About This Program
This program aims to develop anti-CD40 therapeutic monoclonal antibody for immuno-oncology.
CD40 agonist mAb can upregulate MHCII, CD80, and CD86 on DCs and macrophages, promote CD8+ T cell activation in vivo, and likely alter TME myeloid component. Besides, induction of CD40 signaling on DCs using agonistic anti-CD40 antibodies can substitute for CD4+ T-cell help and directly stimulate a specific CD8+ CTL response, highlighting a clear rationale for CD40-based cancer immunotherapy. In this case, anti-CD40 antibody has great potentials to be widely applied in the treatment of various diseases with high efficacy.
CD40
CD40 is a member of the tumor necrosis factor (TNF) receptor superfamily and is expressed in many immune cell types including DCs, macrophages, B cells and NK cells. CD40 signaling induced by CD40L-expressing T helper (Th1) cells on CD40-expressing antigen presenting cells (APCs) is critical for APC licensing. Here, we show the CD40-CD40L interactions at DC-T cell and T cell-T cell interface (see fig1).
-
Using CD4+ T-helper cells, CD40L mediates CD40 costimulation of DCs and is critical for generation of antitumor T-cell responses.
-
The binding to the natural ligand CD40L, which is expressed on T helper cells, leads to APC activation and thus enhancement of immune responses.
-
Cognate interactions between CD40 and CD40 affect humoral immunity, cell mediated immunity, and inflammation.
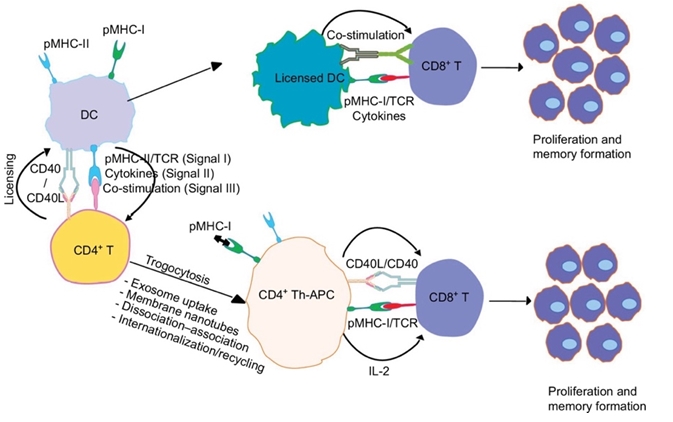 Fig.1 Fig.1 CD40-CD40L interactions at DC-T cell and T cell-T cell interface.1
Fig.1 Fig.1 CD40-CD40L interactions at DC-T cell and T cell-T cell interface.1
Anti-CD40 Antibody in Cancer Studies
Anti-CD40 antibody is often used in combination with other immunology agents in clinical researchs to achieve anti-tumor effects. Here are some published data about CD40 work as a potential target for cancer immunotherapy.
-
A CD40 agonist increases antitumor efficacy of vaccine + anti-PD-1 therapy (triple therapy).
-
The effect of an agonist CD40 antibody (CDX-1140) therapy on bladder cancer (cell line EJ138).
Indication
Based on the published data, CD40 antibodies are being tested as treatment for malignancies such as chronic lymphatic leukemia (CLL), Multiple Myeloma (MM), and non-Hodgkin's lymphoma (NHL). Therefore, we hope to develop multiple mAb programs that apply to different indications (not limited to one specific tumor type), of which CD40 is highly expressed.
Clinical Trials under Progress
Currently, several anti-GITR therapeutic monoclonal antibodies (Dacetuzumab and Lucatumumab are in the most advanced stage) are tested in clinical trials for several types of solid tumors. An increasing number of agonistic mAbs is getting confirmed on its role to stimulate T cells and early clinical trial data shows great promise. However, the efficacy, safety, and combination strategies with anti-ICOS agonists or antagonists have not yet to be specified.
In this case, CD40 is a compelling target for cancer immunotherapy. In an effort to optimally leverage CD40-mediated immune response, the next generation of ICOS targeting treatment attempts to explore combination therapy trials with other immunomodulatory agents.
Program Plan
We have extensive knowledge for end-to-end program development. For each program, we are committed to delivering the final complete program to our clients within 1.5 years prior to entering the IND stage.
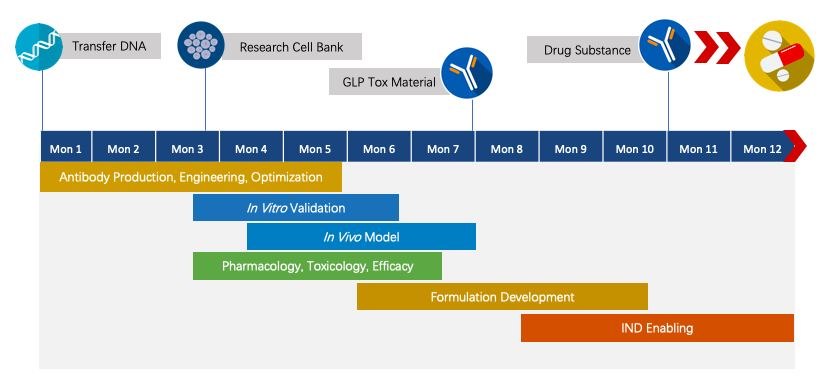 Fig.2 The timeline of Next-IOᵀᴹ programs.
Fig.2 The timeline of Next-IOᵀᴹ programs.
Cooperation
Creative Biolabs is looking for potential partners (include but not limit to major pharma or biotech firms) to develop anti-CD40 Therapeutic Monoclonal antibody program together. Our scientists are dedicated to bringing together years of valuable experience to our partner and achieve a meaningful partnership. By doing so, we wish to help both parties to proceed with IND and many stages of clinical trials beyond.
If you are interested, please feel free to contact us so that we can discuss the program and other possible opportunities for cooperation. Look forward to working with you in the near future.


 Fig.1 Fig.1 CD40-CD40L interactions at DC-T cell and T cell-T cell interface.1
Fig.1 Fig.1 CD40-CD40L interactions at DC-T cell and T cell-T cell interface.1
 Fig.2 The timeline of Next-IOᵀᴹ programs.
Fig.2 The timeline of Next-IOᵀᴹ programs.
 Download our brochure
Download our brochure