According to the World Health Organization (WHO), cancer is the second leading cause of death in the world. In 2018 alone, 9.6 million people worldwide died of cancer, accounting for about 1/6 of those deaths. Many pharmaceutical companies are stepping up efforts to develop new attack lines against cancer. They believe that if cancer statistics are to be made less daunting, it is necessary to explore alternatives to traditional anti-cancer methods.
It is well known that in the past cancer research focused on the potential genetic and epigenetic defects of the disease. Today, however, pharmaceutical companies are turning their attention to tumor microenvironment (TME), a battlefield of elements around cancer cells.
What is tumor microenvironment?
In addition to cancer cells, tumors are also composed of many other cell types, which together form the tumor microenvironment. These include stroma cells, fibroblasts, endothelial cells that make up the blood vessels in the tumor, extracellular matrix, and many different immune cells that researchers are most interested in. Tumors affect their microenvironment by secreting growth factors, immunosuppressive molecules and other signal molecules, which provide the perfect environment for tumor reproduction.
On this battlefield, the fog of war may confuse the immune system, making it impossible to distinguish between friends and enemies. On the contrary, dispelling the fog will help the immune system fight it more effectively.
The existing immunotherapy has been commercialized and has achieved great success. It is worth noting, however, that only a small number of cancer patients benefit. People began to think that if the newly developed immunotherapy began to target other factors of TME, it might achieve better results. Last year, the 10th European Protein and Antibody Engineering Summit (PEGS) discussed the possibility of TME targeting. Common strategies may include, but are not limited to, antibody drugs, cell therapy, oncolytic virus, and combination therapy.
1.Antibody Drugs
Monoclonal antibody (mAb) is the most common immunotherapy. These antibodies grow in cloned immune cells and target specific antigens expressed by the tumor. Monoclonal antibodies in immunotherapy have many different mechanisms. For example, some mAb label tumors so that they are attacked by immune cells, while others block antigens that contribute to tumor growth and spread. Or, the antibody drug conjugates (ADC) transfer the drug directly to the tumor by binding the antibody to toxins or radioactive particles. Bispecific antibodies and multispecific antibodies are also becoming options for cancer immunotherapy that can provide significant advantages.
Several drugs are being developed in preclinical research and testing, including the ADC of tough mucin C and the extra domain B (EDB) of fibronectin. Clinically, antibodies that recognize specific fibronectin splicing variants EDB, which are conjugated with IL2 and TNF to recruit immune cells into tumors for use in advanced melanoma, are being studied and phase 3 trials are underway (clinialtrials.gov: NCT02938299).
TNFR2 is very rare in healthy people, but it is abundant in the microenvironment of some tumors. “Of all the cancer targets so far, I have never seen a preferred expression in TME and as a T cell inhibitor and carcinogenic gene, as well as low expression in humans.” Dr. Denise L. Faustman, immunobiology director of Massachusetts General Hospital, pointed out that TNFR2 is overexpressed on Treg in TME, making it super-inhibitory. TNFR2 itself is also a way of proliferation, so it can also promote cell expansion and prevent effective treatment, which is the difference. Through the expression of oncogenes or the overexpression of regulatory T cells and myeloid inhibitory cells in tumor, the direct proliferation of tumor is promoted. According to research led by Dr. Denise Faustman, multifunctional TNFR2 antagonistic antibodies may specifically alter TME.
Bicycle Therapeutics is developing the bicycle peptide-drug conjugate (BDC). Dr Kevin Lee, chief executive officer, said the problems with monoclonal antibodies, especially ADC, included large volumes and long half-lives in the body. When a variety of long-acting antibodies are used in combination immunotherapy, they may lead to excessive activation of the immune system, resulting in side effects and loss of efficacy. Lee stresses that bicycle peptides are much smaller than antibodies and can penetrate tumors quickly. Therefore, it can play its role without having to rely on the long cyclic half-life of the body. Its research drug is currently in the first phase.
