
Single domain antibodies (sdAbs, or VHH) are the smallest antigen-binding units of antibodies, comprising either one variable domain or one engineered constant domain that solely facilitates target binding. They are popular as a novel class of proprietary therapeutic proteins containing unique structural and functional properties. At Creative Biolabs, we offer one-stop solutions for sdAb development and characterization to promote the identification of novel therapeutic sdAbs to meet your specific project demands.
As one of the novel antibody-based therapeutic agents, sdAbs display several advantages when compared to conventional antibodies. Firstly, as the smallest antigen-binding units of antibodies, sdAbs exhibit similar affinities as compared to full-sized antibodies. Due to their long, protruding and highly variable CDR3 section, sdAbs have the ability to access epitopes normally hidden for conventional antibodies. Secondly, sdAbs are readily expressed in and purified from different host systems, including E. coli, yeast, plant, and mammalian cells. Thirdly, they have exhibited high solubility and thermal stability, high refolding capacity, and good in vivo tissue penetration. Moreover, sdAbs can be engineered into various formats by fusion to other proteins or peptides, thereby tailoring their utility for certain therapeutic applications. The above advantages make them ideally suited for various therapeutic applications.
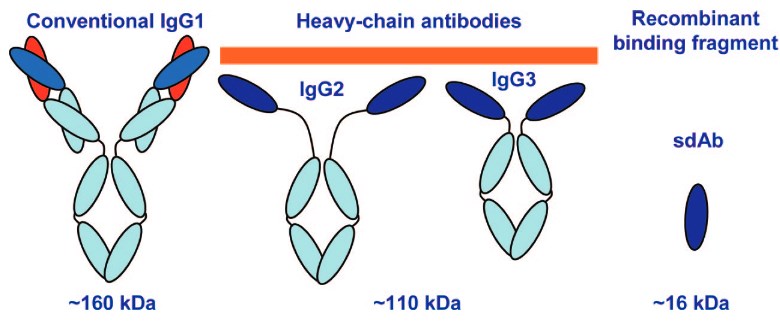 Fig.1 Conventional IgG (IgG1), heavy chain only subtypes (IgG2 and IgG3), and sdAb. (Anderson, 2008)
Fig.1 Conventional IgG (IgG1), heavy chain only subtypes (IgG2 and IgG3), and sdAb. (Anderson, 2008)
Potential targets for sdAb include cell surface proteins, cytokines, secreted proteins, intracellular proteins, etc. Depending on the targets, the therapeutic effects of sdAbs can be mediated by different mechanisms, including but not limited to:
Thanks to their single domain nature, sdAbs can be readily equipped with various effector functions through molecular or biochemical engineering. These effector functions include antibody-dependent cellular cytotoxicity (ADCC), antibody-dependent cellular phagocytosis (ADCP), complement-dependent cytotoxicity (CDC), thereby enhancing and broadening the efficacy of sdAbs. Moreover, the fusion has been also reported to result in half-life extension.
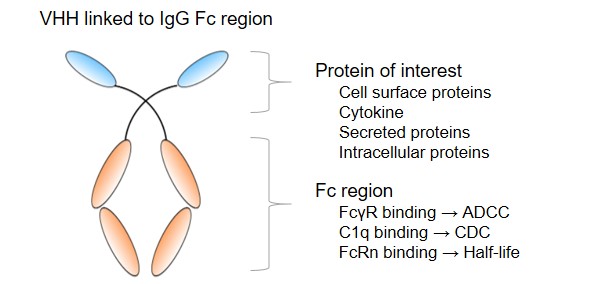 Fig.2 VHH linked to IgG Fc region.
Fig.2 VHH linked to IgG Fc region.
Given that therapeutic antibodies rely on different types of MOAs to achieve clinical efficacy, it is essential to apply different strategies to assess and identify the functions of these sdAb candidates. Here at Creative Biolabs, we are proud to present the most comprehensive list of assays for screening and evaluating the functionality of sdAb therapeutics, including but not limited to:
Creative Biolabs is devoted to tailoring the best-fit proposal to screen and identify the desired sdAb candidates with expected functionalities to meet your objectives. If you are interested in learning more about our capacity for functional assays, please do not hesitate to contact us.
Reference
We are offering highly customized CRO services to assist your Single Domain Antibody (sdAb) related projects. Please Contact Us for more details.
Enter your email here to subscribe.
Submit

Follow us on