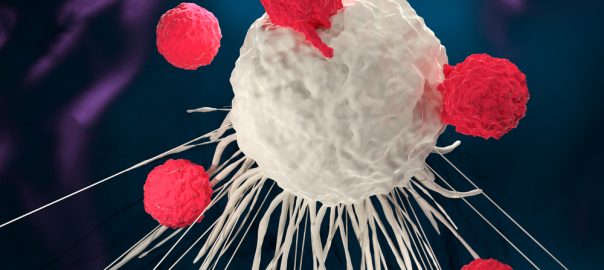
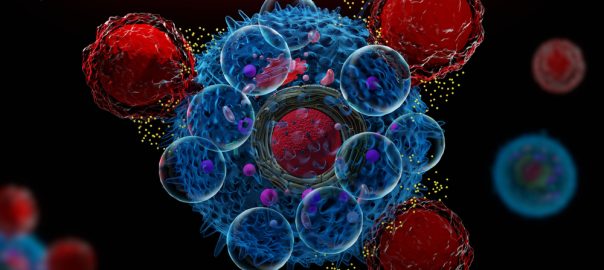
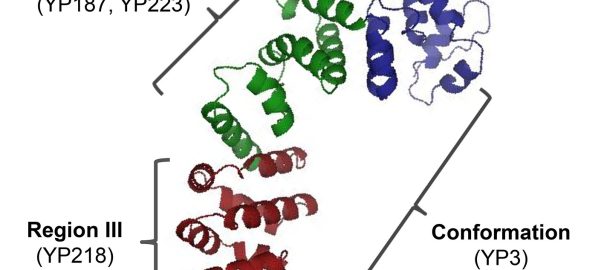
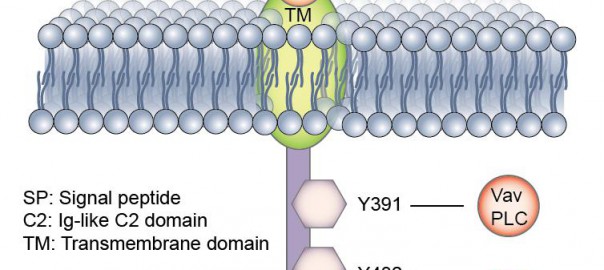
A study at the Stanford University School of Medicine showed that altered immune cells can eliminate a fatal pediatric brain tumor in a mouse model, which was published in the April 16Read More…
Nat Biotechnol: New Generation of CAR-T Can Easily Eliminate Solid Tumors
Huge progress has been made in the CAR-T field recently. Professor Koji Tamada from Yamaguchi University, Department of Immunology, has successfully developed a new generation of CAR-T. Meanwhile, in a variety of solidRead More…
CAR-T Therapy Breakthrough Results Successively Published in Science and Nature
Recently, CAR-T therapy has achieved continuous breakthrough results. First, scientists from the United States and Italy found a better target for CAR-T against a fatal brain tumor. Then, a research team fromRead More…
Oncolytic Virus Significantly Prolong Patients’ Survival in Brain Tumor Treatment
The latest achievement published in Journal of Clinical Oncology showed that a common cold virus could attack deadly brain tumors to prolong patient survival after genetic modification. Researchers from the MD AndersonRead More…
Carl June: The World’s First CAR-T Therapy Released the Latest Data
On February 1, NEJM, the top medical journal, published the results of a pivotal multi-center critical phase 2 trial in support of the “first CAR-T therapy Tisagenlecleucel listed”. The overall survival of patientsRead More…
CAR T Cells Manufacture Upgrades
Chimeric antigen receptor, or CAR, T cells have considerably improved cancer treatment outcomes for certain malignancies. CAR T cells are produced when T cells isolated from the patient are modified to targetRead More…
What Is Mesothelin?
As we all know, CD19 is the most frequently used target marker for CAR-T cell therapy clinical trials. But not much people knows about the second frequently used target biomarker is mesothelin. What’s more,Read More…
What Is CD19 Antigen?
As the data from clinicaltrials.gov shows, CD19 antigen is the most frequently used biomarker in CAR-T cell therapy clinical trials for hematological malignancy such as leukemia and lymphoma. According to our statistics report, overRead More…
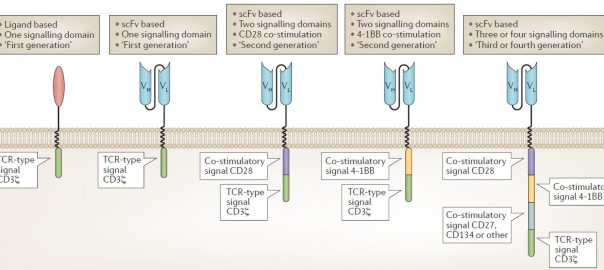
What the Next Generation T Cell Therapy Looks Like
T cell receptors (TCRs) provide a recognition signal for T cells complemented by a co-stimulatory signal that can provide an on/off signal to regulate the activation of T cells. Since Medawar andRead More…
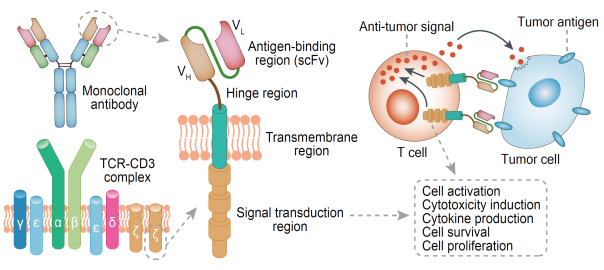
What Is the Important Role of the Transmembrane Spacer Domain in CAR Design?
As we know, a typical design of 2rd generation Chimeric antigen receptor contains 3 functional units: a recognition domain, a transmembrane spacer domain and an intracellular signal transduction domain. Antigen recognition domainRead More…