Short transient receptor potential channel 3 (TRPC3), encoded by the TRPC3 gene, is one of the classical members of the mammalian transient receptor potential (TRP) superfamily of ion channels. TRPC3 is a non-specific ion channel permeable to calcium and many other cations. Similar to other TRPC channels, TRPC3 features six transmembrane spanning segments with nitrogen (N) and carbon (C) termini residing in the cytoplasm. TRPC3 assembles into tetrameric complexes, which form a cation permeation path with a selectivity filter harboring negatively charged residues to determine calcium ion (Ca²⁺) transport within the pore domains, connecting transmembrane domains 5 and 6 (TM5 and TM6).
| Basic Information of TRPC3 | |
| Protein Name | Short transient receptor potential channel 3 |
| Gene Name | TRPC3 |
| Aliases | Transient receptor protein 3 |
| Organism | Homo sapiens (Human) |
| UniProt ID | Q13507 |
| Transmembrane Times | 6 |
| Length (aa) | 836 |
| Sequence | MREKGRRQAVRGPAFMFNDRGTSLTAEEERFLDAAEYGNIPVVRKMLEESKTLNVNCVDYMGQNALQLAVGNEHLEVTELLLKKENLARIGDALLLAISKGYVRIVEAILNHPGFAASKRLTLSPCEQELQDDDFYAYDEDGTRFSPDITPIILAAHCQKYEVVHMLLMKGARIERPHDYFCKCGDCMEKQRHDSFSHSRSRINAYKGLASPAYLSLSSEDPVLTALELSNELAKLANIEKEFKNDYRKLSMQCKDFVVGVLDLCRDSEEVEAILNGDLESAEPLEVHRHKASLSRVKLAIKYEVKKFVAHPNCQQQLLTIWYENLSGLREQTIAIKCLVVLVVALGLPFLAIGYWIAPCSRLGKILRSPFMKFVAHAASFIIFLGLLVFNASDRFEGITTLPNITVTDYPKQIFRVKTTQFTWTEMLIMVWVLGMMWSECKELWLEGPREYILQLWNVLDFGMLSIFIAAFTARFLAFLQATKAQQYVDSYVQESDLSEVTLPPEIQYFTYARDKWLPSDPQIISEGLYAIAVVLSFSRIAYILPANESFGPLQISLGRTVKDIFKFMVLFIMVFFAFMIGMFILYSYYLGAKVNAAFTTVEESFKTLFWSIFGLSEVTSVVLKYDHKFIENIGYVLYGIYNVTMVVVLLNMLIAMINSSYQEIEDDSDVEWKFARSKLWLSYFDDGKTLPPPFSLVPSPKSFVYFIMRIVNFPKCRRRRLQKDIEMGMGNSKSRLNLFTQSNSRVFESHSFNSILNQPTRYQQIMKRLIKRYVLKAQVDKENDEVNEGELKEIKQDISSLRYELLEDKSQATEELAILIHKLSEKLNPSMLRCE |
TRPC3 is ubiquitously expressed in the cerebellum, cerebrum, and smooth muscles, where it appears to control functions of the cardiovascular system and the brain in a highly specific manner. Especially, TRPC3 plays essential roles in the regulation of neurogenesis and extracellular/intracellular calcium signaling. Dysfunction of TRPC3 has been linked to neurodegenerative disease, cardiac hypertrophy, and ovarian adenocarcinoma. Moreover, it has been reported that TRPC3 plays a role in immunity, cancer, and tissue remodeling, generating much interest in TRPC3 as a target for pharmacological intervention. In basal ganglia (BG) output neurons, TRPC3 channels are involved in maintaining a tonic inward depolarizing current that promotes regular neuronal firing and regulates resting membrane potential. On the contrary, inhibiting TRPC3 will facilitate cellular hyperpolarization, which can result in more irregular and slower neuronal firing. TRPC3 can be activated by neuromodulator diacylglycerols (DAG) and substance P.
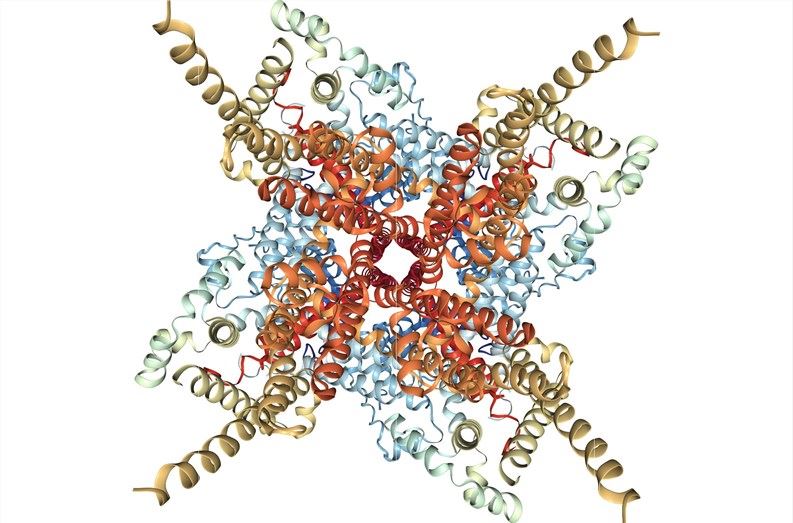 Fig.1 Cryo-EM structure of human TRPC3 at 4.36A resolution. (Tang, 2018)
Fig.1 Cryo-EM structure of human TRPC3 at 4.36A resolution. (Tang, 2018)
This article concludes that postsynaptic GABABRs modulate Purkinje cell responses to strong glutamatergic stimulation through regulation of mGluR1-TRPC3 coupling.
This article determines the structure of human TRPC3 by single-particle cryo-electron microscopy to 4.4 Å resolution.
This article confirms that overexpression of TRPC3 protein may be associated with hyperresponsiveness of airway smooth muscle and remodeling in asthmatic mice, providing evidence for a new target for drug intervention and a new direction of asthma pathogenesis research.
This report provides evidence for a novel concept of lipid-sensing by TRPC ion channels based on a lateral fenestration in the pore domain which accommodates lipid mediators to control gating.
This article demonstrates an important role of TRPC3 with regards to the pluripotency, survival and neural differentiation of mouse embryonic stem cells.
In order to provide high-quality membrane protein preparation service, we have developed a versatile Magic™ membrane protein production platform. Our experienced scientists will do their best to help you find a perfect match in your required formats. Aided by our versatile Magic™ anti-membrane protein antibody discovery platform, we also provide customized anti-TRPC3 antibody development services.
Creative Biolabs is dedicated to providing first-class membrane protein production service using a variety of strategies. Based on our leading-edge platform, we have successfully produced, purified, stabilized and characterized many challenging membrane protein targets for global customers. If you are interested in the service we can provide, please feel free to contact us for more information.
Reference
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.