Cytokine/Chemokine-expressing Oncolytic Virus
With years of experience in oncolytic virus development, Creative Biolabs has built an advanced oncolytic virus engineering platform. Taking advantage of the advanced platform, we provide customized, standardized, and reliable high-quality cytokine/chemokine-expressing oncolytic virus.
Genetic therapies, such as transfected viral vectors and immune cells, continue to display clinical responses as systemically deliverable and targeted therapeutics, with the first such strategy having been approved for cancer treatment. Most of these use cytokine transgenes. Nevertheless, expression of cytokines early after systemic delivery is able to induce improved toxicity and nonspecific induction of the immune response. Besides, genetic therapies may result in premature immune-mediated clearance, especially for viral-based methods. Recently, the field of oncolytic virology has made great strides with the publication of exciting phase II clinical results from many different cytokine-expressing vectors, containing confirmation of the ability for systemic viral delivery to the tumor in the clinic. Based on our comprehensive oncolytic virus engineering platform, our scientists are confident in providing the first class cytokine/chemokine-expressing oncolytic virus to meet your requirements of research, preclinical study and drug development requirements.
Example 1: CCL5 Expressing Oncolytic Virus
The chemokine CCL5 is a RANTES, which is able to regulate upon activation and normal T cell expressed. It plays an important part in several human diseases and binds to three receptors: CCR1, CCR3, and CCR514, which are expressed on a variety of types of effector and regulatory T cells. It is formed by both activated T lymphocytes and antigen presenting cells, and is widely chemoattractive. Recently, researchers have demonstrated that vvCCL5 (an oncolytic vaccinia virus expressing CCL5) constitutively expressing CCL5, promotes immune infiltration of mouse colorectal tumors in vivo and improves therapeutic effects. Besides, CCL5 expression also induces prolonged persistence of the virus exclusively within the tumor. What's more, further improvement of antitumor effectiveness is observed when vvCCL5 is used together with type-1-polarized dendritic cells (DC1) vaccination, along with enhanced immune cell infiltration into the tumor and an obvious T1 skewing of the infiltrating T-cells.
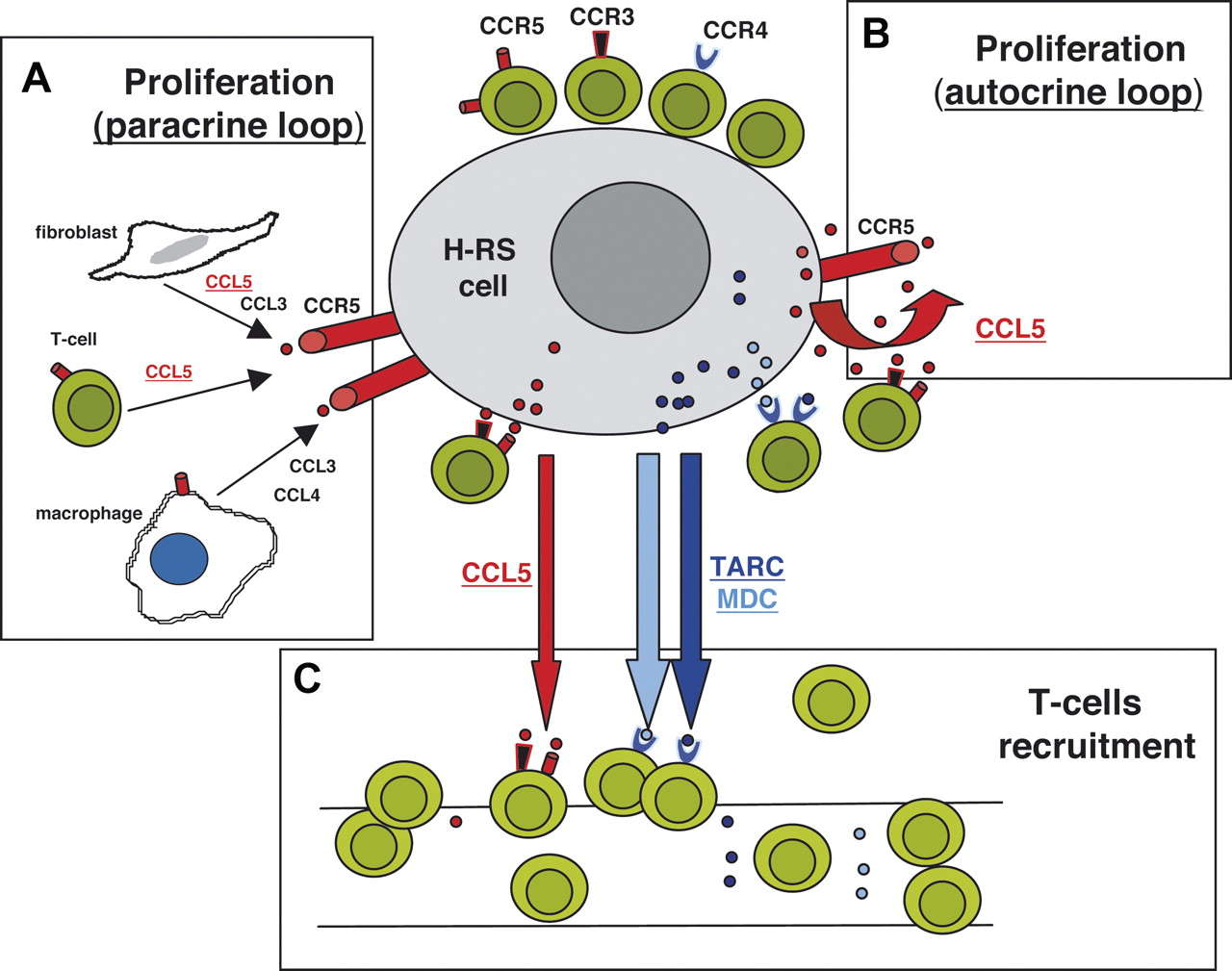 Fig 1. Schematic diagram displaying the involvement of TARC, MDC, and CCL5 in microenvironment formation and RS cell growth. (Aldinucci, D., 2010)
Fig 1. Schematic diagram displaying the involvement of TARC, MDC, and CCL5 in microenvironment formation and RS cell growth. (Aldinucci, D., 2010)
