Chimeric antigen receptor (CAR) modified T cells (CAR-T) as a new field of adoptive immune cell therapy has become a hot spot in recent research. In 1989, Israeli scholar Gross et al. First proposed the concept of CAR-T cells, which blend the antibody-variable region of the tumor antigen with the constant region of the T cell receptor (TCR) to construct a chimeric antigen Receptor (CAR). It was found that CAR T cells could recognize specific tumor antigens without MHC restriction while activating killer T cells (CTL) and playing the role of killing tumor cells. As the CAR-T cells technology develops, more hope has been brought to these incurable diseases, such as autoimmune disease, blastoma, carcinoma, leukemia, and lymphoma.
Carcinoma is a type of cancer that originates in epithelial cells. In particular, a carcinoma refers to cancer that begins in a tissue that lines the inner or outer surfaces of the body, and that progress develop from cells originating in the endodermal or ectodermal Germ layer during embryogenesis.
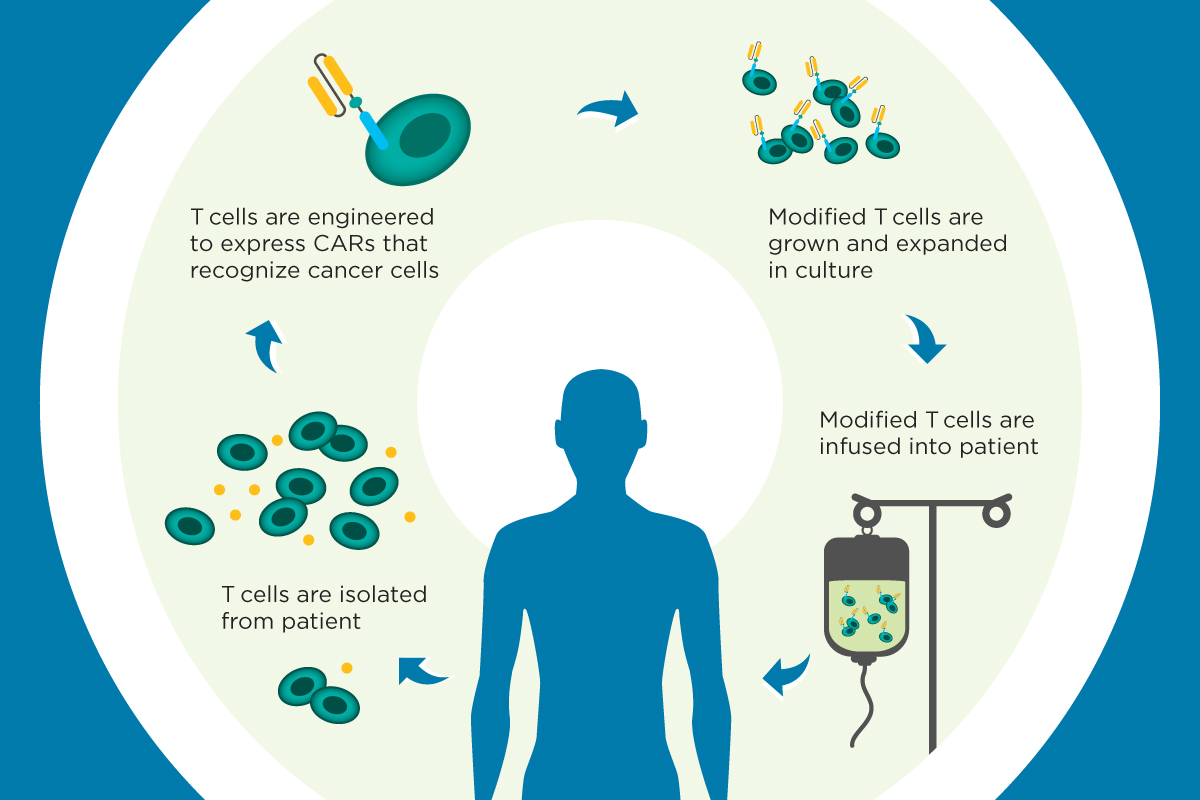
Like other immunotherapies, the basic principle is to clear cancer cells by taking advantage of immune cells itself. But the difference is that this is a cytotherapy not a drug. Car T for Carcinoma Therapy can be divided into 5 steps. The cell is separated from the body of cancer patients. Then a cell that can confirm tumors is added into the T cell by the genetic engineering technology and at the same time, T cell is activated to kill chimeras of tumor cells, turning into CAR-T Cells to target and kill cancer cells at any time. The third stage belongs to in vitro culture. A great number of CAR-T cells are amplified. Normal speaking, a patient needs billions of CAR-T cells. The larger patients body, more Cells will be needed. Next, these amplified CAR T cells are transfused back into the patient body. Eventually, patients will be monitored closely to control their acute reactions.
Leukemia is a group of cancers that usually start in the bone marrow and result in high numbers of abnormal white blood cells which are not fully developed and are called blasts or leukemia cells. Symptoms of Leukemia may include bleeding and bruising problems, feeling tired, fever, and an increased risk of infections.
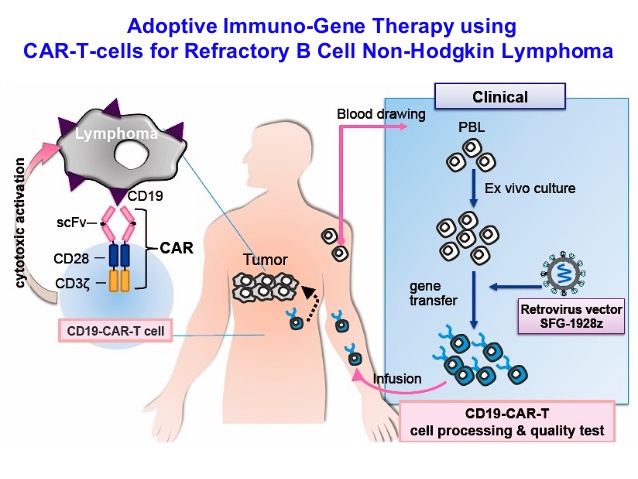
In general, CAR is composed of the extracellular antigen recognition domain of the antibody and the intracellular signal transduction mechanism of the T cell receptor (TCR). Since T cells require co-stimulus to be fully activated, the second generation of CAR is designed with additional intracellular domains (usually from CD-28 or 4-1BB) and the second signal is delivered at the time of receptor binding. For acute lymphoblastic leukemia, early B cells (blasts) out of control are mainly in the periphery. These cells typically exhibit high levels of CD19, which appear in almost all B cell lineages but do not appear elsewhere. As for other blood cancers, even if they express CD19 antigens, such as chronic lymphocytic leukemia (CLL), they are often tolerated by CAR-T therapy because these cell types provide a more challenging environment for T cell binding. At the same time, they may exist in other isolated parts of the body.
Lymphoma is a group of blood cell tumors that origin in lymphocytes (a type of white blood cell). Signs and symptoms may include enlarged lymph nodes, drenching sweats, fever, itching, unintended weight loss, and constantly feeling tired.
Car T for Lymphoma therapy usually are targeted to CD19 antigen. In fact, the first CAR-T cell clinical trial was conducted in patients with NHL. Many clinical trials use CAR-T cells to treat low-risk (FL) and intermediate-risk (DLBCL). The results of these trials are largely encouraging but are accompanied by adverse reactions in patients with similar leukemia.