Pseudotyping Services of Lentiviral Vectors with Rabies Virus
At Creative Biolabs, we focus on the design, production and validation of high-quality pseudotyped lentiviral vectors. Our comprehensive service platform combines the most advanced molecular virology with strict quality control, providing researchers with reliable and directly usable viral vectors that maintain stable performance in both in vitro and in vivo environments. Whether you are studying basic neurobiological mechanisms or developing next-generation therapeutic strategies, we can offer comprehensive design solutions to help you solve problems quickly and efficiently.
Pseudotyping Introduction
Pseudotyping as a cornerstone technique in modern virology and genetic engineering, enables the structural adjustment of viral vectors according to specific research and treatment requirements. By replacing the natural envelope protein of lentiviral vectors with the rabies virus glycoprotein (RV-G), researchers can utilize an efficient retrograde transport system to achieve a unique function, allowing these vectors to move retrogradely along the axons of neurons. Therefore, rabies virus phage-displayed lentiviral vectors (RV-LVs) have become an indispensable tool for mapping complex neural circuit diagrams, delivering genetic material to specific neuronal populations, and developing novel gene therapies for neurological diseases.
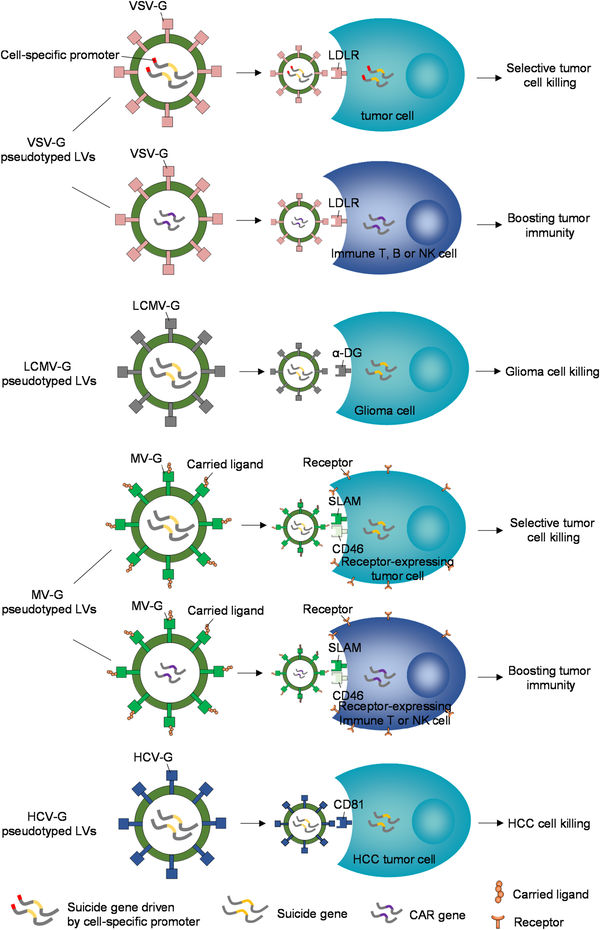 Figure.1 Applying pseudotyped LVs for cancer gene therapy.1
Figure.1 Applying pseudotyped LVs for cancer gene therapy.1
What are Rabies Virus (RV) Pseudotyped Lentiviral Vectors?
- Pseudotyping is the process of replacing the native viral envelope protein (typically the Vesicular Stomatitis Virus G protein, or VSV-G, for lentiviruses) with the envelope protein of a different virus. In this context, we integrate the Rabies Virus Glycoprotein (RV-G) into the HIV-1-based lentiviral membrane.
- Structure and Function of RV-G The RV-G is a class III viral fusion protein. It is the sole protein on the surface of the rabies virion, responsible for both receptor binding and pH-dependent membrane fusion.
- The Core Difference: RV-G vs. VSV-G While VSV-G is the "gold standard" for broad-spectrum cell transduction due to its interaction with the ubiquitous LDL receptor, it lacks tissue specificity. In contrast, RV-G confers neurotropism. While VSV-G pseudotypes primarily transduce the area around the injection site, RV-G pseudotypes excel at long-range transport.