Example 2: Interleukin 2 Expressing Oncolytic Virus
Interleukin 2 (IL-2) is one of the most widely studied cytokines. IL-2 plays its effects on a number of cell types, the most highlighted of which is the T lymphocyte. In fact, one of the most rapid consequences of T cell activation via its antigen receptor is the de novo synthesis of IL-2. Then, a high affinity IL-2 receptor is expressed, therefore permitting rapid and selective expansion of effector T cell populations activated by antigen. Moreover, a main function of IL-2 is to enhance proliferation of CD4C and CD8C T cells. IL-2-induced proliferation takes place through pro-proliferative signals via the proto-oncogenes c-myc and c-fos, in combination with anti-apoptotic signals by Bcl-2 family members. What's more, it has been proven that in addition to anti-apoptotic signals, IL-2 also has effects on cellular metabolism and glycolysis which are necessary for long-term survival of T cells. In the past studies, scientists have demonstrated that IL-2 expression from a systemically delivered oncolytic virus led to decreased oncolytic activity and suboptimal immune activation. They also showed that temporally regulated expression of the inflammatory cytokine IL2 in conjunction with oncolytic vaccinia activity offered greatly promoted therapeutic activity, as well as significantly increased safety profile. In addition, regulating the expression of IL2 leads to improved tumor-specific replication of vvdd (oncolytic vaccinia strain vaccinia virus double deleted).
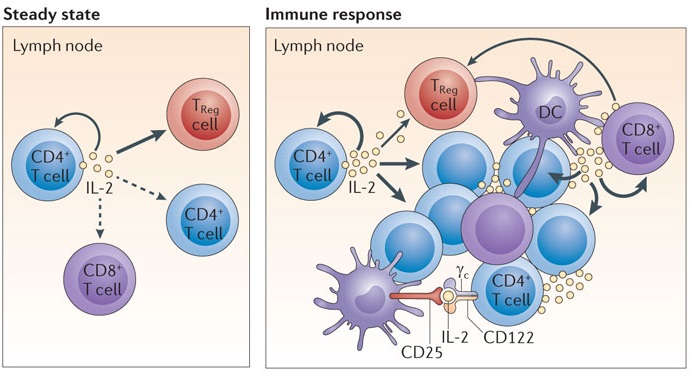 Fig 2. Schematic diagram of IL‑2 homeostasis in steady-state conditions and during an immune response. (Boyman, O. 2012)
Fig 2. Schematic diagram of IL‑2 homeostasis in steady-state conditions and during an immune response. (Boyman, O. 2012)
Other Services in Oncolytic Virus Engineering Platform
- Pathogenicity Manipulation (Attenuation)
- Immunogenicity Manipulation
- Antibody-expressing Oncolytic Virus
- Immune Checkpoint Inhibitor-expressing Oncolytic Virus
With our well-established oncolytic virus engineering platform, the experienced scientists at Creative Biolabs are dedicated to helping you develop unique oncolytic virus. We also provide other various services regarding oncolytic virus development. Please feel free to contact us for more information and a detailed quote.
Reference:
- Chen, H., (2013). "Regulating cytokine function enhances safety and activity of genetic cancer therapies." Molecular Therapy, 21(1), 167-174.
- Aldinucci, D., (2010). "Chemokine receptors as therapeutic tools in Hodgkin lymphoma: CCR4 and beyond." Blood, 115(3), 746-747.
- Boyman, O., (2012). "The role of interleukin-2 during homeostasis and activation of the immune system." Nature reviews. Immunology, 12(3), 180.
