Glycoprotein Detection Services
Glycoprotein detection is an important part of glycosylation engineering. Combining first-in-class technologies with experienced technical team, Creative Biolabs has successfully developed a high-sensitive glycoprotein detection platform to offer the most diverse portfolio of glycoprotein detection products and services. The values of quality, timing and price is our basic criterion. We will try our best to help you get milestone success in your glycoprotein project.
Glycoprotein Detection Is Needed for Therapeutic Glycoproteins Development
As a common co-/post-translational modification, glycosylation plays profound effects on protein structure and function. Many biologically interesting proteins, such as vaccines, antibodies, and enzymes, are glycosylated at their asparagine, serine, and threonine residues. Glycoproteins have been shown to associate with increasing applications in therapeutics, such as plasminogen activator for the treatment of myocardial infarction and strokes, erythropoietin for anemia and various monoclonal antibody-based treatments for cancer. But the complexity of the glycan structures, the multiple substitutions (microheterogeneity) at glycosylation sites, and the structural diversity associated with the protein backbone itself makes the glycoprotein analysis becomes an enormous task. The key effects that glycoproteins have on biological processes and clinical applications stimulate the development of detection and analytical strategies with increasing sensitivity and throughput.
Glycoprotein Detection in Creative Biolabs
In order to perform reliable, detailed detection of glycoproteins, we provide a series of technologies for glycoprotein detection and analysis. Staining and affinity-based methods are two generally used methods.
-
Glycoprotein gel-staining is one of the simplest ways to estimate whether a protein is glycosylated. During the staining procedures, sugar groups of glycoproteins are often chemically restructured with periodic acid to overcome the problem that glycan sugar moieties are not reactive to staining or labeling molecules. The periodic acid oxidizes vicinal hydroxyls on sugars (especially sialic acid) to aldehydes or ketones, which reacts with the Schiff reagent to give a magenta color. This periodic acid-Schiff (PAS) stain can be used to detect and quantify glycoproteins in various biological samples. Notably, it can also be used to make sugars reactive towards crosslinkers, which can be covalently bound to labeling molecules (e.g., biotin) or immobilized support (e.g., streptavidin) for detection or purification.
-
The affinity-based glycoprotein detection is widely used and facilitate the determination of the glycosylation type. According to the different molecules that glycoprotein combined, affinity-based detection can be classified into three types, namely saccharide-binding proteins, enzyme-based methods, and antibody-based methods. These biomolecules have been used to label and isolate glycosylated proteins containing a specific sugar (such as O-GlcNAc or mannosamine) with high specificity and efficiency. The glycan-specific antibodies can specifically recognize the specific sugar on a given protein. Moreover, our glycoprotein labeling kits can tag sugars with fluorescent moieties, which is convenient for direct detection or improved mass spectrometric (MS) ionization.
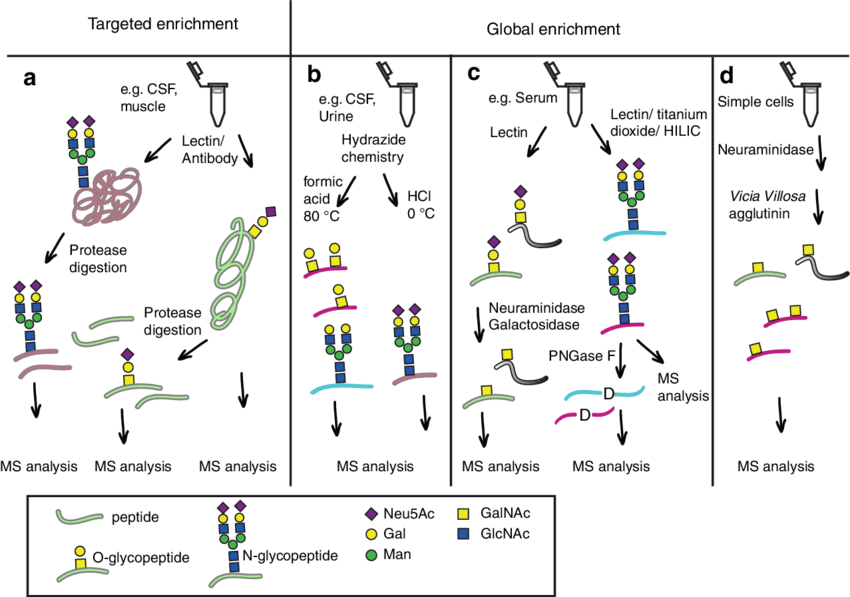 Fig.1 Schematic view of glycoproteomics methods for glycoproteins and glycopeptides.1
Fig.1 Schematic view of glycoproteomics methods for glycoproteins and glycopeptides.1
Why Choose Us?
-
Specificity: our detection methods can specifically detect glycosylated proteins with accuracy.
-
High sensitivity: very small amounts of glycoproteins can be tested through our detection system.
-
Stability and consistency: minimized batch-to-batch variations.
-
Best after-sale service.
Working with Us to Promote Your Success!
With decades of experience, Creative Biolabs has successfully completed a lot of glycoprotein detection projects. We are committed to applying high-quality products, services, and communications to meet your specific project needs. Our off-the-shelf product portfolio and services can help you get landmark development. If you are interested, please contact us without hesitation.