Mechanism of Cell Targeting and Retrograde Transport
Receptor Recognition and Membrane Fusion Process
The acidic environment of the endosome induces a major conformational change in RV-G, exposing its fusion peptide. This peptide inserts into the endosomal membrane, driving the fusion of the viral envelope with the endosomal lipid bilayer, thereby releasing the lentiviral core (containing the RNA genome) into the neuronal cytoplasm.
Molecular Basis of Retrograde Axonal Transport
The cytoplasmic tail of RV-G contains motifs that interact with the dynein motor complex. Once the vector core is released in the axon terminal, these interactions facilitate the active, microtubule-dependent transport of the viral capsid in a retrograde direction—towards the neuronal cell body. This journey can span significant distances within the nervous system.
Analysis of Neuron-Specific Transduction Advantages
This two-step mechanism—specific receptor binding followed by retrograde transport—ensures that transduction is largely restricted to neurons and, more specifically, to those neurons synaptically connected to the injection site. It minimizes off-target transduction of glial cells or local interneurons that do not project to the area, providing exceptional anatomical precision.
Applications of Rabies Virus–Pseudotyped Lentiviral Vectors
- Neural Circuit Tracing and Functional Analysis: The gold-standard tool for monosynaptic retrograde tracing when combined with complementation systems.
- Retrograde Gene Delivery for CNS Research: Targeted delivery of therapeutic genes to specific neuronal populations affected in Parkinson's disease, epilepsy, or chronic pain.
- Neurodegenerative Disease Model Construction: Selective expression of disease-associated proteins in vulnerable, projection-defined neuronal populations.
- Brain Region-Specific Genetic Manipulation: Functional studies via retrograde delivery of optogenetic actuators or chemogenetic receptors.
Our Rabies Virus Pseudotyping Service Capabilities
At Creative Biolabs, we offer end-to-end expertise in the design, production, and validation of high-titer, research-grade RV-LVs. Our core capabilities include:
- RV-G Construct Engineering and Optimization: We provide both wild-type and engineered RV-G variants to balance transduction efficiency, stability, and specificity.
- Custom Lentiviral Backbone Design: Tailoring of transfer plasmids to meet specific experimental goals, including the incorporation of fluorescent reporters, optogenetic tools, or therapeutic genes.
- Support for Single/Multi-Gene/Functional Expression Systems: From simple reporter expression to complex, logic-gated circuits.
- Scalable Titer and Bulk Production: Services range from small-scale pilot batches for method optimization to large-scale production for preclinical studies.
Comparison with Other Pseudotyping Strategies
| Feature | RV-G Pseudotyped LV | VSV-G Pseudotyped LV | Retro-AAV |
|---|---|---|---|
| Transport Direction | High Retrograde | Local/Anterograde | High Retrograde |
| Cargo Capacity | Large (~9 kb) | Large (~9 kb) | Small (~4.7 kb) |
| Neurotropism | High | Low/Broad | Moderate |
| Immunogenicity | Moderate | Moderate | Low |
Our Workflow: From Concept to Ready-to-Use Vectors
We follow a structured, phase-gated process to ensure transparency, quality, and reproducibility at every stage of vector development:
-
Phase I
Consultation & Vector Design
When initiating each project, we will thoroughly discuss your research objectives, experimental system and specific requirements with you. Our experts will provide you with the best guidance on vector design, including promoter selection, reporter gene selection and cassette configuration, to maximize your experimental results.
-
Phase II
Plasmid Construction & Validation
Our molecular biology team will employ advanced cloning techniques to clone your gene fragment into an optimized lentiviral backbone vector. All the constructed entities have been fully verified by Sanger sequencing to ensure the complete accuracy and genetic integrity of the sequences.
-
Phase III
Virus Production & Purification
We employ an expandable production system, using HEK293T cells and advanced transfection reagents to prepare high-titer virus samples. Subsequently, the samples are purified through a benzyl alcohol gradient ultracentrifugation to remove impurities and enrich fully functional virus particles.
