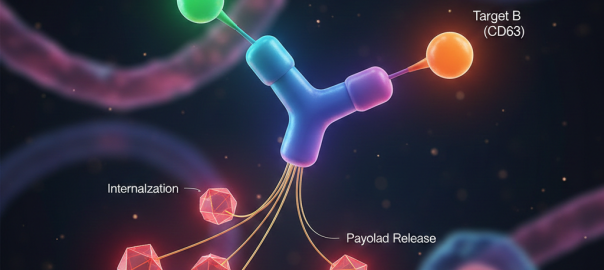
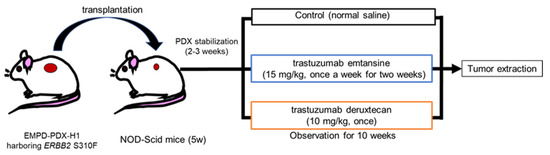
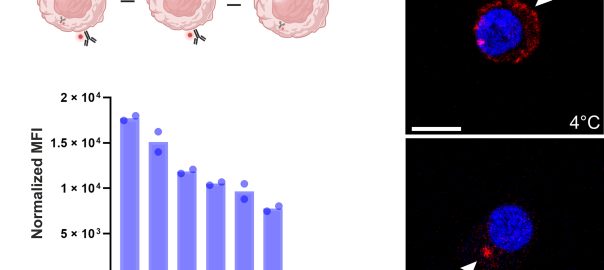
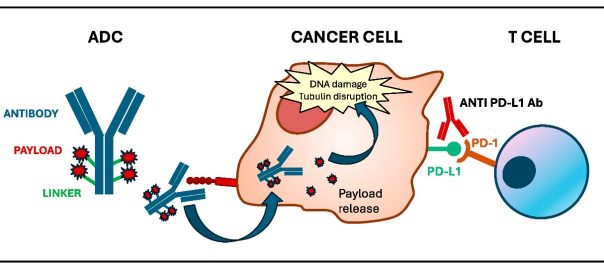
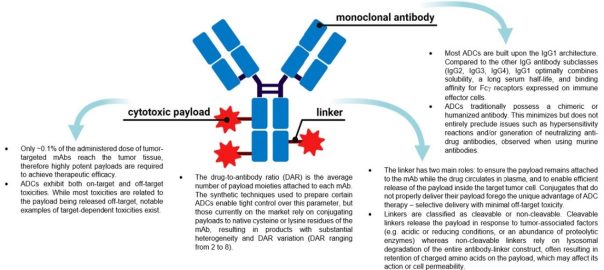
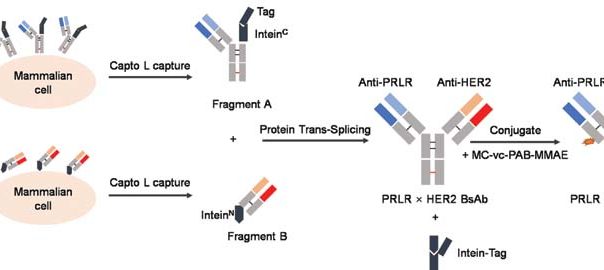
Engineering Smarter Payloads: The Rise of Next-Generation Bispecific ADC Strategies
A New Generation of Precision Delivery Antibody-drug conjugate (ADC) science is undergoing a quiet revolution. The core concept — linking a cytotoxic payload to a tumor-targeting antibody — remains unchanged. But theRead More…