detailed Breakdown
| Service Module | Specific Service | Description & Key Benefits for X-CGD Programs |
|---|---|---|
| Gene Optimization | Codon Optimization of CYBB |
Description: Redesign of the CYBB coding sequence to match the tRNA abundance in human hematopoietic stem cells (HSCs) and phagocytes without altering the amino acid sequence. Benefit: Enhances translational efficiency, increases protein yield of gp91^phox^, and ensures stable, high-level expression in target cells. |
| Promoter Engineering | Myeloid-Specific Promoter Selection |
Description: Utilization of promoters (e.g., mimetic of the endogenous CYBB promoter, synthetic myeloid promoters) to drive gp91^phox^ expression specifically in neutrophils, monocytes, and macrophages. Benefit: Mimics physiological expression patterns, potentially improving safety by reducing expression in non-target cells and enhancing efficacy by focusing the therapeutic protein where it is needed most. |
| Constitutive Promoter Selection |
Description: Incorporation of strong, ubiquitous promoters for broad and high-level transgene expression. Benefit: May be suitable for specific research or preclinical applications requiring maximal expression, though with trade-offs in cell-type specificity. |
|
| Vector Architecture | Self-Inactivating (SIN) Vector Design |
Description: Engineering the vector with a deletion in the U3 region of the 3' long terminal repeat (LTR). This is transferred to the 5' LTR during reverse transcription, inactivating viral promoter/enhancer activity in the integrated provirus. Benefit: Drastically reduces the risk of insertional activation of proto-oncogenes, representing a key safety feature for clinical-grade vectors. |
| 4th Generation Backbone |
Description: Use of advanced, split-genome packaging systems to enhance safety by minimizing the potential for recombination and generation of replication-competent lentivirus (RCL). Benefit: Provides the highest level of biosafety for your therapeutic vector. |
|
| Advanced Functional Elements | Inclusion of Chromatin Insulators |
Description: Incorporating genetic elements (like cHS4) that act as barriers to prevent interference from nearby chromosomal elements and reduce position effect variegation. Benefit: Promotes more consistent, stable, and long-term transgene expression, protecting against gene silencing over time. |
| Woodchuck Hepatitis Virus Posttranscriptional Regulatory Element (WPRE) |
Description: Adding WPRE to the construct to enhance polyadenylation and nuclear export of the CYBB mRNA transcript. Benefit: Significantly boosts transgene expression levels, potentially allowing for lower vector doses and improved efficacy. |
|
| Tracking/Selection Markers |
Description: Co-expression of marker genes (e.g., truncated CD34, NGFR, GFP) via an internal ribosome entry site (IRES) or a 2A peptide sequence. Benefit: Enables easy tracking of transduced cells in vitro and in vivo, facilitates the enrichment of successfully transduced HSCs prior to infusion, and aids in preclinical biodistribution studies. |
High-Titer Vector Production and Characterization
Producing high-quality, high-titer vector is the cornerstone of a successful gene therapy program. Our advanced production platforms ensure you have the material you need, when you need it.
- Scalable Transfection Systems: We utilize both adherent and suspension cell culture systems for transient transfection, allowing us to produce research-grade and preclinical vectors rapidly. Our processes are optimized for maximum yield of functional, full-length vector particles.
-
Comprehensive Characterization: We provide full characterization of your vector, including:
- Physical Titer: p24 ELISA or qPCR/ddPCR for viral RNA.
- Functional Titer: Transduction efficiency on relevant cell lines and primary HSCs, measured by flow cytometry or qPCR.
- Identity and Purity: Assessment of residual host cell DNA, plasmid DNA, and BSA.
- Potency (Critical for X-CGD): We can develop and perform functional potency assays to confirm that your vector-transduced cells exhibit restored NADPH oxidase activity, measured by standard assays like the DHR (dihydrorhodamine-123) flow cytometry test.
Rigorous Quality Control and Validation
Success in X-CGD therapy depends on the precision of the vector. Our validation suite includes:
I. Identity and Purity
- Physical Titer (p24 ELISA) and Functional Titer (qPCR/ddPCR).
- Aggregation Analysis and Residual Impurity Testing (HCP, HCD).
II. Functional Potency Assays (The Gold Standard)
- DHR (Dihydrorhodamine 123) Assay: We test the ability of transduced CGD-patient-derived cells to produce superoxide upon stimulation.
- NBT (Nitroblue Tetrazolium) Test: Visual confirmation of restored NADPH oxidase activity.
- Bactericidal Activity: In vitro assays to confirm that corrected neutrophils can effectively kill pathogens like Staphylococcus aureus or Aspergillus.
III. Safety Assessment
- VCN (Vector Copy Number) Analysis: Ensuring optimized transduction (typically 1-3 copies per cell) to balance efficacy and safety.