-
Phase IV
Comprehensive Quality Control
- Quantitative PCR for genomic titer determination
- p24 ELISA for physical particle quantification
- Functional titration in relevant cell lines
- Endotoxin and sterility testing
- SDS-PAGE analysis for protein purity assessment
-
Phase V
Delivery & Documentation
After you complete the order, we will immediately send your virus samples according to the concentration you specified. These samples will be safely packaged with dry ice to ensure their stability. Each delivery will come with a detailed analysis certificate, which includes complete quality control results, as well as a comprehensive project report that outlines the entire production process and various characteristic data.
Key Advantages of Our Pseudotyping Services
High Transduction Efficiency and Expression Levels
Through optimized production protocols and rigorous QC, we ensure that our vectors deliver robust gene expression in both in vitro and in vivo applications. Our vectors consistently achieve high transduction rates across various neuronal cell types.
Rigorous Quality Assurance
We implement a multi-tiered QC approach that exceeds industry standards. Every viral batch is thoroughly characterized for titer, purity, functionality, and safety, providing you with reliable, reproducible results across experiments.
Expert Scientific Collaboration
Our team includes experienced virologists and neuroscientists who provide end-to-end project support from experimental design to data interpretation. We function as an extension of your research team, offering technical insights and troubleshooting assistance throughout your project.
Flexible and Customizable Solutions
We understand that every research project has unique requirements. Our services are highly adaptable to accommodate specific promoter choices, reporter combinations, serotype preferences, and purification specifications.
Client Testimonials

Frequently Asked Questions
Q: What are the specific advantages of using Rabies-G pseudotyped lentivirus compared to other envelopes?
A: Rabies-G confers efficient retrograde trafficking capabilities, enabling transduction of neurons that project to the injection site—a property absent in most other pseudotypes. This makes RV-LVs particularly valuable for neural circuit mapping studies. Additionally, Rabies-G pseudotyped vectors exhibit excellent stability and broad neuronal tropism, making them suitable for targeting diverse neuronal populations.
Q: Can these vectors be used in non-neuronal tissues or applications outside neuroscience?
A: While Rabies-G is particularly renowned for its neuronal tropism and retrograde transport properties, we also offer pseudotyping services with other envelope proteins (e.g., VSV-G, MLV, Ross River Virus) for applications requiring broad tropism or targeting specific non-neuronal cell types. Our team can advise on the optimal pseudotype for your specific research needs.
Q: Do you offer single-cycle (G-deleted) Rabies systems for restricted spread?
A: Yes, we provide both full-length and G-deleted Rabies glycoprotein systems. The G-deleted system offers enhanced biosafety and restricted spread, limiting transduction to directly infected cells without secondary transmission. This is particularly valuable for precise circuit mapping and reduced off-target effects.
Q: What level of documentation and quality control data is provided with the vectors?
A: Each viral preparation is accompanied by a comprehensive Certificate of Analysis that includes detailed information on genomic and physical titers, endotoxin levels, sterility testing results, and functional validation data. Additionally, we provide a complete project report documenting all production and QC processes, ensuring full transparency and reproducibility.
Q: How do I determine the appropriate titer and volume for my experiments?
A: Our technical team provides personalized guidance on dosing based on your specific experimental system, target region, and application. We consider factors such as injection volume, neuronal density, and expression level requirements to recommend optimal vector quantities for your study.
Connect with Us Anytime!
Unlock the full potential of neural circuit research with custom pseudotyped lentiviral vectors from Creative Biolabs. Our comprehensive service platform combines scientific expertise with rigorous quality control to deliver viral vectors that meet the most demanding research standards. Whether you're launching a new research direction or scaling up existing studies, our team is ready to collaborate with you to design, produce, and validate vectors that will drive your discoveries forward. Contact us today to schedule a confidential consultation with our technical team. Let us help you accelerate your research timeline and achieve your scientific goals with precision-engineered viral vector solutions.
Reference
- Deng,Longfei, et al. "Pseudotyped lentiviral vectors: Ready for translation into targeted cancer gene therapy?" Genes & Diseases (2023) 10, 1937-1955. https://doi.org/10.1016/j.gendis.2022.03.007 (Distributed under Open Access license CC BY 4.0, without modification.)
