Vaccines for Virus from Orthomyxoviridae Family
The ravages of seasonal flu are a worldwide problem, and vaccines are a proven and effective option. Creative Biolabs has a lot of voice in vaccine research, and more than a decade of development has made the company a leader in the world's vaccine industry. We have accumulated rich experience and fruitful results in the research of influenza virus vaccine, which can perfectly solve all your problems in the development of influenza vaccine!
Orthomyxoviridae
Orthomyxoviridae is a family of RNA viruses comprising seven genera of Influenzavirus A, Influenzavirus B, Influenzavirus C, Influenzavirus D, Isavirus, Thogotovirus, and Quaranjavirus, among which the first four genera are pathogens of human, avian, and other mammals influenza, while the host of Isaviruses is salmon and the thogotoviruses as an arbovirus can infect both vertebrates and invertebrates. The four influenza viruses in the family are divided into four genera due to their antigenic structure of matrix protein and nucleoprotein, which are influenza virus A, influenza virus B, influenza virus C, and influenza virus D. What they have in common is the infection of vertebrates.
Table1. Orthomyxovirus genera, serotypes, and hosts.
| Genus | Serotypes | Hosts |
|---|---|---|
| Influenzavirus A | H1N1, H1N2, H2N2, H3N1, H3N2, H3N8, H5N1, H5N2, H5N3, H5N8, H5N9, H7N1, H7N2, H7N3, H7N4, H7N7, H7N9, H9N2, H10N7 | Vertebrate like bat, bird, pig, horse, and human |
| Influenzavirus B | Yamagata, Victoria | Seal and human |
| Influenzavirus C | Dog, pig, and human | |
| Influenzavirus D | Cattle and pig | |
| Isavirus | Salmon | |
| Thogotovirus | Jos virus, Batken virus, and Bourbon virus | Mosquito, tick and mammal |
| Quaranjavirus | Arthropods and birds |
Features of Orthomyxoviruses
The viruses of the Orthomyxoviridae family are mostly polymorphic, and their envelopes are spherical or filamentous. Typically, the virions are presented as ovals having a diameter of 80-120 nm or filaments having a diameter of 80-120 nm and a length of no more than 20 μm. There are approximately 500 spikes on the surface of each envelope that are 10-14 nm in length. The major glycoprotein HA of the virus is inserted into neuraminidase (NA) in a ratio of 4-5:1 (HA : NA). The membrane containing glycoprotein protrusion and cholesterol envelops the nucleocapsid, while the genome of the virus is a linear negative-sense single-stranded RNA containing 6-8 segments, which encode PB1, PB2, PA as polymerase, hemagglutinin HA, nucleoprotein NP, neuraminidase NA, membrane protein M, and non-structural protein NS, respectively. Neuraminidase is responsible for the release of progeny virus from infected cells, and hemagglutinin is involved in mediating the binding of the virus to target cells and the entry of the viral genome into target cells. Therefore, these two proteins have also become targets of antiviral drugs, and the responses of antibodies against these proteins with the proteins has also become the basis for distinguishing different serotypes of influenza A viruses.
The influenza can spread through saliva, blood, feces, and nasal secretions. The hemagglutinin on the surface of the virus interacts with sialic acid on the surface of the epithelial cells of the host's larynx or lung, leading to entry of the virus into the target cell by endocytosis. In endosomes, haemagglutinin mediates fusion of the viral envelope with the vacuole membrane, releasing viral RNA and related proteins into the cytoplasm, which subsequently form complexes that are transported into the nucleus and thereby initiate replication of viral nucleic acids and transcription. The newly synthesized viral proteins can be transported to the cell surface or transported back to the nucleus to bind to the viral RNA and assemble into new viral genomic particles. Some viral proteins also have the function of degrading cellular mRNA and taking advantage of the released nucleotides to synthesize viral RNA.
