Glycan Sequencing Service
As a forward-looking company as well as a leading custom service provider in the field of glycoprotein analysis, Creative Biolabs has won a good reputation among worldwide customers. With our extensive experience and advanced platform, we have successfully accomplished numerous challenging glycan sequencing projects for glycoprotein structure analysis. We are glad to help you get milestone success in your glycoprotein project.
Glycan Sequencing for Glycoprotein Structure Analysis
Glycan sequence is one of the main characters of glycopeptide, standing for the monosaccharide sequence and branching. Glycan sequencing can provide an indication of the structure, sequence, and branching of glycan chain, illuminating the protein’s glycosylation profile, microheterogeneity and its activity. Glycosylation has great importance in the development of biological drugs because their glycan chains markedly affect product stability, activity, antigenicity, and pharmacodynamics. Glycan sequences mainly contribute to glycan microheterogeneity.
Technologies for Glycan Sequencing in Creative Biolabs
-
Mass Spectrometry (MS)
Based on glycan ionization, fragmentation, and mass identification of the fragments, MS analysis is usually used for glycan sequencing. Electrospray ionization (ESI) MS/MS and matrix-assisted laser desorption/ionization (MALDI) MS/MS are two main MS methods for oligosaccharide analysis. Before analysis, the glycan sample must be desalted to prevent ionization masking in ESI and MALDI analysis and fragment ions obtained from permethylated derivatives are easily assignable to glycan sequences. Collision-induced dissociation (CID) is the most common method to form glycan fragmentation. High CID causes glycan cross-ring cleavages, providing data on the monosaccharide linkages; while low CID energy yields glycosidic cleavages, providing data on the monosaccharide sequence and branching. CID MS/MS scans can identify putative glycoforms associated with the same glycosylation site.
-
Chromatographic Methods