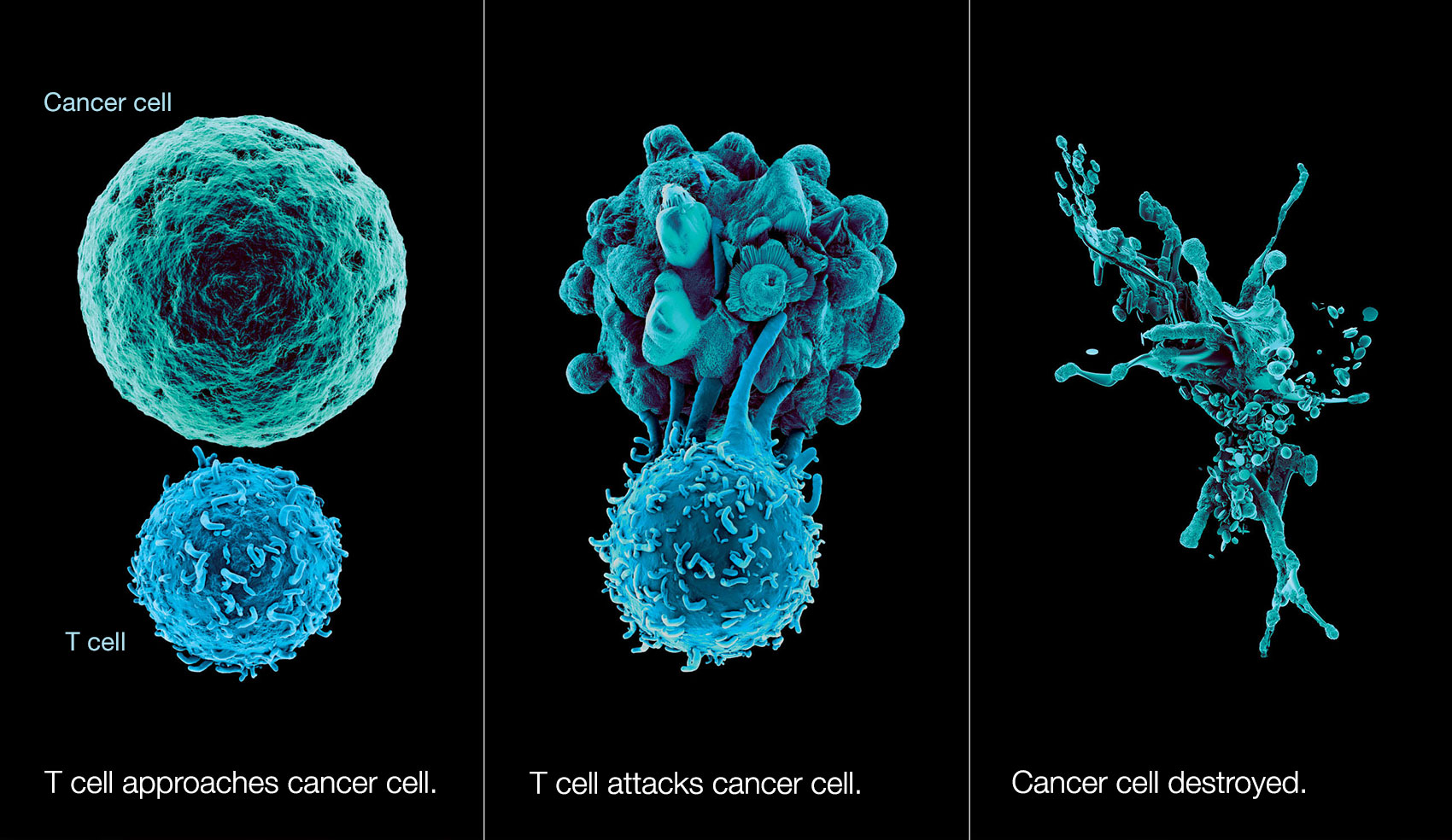
2.Cell Therapy
Different from blood tumors, the application of CAR-T cell therapy in solid tumors is faced with more complex TME. In many cases, we pay more attention to the direct attack on tumor cells, and there are relatively few studies on the microenvironment on which tumor cells live. One of the reasons for the slow progress of CAR-T in the treatment of solid tumors is that the immunosuppressive effect of TME on CAR-T does not produce good therapeutic effect.
