Anti-CD20/IL-21 Immunocytokine Development Service
Creative Biolabs provides a proprietary, integrated platform for the development and validation of customized Anti-CD20/IL-21 immunocytokine candidates. We cover all critical stages: from in silico design and high-yield mammalian expression to purification and CMC documentation. Clients gain significant time savings and a de-risked asset, receiving a meticulously characterized drug substance with documented superior therapeutic potential. This accelerates their program toward subsequent advanced preclinical and regulatory milestones.
Introduction What We Can Offer Workflow Why Creative Biolabs Customer Reviews FAQs Related Services Contact Us
Precision-Targeted CD20/IL-21 Immunocytokine Development
The Anti-CD20/IL-21 IC strategy is a cutting-edge approach to overcome chemo- and Rituximab resistance in B-cell cancers. Immunocytokines resolve the two major limitations of systemic cytokine therapy-short half-life and dangerous off-target toxicity leveraging the Anti-CD20 antibody to concentrate the potent IL-21 payload exclusively at the tumor site. Published data confirm IL-21's superiority over IL-2 by preferentially activating NK and CTL cells while mitigating the systemic expansion of inhibitory Tregs. This targeted delivery is confirmed by advanced-stage IC programs, lending strong credibility to this next-generation approach.
Request a consultation for more information and to discuss your project.
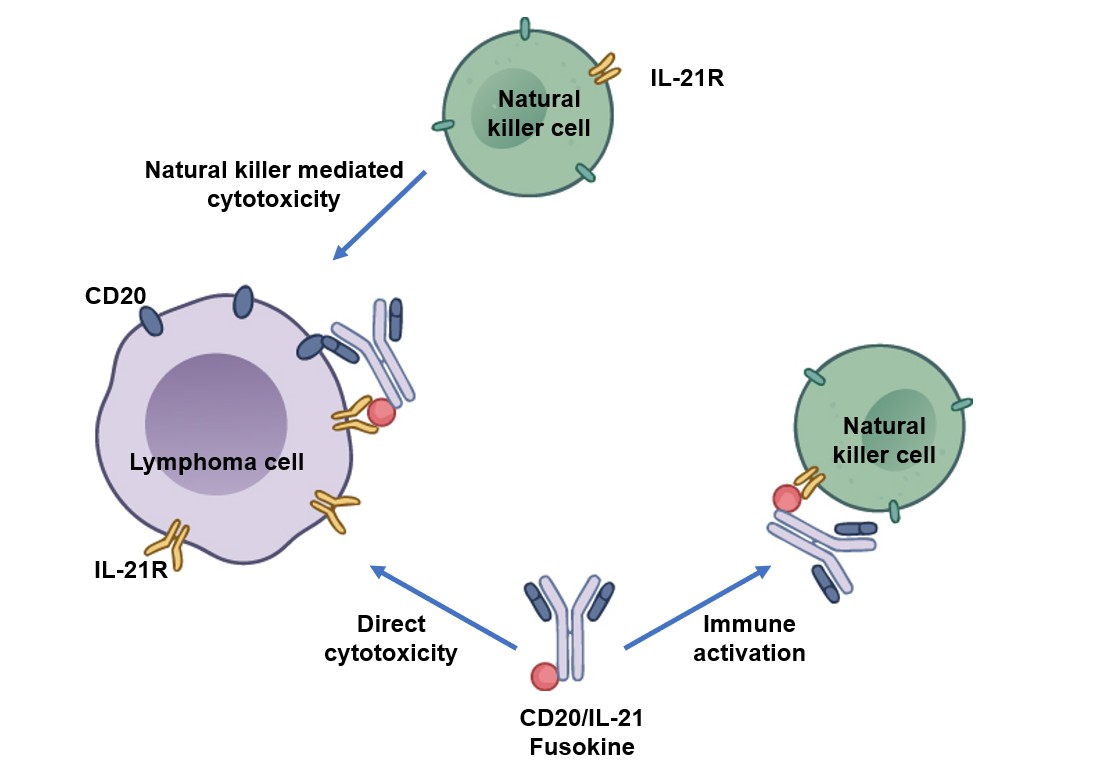 Fig.1 IL-21 conjugated to an anti-CD20 antibody provides 2 additional mechanisms of cytotoxicity. 1
Fig.1 IL-21 conjugated to an anti-CD20 antibody provides 2 additional mechanisms of cytotoxicity. 1
What We Can Offer
Fully Customizable Construct Design
We integrate your proprietary Anti-CD20 antibody sequence with customized IL-21 payloads, including specific stability-enhancing mutations (e.g., R9E, R76A), for a truly bespoke therapeutic asset, ensuring maximum specificity and efficacy.
Superior Therapeutic Window by Design
Engineering the IL-21 payload to proactively mitigate the off-target systemic toxicity and peripheral regulatory T-cell expansion associated with legacy IL-2 analogues, yielding a safer candidate for further development.