Published data
Ligation causes atrophy of the submandibular gland in rats, but after the removal of the ligature, the submandibular gland regenerates its secretory functions through the proliferation of residual acinar cells and the differentiation of new acinar cells, thereby restoring normal functions and morphology. The authors selected rats as experimental subjects and used the duct ligation-deligation technique to explore the regeneration process of the submandibular gland in rats. They used metal clips to cause atrophy of the submandibular gland ducts in rats, then collected the glands, prepared tissue sections, and performed immunohistochemical analysis. They homogenized, centrifuged, and boiled the glandular tissue in a buffer, then separated the samples by SDS-PAGE gel, and detected glycoproteins using periodic acid Schiff staining. The results showed that compared with the control group, on the 5th and 7th days after the removal of the ligature, the weight of most glands was restored, the acinar cells restored the ability of functional proteins (such as AQP5) and secretion of glycoproteins, and the glycoprotein content was significantly restored. However, these glycoproteins were lost in large quantities during the atrophy process. This study provided research support that ligation-induced submandibular gland regeneration and provided valuable reference information for research on restoring gland functions.
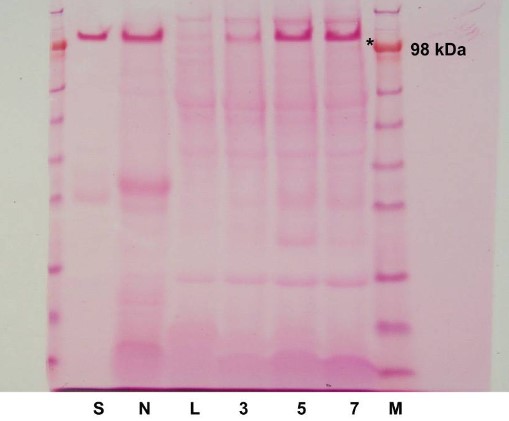 Fig.2 PAS staining of glycoproteins.2
Fig.2 PAS staining of glycoproteins.2
FAQs
Q1: Can you explain the sensitivity of PAS staining in detecting glycoproteins?
A1: The sensitivity of PAS staining mainly depends on the content of sialic acid in glycoproteins. Because periodic acid will oxidize the ortho-hydroxyl groups on sialic acid to aldehydes or ketones, and then react with Schiff's reagent to show magenta. Usually, the detection limit of PAS staining is at the microgram level. For sensitivity detection, we perform quantitative analysis through standard curves, but for very low-concentration samples, we recommend using affinity methods or mass spectrometry to improve detection results.
Q2: Which affinity method do you recommend for specific glycosylation type detection?
A2: We will recommend and determine the final affinity method according to the client's research goals.
-
Glycobinding protein method: It is suitable for identifying specific sugar residues and is recommended for broad screening and preliminary analysis.
-
Enzyme-based method: It is suitable for detecting low-abundance glycosylation sites and types.
-
Antibody-based method: It is very effective for specifically identifying a certain type of glycosylated protein, especially if you need to detect proteins bound by a specific antibody.
In addition, you can also combine our glycoprotein labeling kit to further improve the efficiency and sensitivity of the detection.
Q3: How to solve the impact of glycosylation heterogeneity in glycoprotein samples on analysis?
A3: Glycosylation heterogeneity affects the analysis of glycoproteins. We use affinity methods for specific glycans and binding sites to enrich specific types of glycoproteins. The average is taken by multiple repeated measurements to reduce the impact of random errors. Moreover, we determine glycosylation sites and types by high-resolution mass spectrometry to help distinguish microheterogeneity. These strategies help improve the accuracy and reproducibility of analysis, thereby reliably evaluating the glycosylation status of glycoproteins.
Customer Review
Accurate Glycoprotein Analysis
"Creative Biolabs performed accurate glycoprotein analysis for our therapeutic antibodies. The results showed that the PAS stain was very sensitive in identifying antibody glycosylation sites, and the combination with fluorescent labeling allowed us to efficiently quantify different glycoproteins. This diverse detection method has greatly promoted the progress of our project, and we look forward to continued cooperation in the future."
Good Analytical Service Experience
"The Creative Biolabs team is very professional. From the initial needs assessment of the project to the final result interpretation, every step was communicated and explained in detail. Their technicians answered our various questions about glycoprotein detection and gave multiple optimization suggestions. This customer support experience is an important reason why we choose to continue cooperation."
References
-
Nilsson, Jonas, et al. "Targeting the glycoproteome." Glycoconjugate journal 30 (2013): 119-136. Distributed under Open Access license CC BY 2.0, without modification.
-
Cotroneo, Emanuele, Gordon B. Proctor, and Guy H. Carpenter. "Regeneration of acinar cells following ligation of rat submandibular gland retraces the embryonic-perinatal pathway of cytodifferentiation." Differentiation 79.2 (2010): 120-130. Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only.
Related Services

 Fig.1 Schematic view of glycoproteomics methods for glycoproteins and glycopeptides.1
Fig.1 Schematic view of glycoproteomics methods for glycoproteins and glycopeptides.1
 Fig.2 PAS staining of glycoproteins.2
Fig.2 PAS staining of glycoproteins.2