The dynamic interaction between tumor cells and their surrounding matrix leads to the formation, progress, metastasis and drug resistance of solid tumor. Previous studies have shown that the key components of matrix in tumor microenvironment are beneficial to the growth and metastasis of tumor cells. In recent years, researchers have found that tumor matrix also affects the immune effect of anti-tumor. Therefore, in the process of effective T cell treatment of tumor, targeting related stroma cells is also very necessary.
Among them, the protein of, FAP (fibroblast activating protein (protein cleaving enzyme) plays an important role in the physical properties of matrix formation. In 2017, researchers from the University of Pennsylvania found that FAP was overexpressed in pancreatic tumors compared with normal pancreatic tissues. The high expression of FAP was associated with poor prognosis. When they deleted the gene encoding the FAP protein in the mouse model with the disease the life span of the mice was significantly prolonged and the spread of the tumor to other organs was effectively reduced. Previous preclinical studies have shown that CAR-T therapy targeting FAP suppresses tumor formation, reduces tumor vascular density, and destroys the spatial orientation of tumor cells.
3.Oncolytic Virus
Oncolytic virus can recruit tumor infiltrating lymphocytes (TIL) into tumor microenvironment, which can change tumor immune rejection and immune depletion. In addition, oncolytic virus helps to maintain the phenotype of effective TIL, which may reverse immunosuppression in immunoinfiltrating microenvironment.
As we learn more about checkpoint suppression, we understand that it doesn’t work for everyone, said Dr. Philipp Mueller, chief scientist of cancer immunology and immunomodulation at Boehringer Ingelheim. Depending on the tumor indications, about 10% of patients can benefit, but there are still most patients who do not benefit much.
Boehringer Ingelheim is conducting a preclinical test of the oncolytic virus, a vesicular stomatitis virus, VSV-GP, that carries a modified glycoprotein. The VSV-GP selectively targets cancer cells, and the company hopes it will benefit patients who do not respond to checkpoint therapy. This group of patients usually suffer from so-called “cold tumors”, which usually have low levels of mutation and express very little newborn antigen (neoantigen).
The innate immune response pathway of cancer cells is defective and vulnerable to virus infection, Mueller explained. The purpose of oncolytic virus therapy is to enable the virus to selectively penetrate and attack tumor cells without affecting healthy tissue, thus inducing the activation of the patient’s immune system to fight the tumor.
Last November, researchers at Oxford University and PsiOxus Therapeutics designed an oncolytic virus that encodes BiTE (bispecific T-cell engager) based on clinical oncolytic virus enadenotucirev. It can target cancer cells and immunosuppressive stromal cells at the same time. The study was published on Cancer Research.
This is also the first time that scientists have specifically targeted cancer-related fibroblasts in solid tumors in this way. If further safety tests are successful, clinical trials could be conducted as early as this year, the researchers said.
4.Combination Therapy
TME may also be the key to improving existing treatments. The track of oncolytic virus combined with checkpoint inhibitors is hot because oncolytic virus is considered to be a way to turn on the local immune response to treat tumors. This is a timely help for checkpoint inhibitors, and the combination is expected to be applied to more types of cancer. In addition, potential combinations include the combination of CAR-T cell therapy and oncolytic virus. For example, on February 20, 2019, Mustang Bio cooperated with the Nationwide Children’s Hospital and signed an exclusive global license agreement to develop a combination of MB-101 (IL13R α 2 specific CAR) and C134 for glioblastomas.