- Integration Site Analysis (ISA): Utilizing Next-Generation Sequencing (NGS) to map integration sites and monitor for clonal expansion.
- RCL Testing: Ensuring the final product is free of replication-competent viruses.
Why Choose Creative Biolabs for Your X-CGD Research?
X-CGD is a rare disease with a small patient population, but it demands the same level of scientific rigor, technical innovation, and regulatory compliance as any other gene therapy program. At Creative Biolabs, we are committed to advancing X-CGD gene therapy by providing the highest-quality lentiviral vector solutions – and we bring three key strengths to every project:
- Specialization: X-CGD is a core focus of our gene therapy research, and we have developed a deep understanding of the unique challenges of CYBB gene delivery and expression in hematopoietic cells.
- Innovation: We continuously invest in R&D to refine our lentiviral vector technology, incorporating the latest advances in bioinformatics, synthetic biology, and clinical gene therapy to deliver state-of-the-art vectors for X-CGD.
- Partnership: We view our clients as partners, and we work closely with your team to understand your research goals, address your technical challenges, and accelerate your path to success. Our goal is not just to deliver a vector – but to help you achieve your research milestones and bring curative therapies to X-CGD patients.
Frequently Asked Questions
Q: What makes developing a lentiviral vector for X-CGD uniquely challenging compared to other blood disorders?
A: The primary challenge lies in the biology of the disease. Unlike disorders where replacing a secreted protein or a simple enzyme is sufficient, X-CGD requires the restoration of a complex, multi-component enzyme system—the NADPH oxidase—in the correct cell type (phagocytes). The vector must not only efficiently deliver the CYBB gene into HSCs but also ensure that its expression is:
- Lineage-specific: High and functional in neutrophils, monocytes, and macrophages.
- Physiologically regulated: To avoid potential toxicity from over-expression.
- Functional: The resulting gp91^phox^ protein must correctly assemble with other components (p22^phox^, p47^phox^, etc.) to form a working oxidase. This makes a functional potency assay (like the DHR test on vector-transduced cells) a critical part of development, which is more complex than simply measuring protein levels.
Q: How do you ensure that your vectors will drive sufficient gp91^phox^ expression to restore neutrophil function?
A: We address this through a multi-pronged strategy during the design phase. First, we offer codon optimization of the CYBB gene to maximize translational efficiency in human cells. Second, we carefully select and test promoters. We can use potent constitutive promoters for maximum expression, or more sophisticated myeloid-specific promoters that are designed to drive high levels of expression precisely in phagocytes. Third, we can incorporate elements like the WPRE to boost mRNA stability and export. Ultimately, the final proof is in our potency testing services, where we transduce patient-derived or healthy donor CD34+ cells with your vector, differentiate them into neutrophils, and directly measure the restoration of ROS production using the DHR assay.
Q: What is your strategy for minimizing the risk of genotoxicity, a key concern for HSC gene therapies?
A: Safety is paramount in our designs. All our vectors for X-CGD are built on a 4th generation, self-inactivating (SIN) lentiviral backbone. The SIN design removes the viral enhancer/promoter sequences from the LTRs after integration, which significantly reduces the risk of activating nearby proto-oncogenes. Furthermore, our use of internal promoters (especially cell-specific ones) provides an additional layer of control over expression. For programs where the highest level of safety is desired, we can also explore the inclusion of chromatin insulators to shield the transgene from the influence of the surrounding genome and vice versa. Finally, we offer insertional site analysis (ISA) services to help you profile the integration pattern and clonal outgrowth in your preclinical models.
Q: My program is still in early preclinical stages. Can I start with a small-scale, research-grade vector from you and later scale it up for GMP production?
A: Absolutely. This is the most efficient path to the clinic. We offer a seamless, integrated pipeline. You can begin with our custom design and research-grade vector production services to test your construct in vitro and in small animal models. Once you have proof-of-concept data, we work with you to transition that exact construct and process to our process development and scale-up platforms. We have extensive experience in transferring processes, optimizing them for larger-scale production, and ultimately locking them down for technology transfer to our cGMP manufacturing suites. This continuity minimizes changes and de-risks the journey to IND.
Connect with Us Anytime!
Creative Biolabs provides lentiviral vectors for X-linked CGD gene therapy according to customer needs through rigorous quality control. We will optimize the vector to make it more genetically stable and can be permanently integrated into the genome of transduced cells to provide long-term stable gene expression in vivo or in vitro. If you have any questions or have any difficulties, you can contact us by email or send us an inquiry to find a complete solution.
Reference
- Bailey J A, Kong M D, Piamjitchol C, et al. X-Linked CGD Chorioretinitis in Two Young Girls. Biomedicines, 2025, 13(2): 323. https://doi.org/10.3390/biomedicines13020323 Distributed under Open Access license CC BY 4.0, without modification.
