About This Program
This program aims to develop anti-OX40L therapeutic fusion protein for immuno-oncology.
Cancer immunotherapy can be improved through combinations of individual agents, as well as through engineering biologic molecules endowed with multiple functions. Bi-specific fusion proteins are receiving increasing attention and have shown promising activity in blocking or bridging two target molecules simultaneously (PD-1, CTLA-4, CD3, CD123, etc.). We are developing a dual-sided fusion protein program wherein a specific checkpoint and OX40 costimulatory pathways could be adjoined.
OX40L
OX40L (CD252, TNFSF4) is a member of the tumor necrosis factor (TNF) receptor superfamily and is expressed on activated antigen-presenting cells (APCs), including B-cells, dendritic cells (DCs), and macrophages. Ligation of OX40 (CD134, TNFRSF4) on activated T cells by its natural ligand OX40L enhances cellular survival, proliferation, and effector functions such as cytokine release and cellular cytotoxicity.
OX40L Fusion Protein in Cancer Studies
Here are some published data about OX40L fusion protein as a potential combination strategy for cancer immunotherapy.
-
The effect of MEDI6383 fusion protein (OX40L-Fc- TRAF2) therapy on human melanoma tumor (cell line A375).
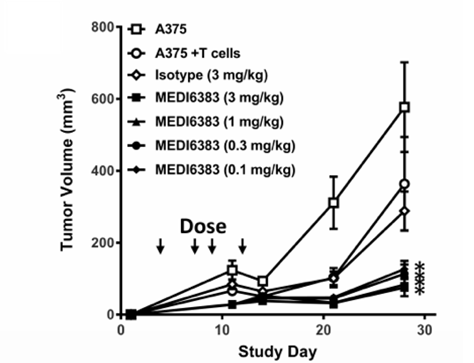 Fig.1 The effect of MEDI6383 fusion protein (OX40L-Fc- TRAF2) therapy on human melanoma tumor (cell line A375).1
Fig.1 The effect of MEDI6383 fusion protein (OX40L-Fc- TRAF2) therapy on human melanoma tumor (cell line A375).1
-
Combination of OX40L-FP (fusion protein) and vaccines reduces lung metastasis and prolongs survival in 4T1 murine breast tumor model.
Clinical Trials under Progress
Currently, to the best of our knowledge, only one OX40 ligand fusion protein (MEDI6383) is undergoing early phase clinical testing for the treatment of advanced solid malignancies. An increasing number of OX40 fusion proteins are getting confirmed on its role to stimulate T cells and shows great promise in cancer treatment. However, further studies are needed to identify engineering and conjugation strategies to improve efficacy, safety, and clinical success.
In this case, OX40L fusion protein is still a compelling target for novel drug development. Currently, Creative Biolabs has established the following programs (including but not limited to):
-
TIM3-Fc-OX40L Fusion Protein Program
-
TRAF2-Fc-OX40L Fusion Protein Program
Program Plan
Creative Biolabs has established the novel chimeric fusion protein platform for Agonist Redirected Checkpoint (ARC) program development. We have extensive knowledge of end-to-end program development. For each program, we are committed to delivering the final complete program to our clients within 1.5 years prior to entering the IND stage.
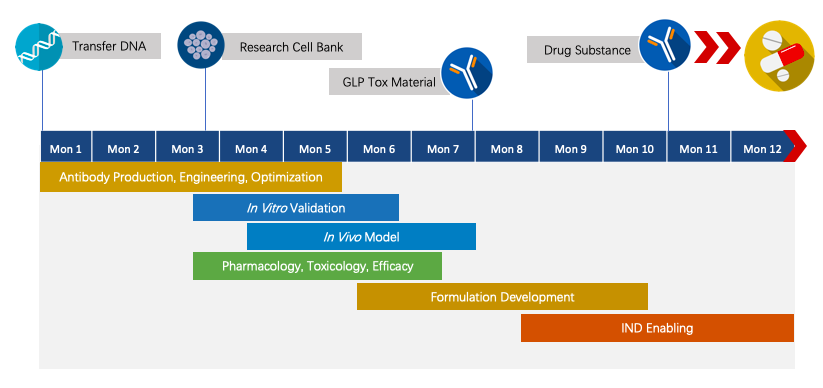 Fig.2 The timeline of Next-IOᵀᴹ programs.
Fig.2 The timeline of Next-IOᵀᴹ programs.
Cooperation
Creative Biolabs is looking for potential partners (include but not limit to major pharma or biotech firms) to develop anti-OX40L therapeutic fusion protein program together. Our scientists are dedicated to bringing together years of valuable experience to our partner and achieve a meaningful partnership. By doing so, we wish to help both parties to proceed with IND and many stages of clinical trials beyond.
If you are interested, please feel free to contact us so that we can discuss the program and other possible opportunities for cooperation. Look forward to working with you in the near future.


 Fig.1 The effect of MEDI6383 fusion protein (OX40L-Fc- TRAF2) therapy on human melanoma tumor (cell line A375).1
Fig.1 The effect of MEDI6383 fusion protein (OX40L-Fc- TRAF2) therapy on human melanoma tumor (cell line A375).1
 Fig.2 The timeline of Next-IOᵀᴹ programs.
Fig.2 The timeline of Next-IOᵀᴹ programs.
 Download our brochure
Download our brochure