High-performance anion-exchange chromatography with pulsed amperometric detection (HPAEC-PAD), normal-phase high-performance liquid chromatography (HPLC) and reverse-phase-HPLC (RP-HPLC) are the most widely used chromatographic methods for glycan separation. It is notable that HPAEC-PAD requires high-pH and high-salt buffers (which must be removed before further analysis of the separated glycans, such as in the case of exoglycosidase sequencing). NP-HPLC, having high resolution and reproducibility, is the most efficient method for glycan sequencing. RP-HPLC can analyze the resulting peptide and glycopeptides fragments after digestion of a glycoprotein with specific proteases after reduction and alkylation.
Technologies for glycan sequencing in Creative Biolabs including but not limited to the above methods.
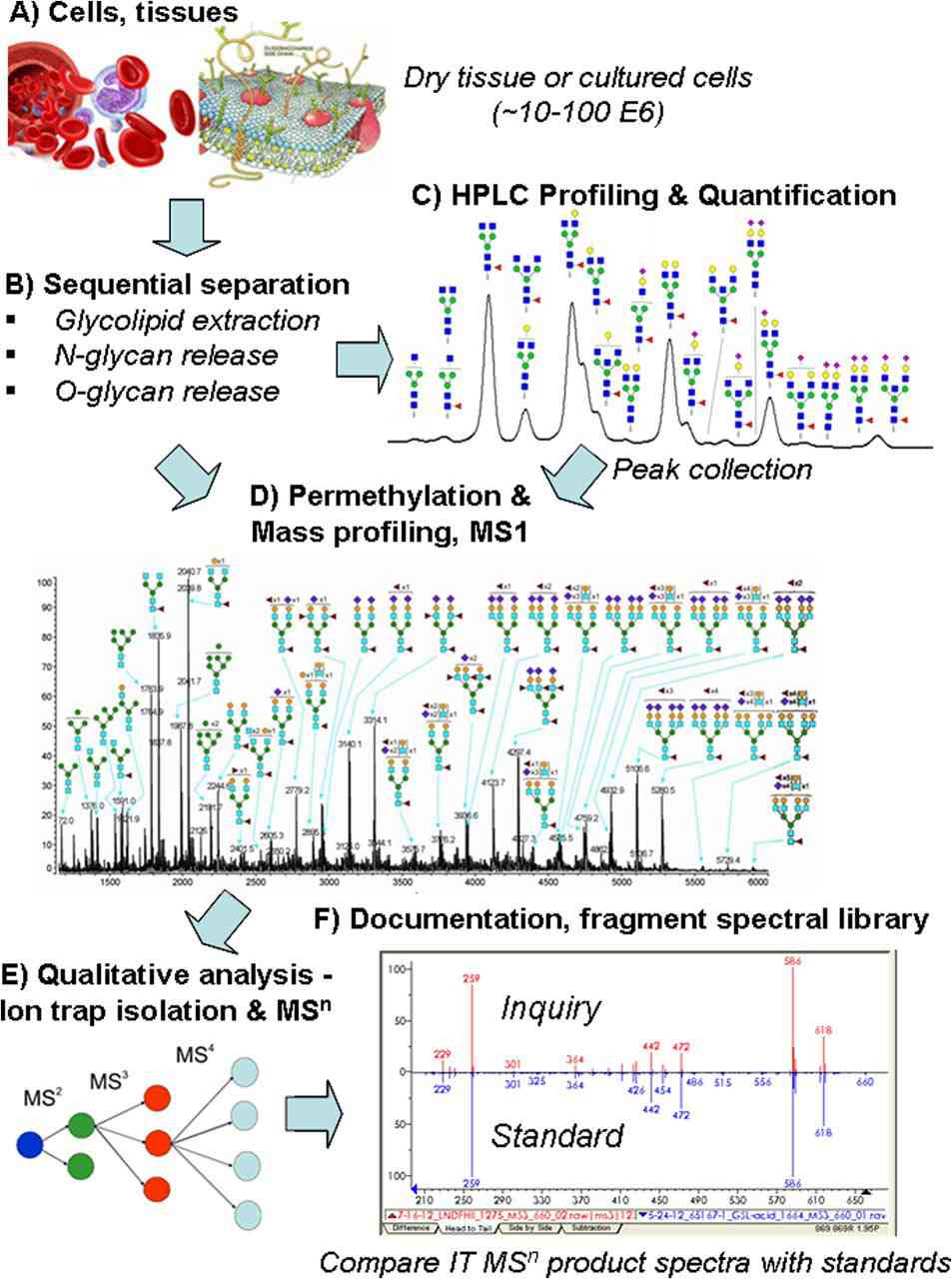 Fig.1 Overview of the integrated glycan sequencing platform.1
Fig.1 Overview of the integrated glycan sequencing platform.1
Features of Our Glycan Sequencing Service
-
Practical and fully experienced technical team
-
High sensitivity and specificity technique platforms
-
Providing accurate and comprehensive analysis data timely
-
High batch-to-batch consistency
-
Competitive prices with quality service
-
Best after-sale service
Working with Us to Promote Your Success!
Creative Biolabs is a leader in the field of glycoprotein analysis and has successfully completed a lot of therapeutic glycoprotein projects. We have experienced experts and advanced platforms that can provide excellent services for glycan sequencing. We can offer a whole set of glycosylation sequencing service to help you get landmark development. We can also customize our offering to meet your specific project needs. If you are interested, please contact us without hesitation.
Published data
Glycans exhibit their activity in a variety of ways. An analytical platform has been proposed for quantitative and qualitative measurement of glycosylation, complete with detailed structural and glycan-peptide linkage understanding. This paper summarizes sample preparation methods and provides some theoretical support for the optimization of the analytical platform. For example, non-reducing O-glycan release strategies in combination with peptide site labeling can be considered. By understanding the sample preparation and subsequent analysis methods in this paper, researchers can better sequence glycans for an in-depth study of their structure and functions.