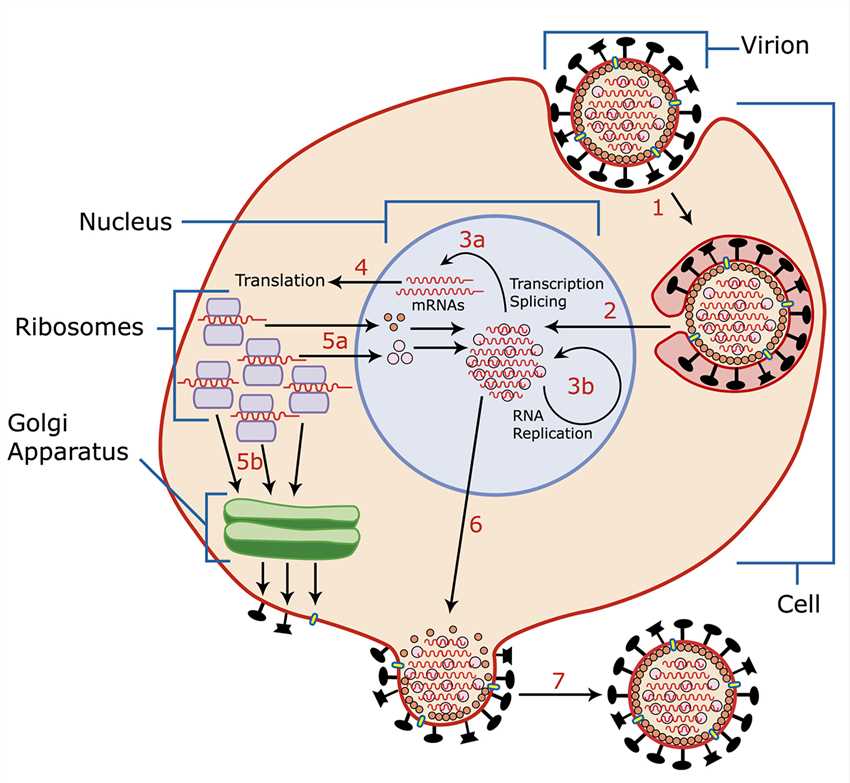 Fig. 1 Invasion and replication of the influenza virus. Distributed under CC BY-SA 3.0, from Wikimedia, the image title was changed to "Invasion and replication of the influenza virus.
Fig. 1 Invasion and replication of the influenza virus. Distributed under CC BY-SA 3.0, from Wikimedia, the image title was changed to "Invasion and replication of the influenza virus.
The Development of Vaccines for Influenza Virus
There are already vaccines and drugs for influenza virus. Such vaccines include attenuated live virus vaccines or inactivated vaccines of human influenza A viruses and influenza B viruses. One problem with such viral vaccines is that since the antigenic structure of wild-type viruses are constantly changing, the corresponding vaccines have to update the paralleled virus seeds every year because once the antigenic structure of the seed virus is inconsistent with the wild virus, the vaccine will be no longer possible to provide protection for vaccines. What's more, even if the two do match, there will also exist the possibility of generation of escape variants. Influenza vaccines are usually produced using eggs that have been fertilized. After the WHO announced the recommended strains for the winter flu season, each vaccine producer began manufacturing vaccines. In addition to eggs, the EU has also adopted the flu vaccine produced by animal cells, which can meet the needs of different mass production and avoid allergies caused by vaccine production using eggs.
The universal influenza vaccine is effective for a wide range of influenza viruses and does not require customization of specific strains. There are generally two strategies for designing such vaccines. One is to create a vaccine that stimulates antibodies against HA, and the other is to include four H protein subtypes within one vaccine. The universal influenza vaccine is already in clinical research.
In addition to the use of eggs, mammalian cells to produce influenza virus, cell culture production of influenza virus vaccine, recombinant protein vaccine have been approved by the FDA, plant-based influenza vaccine is also in the clinical evaluation stage.
The research and development of vaccines are beneficial to people all over the world. Since its inception, Creative Biolabs has continued to expand its professional research team and experience in vaccine development. Now it has successfully established a comprehensive development and service system to provide the most scientific and reasonable solutions and products for your vaccine research.
All of our products can only be used for research purposes. These vaccine ingredients CANNOT be used directly on humans or animals.


