Complement is an essential component of the intrinsic immune system, comprising a set of serum-functioning proteasome cascade pathways that play a crucial role in clearing invading pathogens and senescent dying cells. ProteaseRead More…
Complement Protein C5 Track Heats Up with Two Drug Approvals in August
The complement system is an important part of the innate and adaptive immune system. When activated, it participates in the body’s defense response and immune regulation, including enhancing antibody response, possessing immuneRead More…
Interactions between MSCs and the Immune Complement
Complement, named because it assists antibodies in removing pathogens, is a group of about 20 proteins found in plasma. Complement is mainly derived from the liver or certain cell membrane proteins andRead More…
Biological Significance and Bioanalytical Strategies of Complement Assays
Bordet, the winner of the 1919 Nobel Prize in Physiology or Medicine, found that when serum was heated to 56°C, it lost its ability to kill bacteria, even though the antibodies inRead More…
Br J Pharmacol: Complement C3 as a New Target for Reducing Proteinuria in Patients with Hypertensive Nephropathy
The increasing number of clinical diseases involving pathological contributions of the complement system has prompted renewed interest in therapeutic options to modulate this host defense pathway. Based on these findings, the pharmaceuticalRead More…
Advances in Complement System Targeted Therapy in Neurological Diseases
The complement system, a vital component of innate immunity and a potent promoter of inflammation, has a crucial role in host defense against pathogens and the development of central and peripheral nervousRead More…
Successful Expansion of Complement Drugs into the Market for Major, Chronic, and Common Diseases
On February 17, 2023, Apellis announced the FDA approval of Pegcetacoplan, a complement C3 inhibitor, as the first molecularly targeted drug for the treatment of dry AMD or geographic atrophy. This approvalRead More…
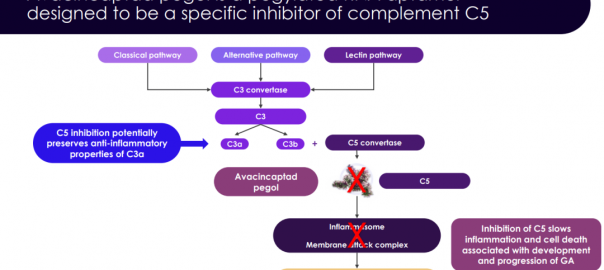
Complement C5 Inhibitor Receives Priority Review by the FDA for Geographic Atrophy
On February 16, Iveric Bio announced in an announcement that the New Drug Application (NDA) for the complement C5 inhibitor Zimura (avacincaptad pegol, ACP) for the treatment of age-related macular degeneration (AMD)Read More…
Application of Nucleic Acid Aptamers in Tumor Immunotherapy
Nucleic Acid Aptamer Nucleic acid aptamers are single-stranded DNA or RNA (ssDNA or ssRNA) that are able to bind specifically to target molecules by folding to form a unique tertiary structure. TheRead More…
Cell sub-journal: Mechanism of conversation between sphingolipid signaling and complement signaling for cancer metastasis revealed
In a new study, researchers have identified a series of molecular interactions that may be key to stopping the spread of cancer cells. The new study identifies, for the first time, aRead More…