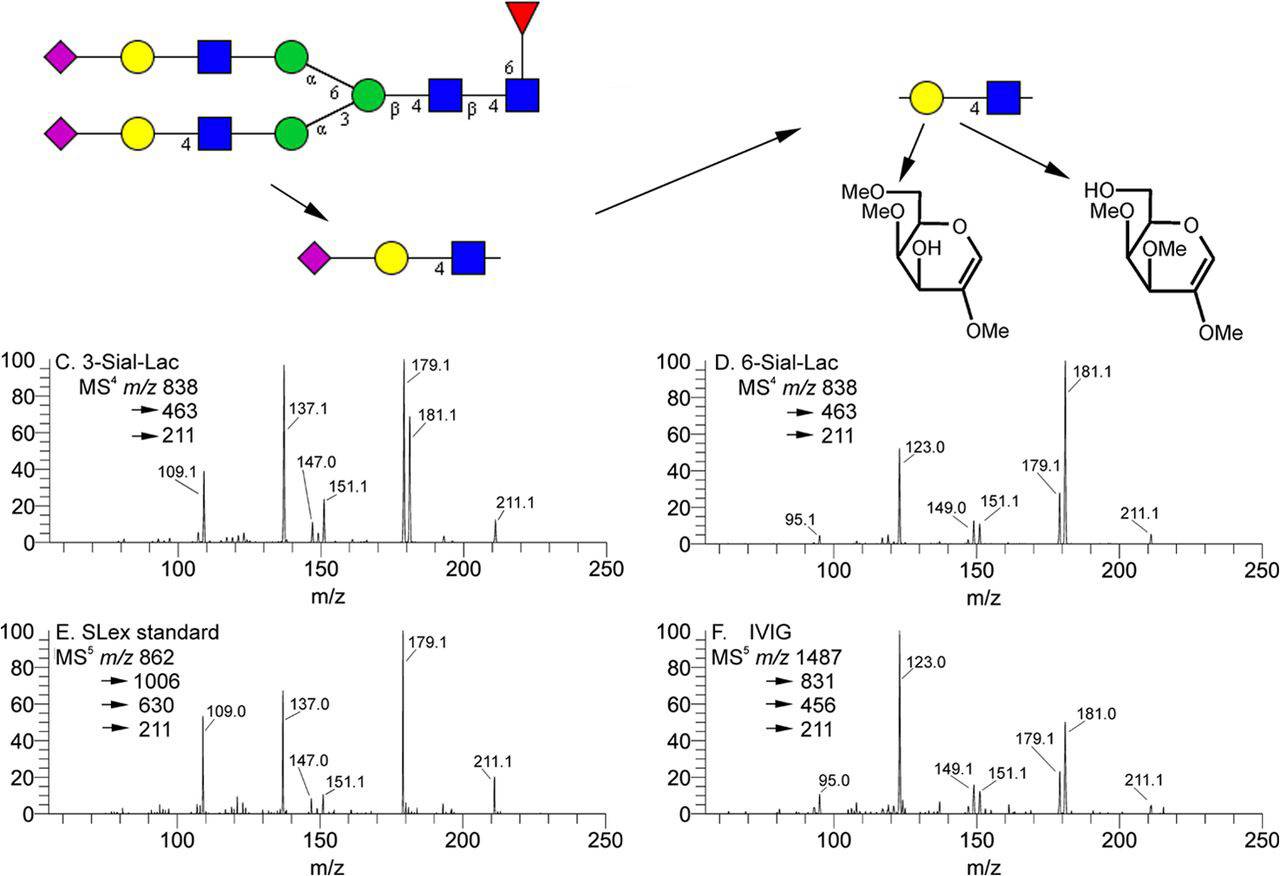 Fig.2 Disassembly of terminal sialyl lactosyl analog structure.1
Fig.2 Disassembly of terminal sialyl lactosyl analog structure.1
FAQs
Q1: Why is glycan sequencing important?
A1: Glycans play key roles in a variety of biological processes such as cell signaling, immune responses, and disease progression. Understanding their structure through sequencing is essential for studying their functions and for disease biomarker discovery. In addition, glycan sequencing has been used to understand the mechanisms of diseases such as cancer and senior immune diseases and to develop disease-targeting therapies.
Q2: Can automated tools assist with glycan sequencing?
A2: Yes, there are software tools available that can assist with glycan sequencing, glycan data analysis obtained from a variety of analysis techniques, and structure elucidation by assisting with glycan sequencing. We have extensive experience in both glycan sequencing and bioinformatics analysis. Combining a variety of advanced tools, we provide an accurate and reliable glycan sequencing service.
Customer Review
High Professionalism and Quality in Glycan Sequencing
"Creative Biolabs impressed us with the professionalism and quality of their glycan sequencing services. Their knowledgeable, experienced, and responsive lab team customized the glycan sequencing protocol in detail. They not only helped us analyze the complexity of glycans but also provided some valuable insights into our research. Their glycan sequencing service was very good and recommended."
Efficient and Timely Glycan Sequencing
"Creative Biolabs was very efficient and prompt with their glycan sequencing service. Not only were the results delivered promptly, but they also provided a detailed interpretation of the data, which greatly enhanced our understanding of the glycan structure. Working with them was a transformative experience."
Reference
-
Reinhold, Vernon, et al. "Toward a platform for comprehensive glycan sequencing." Molecular & Cellular Proteomics 12.4 (2013): 866-873. Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
Related Services

 Fig.1 Overview of the integrated glycan sequencing platform.1
Fig.1 Overview of the integrated glycan sequencing platform.1
 Fig.2 Disassembly of terminal sialyl lactosyl analog structure.1
Fig.2 Disassembly of terminal sialyl lactosyl analog structure.1