Validated Against Clinical Resistance
Guaranteed functional validation in specialized in vitro models that mimic Rituximab-refractory B-cell malignancies, ensuring your lead candidate is effective against the toughest biologically challenging phenotypes right from the start.
Accelerated Half-Life Optimization
Utilization of proprietary linker chemistries and sequence modification expertise to dramatically prolong the IC's systemic half-life, ensuring peak, sustained IL-21 concentration at the tumor site.
Anti-CD20/IL-21 Immunocytokine Development Service at Creative Biolabs
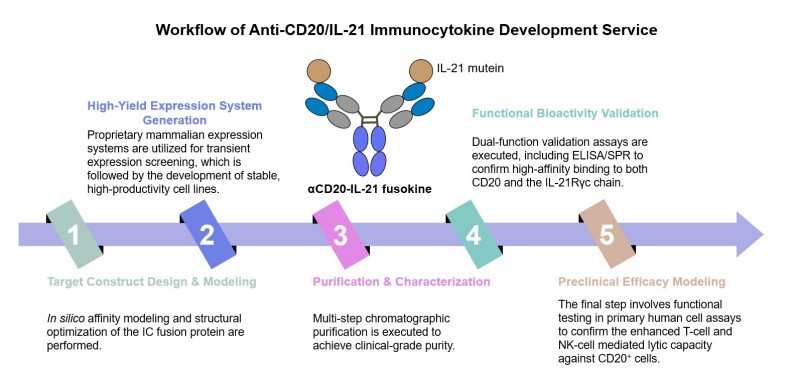
Why Choose Us?
|
Core Advantages
|
Unique Features
|
|
IL-21-Specific Expertise
|
We leverage published data that confirms the IL-21 payload offers a wider therapeutic window and avoids the undesired peripheral Treg expansion commonly associated with IL-2 analogues.
|
|
Stability Engineering
|
We utilize validated linker technologies and sequence mutations known to dramatically prolong the IL-21 systemic half-life, ensuring maximum concentration at the tumor site before clearance.
|
|
Validated Against Resistance
|
Our core assay suite includes specialized functional models using rituximab-refractory B-cell lines, guaranteeing your lead candidate is tested against the most relevant and biologically challenging phenotype.
|
To experience the Creative Biolabs advantage, request a formal quote today.
Customer Reviews
-
Unprecedented Potency
Using Creative Biolabs' service in our research has significantly improved the elimination of rituximab-refractory B-cells. The IC fusion protein showed an increase in STAT3 signaling specificity compared to our internal constructs, allowing us to proceed to advanced development with confidence. - Dr. Ki****a
-
Superior Therapeutic Window
Creative Biolabs' service provided an optimally engineered construct that drastically reduced systemic IL-21 activity while maintaining potent local function. This is a critical advantage over IL-2 analogues, enabling us to project safer dosing in advanced preclinical trials. - Je****y W.
FAQs
Q:
How does the Anti-CD20/IL-21 IC compare to a standard Anti-CD20 monoclonal antibody like Rituximab?
A:
Standard anti-CD20 antibodies primarily rely on ADCC/CDC for B-cell depletion. The IC retains these functions but adds a second, highly potent mechanism: localized immune cell priming via IL-21 signaling. This dual-action approach is proven to kill cells that are Rituximab-refractory, offering a necessary solution for resistant disease.
Q:
What is the primary advantage of IL-21 over IL-2 in an Immunocytokine format?
A:
While both are T-cell stimulants, IL-2 ICs carry a significant risk of systemic toxicity due to the expansion of peripheral regulatory T-cells, which dampens the anti-tumor response. IL-21 is associated with a much wider therapeutic window and preferentially enhances CTL and NK activity, driving a superior, less toxic immune response.
Related Services
Bispecific Antibody Engineering
For clients interested in targeting two antigens simultaneously (e.g., CD20 and another tumor-associated antigen), we offer advanced bispecific IC construction.
Learn More →
Immunostimulatory Cytokine-based Cancer Immunotherapy Development
An alternative to fusion proteins, this service focuses on developing stabilized cytokine-anti-cytokine complexes (ICx) to achieve targeted delivery and extended half-life with unique receptor signaling advantages.
Learn More →
How to Contact Us
Creative Biolabs is uniquely positioned to deliver an optimally engineered, highly stable, and functionally validated IC construct, leveraging our expertise in complex therapeutic biologics and our commitment to accelerated regulatory timelines. Contact us today for a confidential consultation on optimizing your Anti-CD20/IL-21 IC strategy.
For Research Use Only | Not For Clinical Use


 Fig.1 IL-21 conjugated to an anti-CD20 antibody provides 2 additional mechanisms of cytotoxicity. 1
Fig.1 IL-21 conjugated to an anti-CD20 antibody provides 2 additional mechanisms of cytotoxicity. 1

