Next-IO™ Anti-CD73 Therapeutic Monoclonal Antibody Program
About This Program
This program aims to develop anti-CD73 therapeutic monoclonal antibody for immuno-oncology.
CD73 is reported to be overexpressed on tumors and immune cells. Upregulation of CD73 is associated with overproduction of adenosine. In addition, it inhibits anti-tumor immune responses and contributes to proliferation, angiogenesis, and metastasis. These data fully indicate that CD73 may be involved in cancer progression and could be a promising target of next generation cancer immunotherapy.
CD73
CD73, also known as NT5E, is expressed on tumor cells and host immunosuppressive cells, where it converts 5'-AMP into adenosine. Adenosine suppresses antitumor immunity by binding to its receptors, which indirectly suggests the role of CD73 in cancer treatment.
Highlights of CD73 for Cancer Immunotherapy:
-
Overexpression of CD73 in cancer cells results in high levels of adenosine in the tumor site.
-
Adenosine at the tumor site can enhance tumor cell growth and angiogenesis, and inhibit regional immunity.
-
Inhibition of CD73 prevents tumor progression.
-
Anti-CD73 monoclonal antibody and drugs in clinical trials have achieved great results.
-
Blocking CD73 can be considered a reasonable anti-cancer treatment strategy.
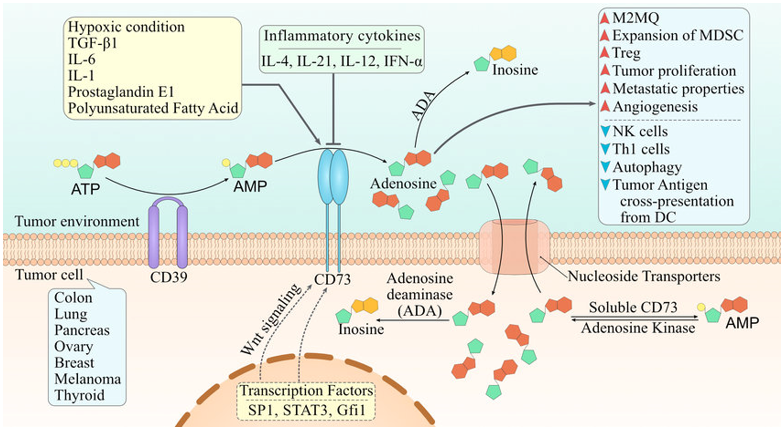 Fig.1 CD73 in the tumor microenvironment. (Ghalamfarsa, 2019)
Fig.1 CD73 in the tumor microenvironment. (Ghalamfarsa, 2019)
CD73 in Cancer Studies
Anti-CD73 antibody is often used in combination with other immunology agents in clinical research to achieve anti-tumor effects. Here are some published data about CD73 molecule work as a potential target for cancer immunotherapy.
-
Combination therapy with anti-CD73 antibody and DTXL significantly inhibits tumor growth and reduces lung metastasis of ovarian cancer.
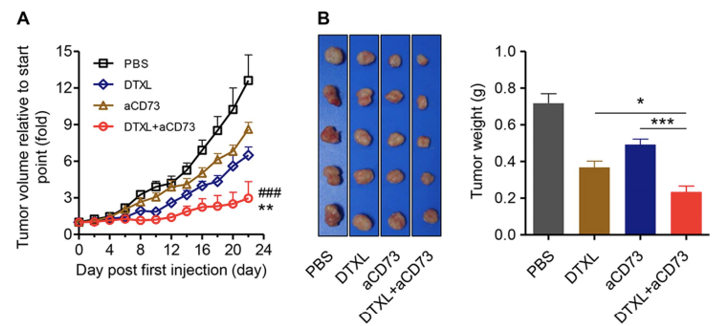 (Li, 2019)
(Li, 2019)
-
Combination therapy of CD73 blockade and anti-4-1BB facilitates tumor regression.
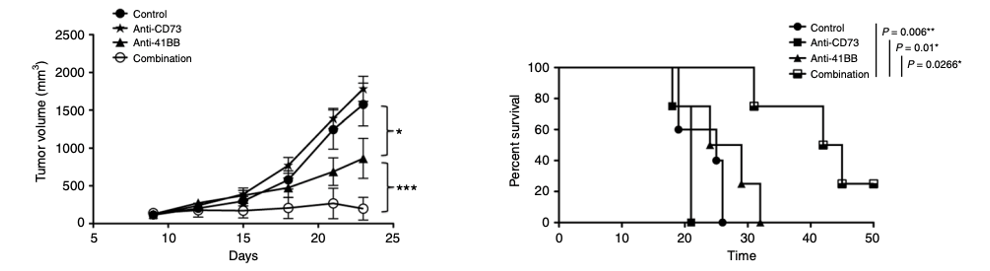 (Chen, 2019)
(Chen, 2019)
Indication
According to published data, CD73 has been shown to be widely expressed in a variety of cancers, including breast cancer, prostate cancer, gastrointestinal cancer, and lung cancer and so on. In our case, we hope to develop multiple mAb programs that apply to different indications (not limited to one specific tumor type), in which CD73 is highly expressed.
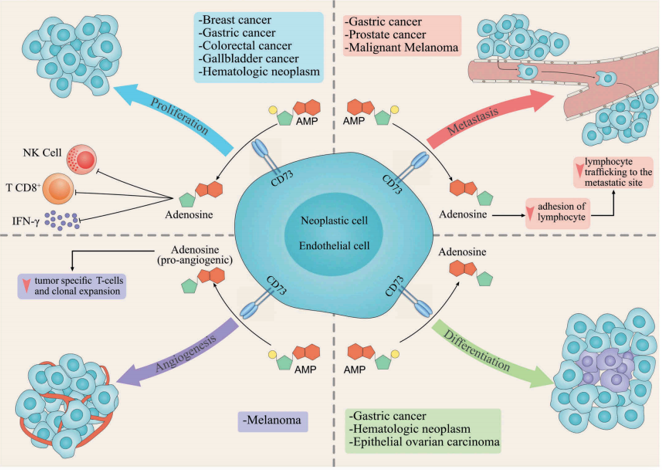 Fig.2 The role of CD73 in tumor development and its underlying molecular mechanisms. (Ghalamfarsa, 2019)
Fig.2 The role of CD73 in tumor development and its underlying molecular mechanisms. (Ghalamfarsa, 2019)
Clinical Trials under Progress
Currently, several studies involving CD73 therapeutic mAbs are undergoing early clinical trials for the treatment of various solid malignancies. Accumulating preclinical evidence supports their clinical development. However, further studies are needed to identify the efficacy and safety of the combination strategies of CD73 therapeutic mAbs.
In this case, CD73 still owns a promising market prospect in the future. In particular, there is increasing interest in combining anti-CD73 with other agents, thus representing a particularly promising approach to achieving greater therapeutic success in various cancers.
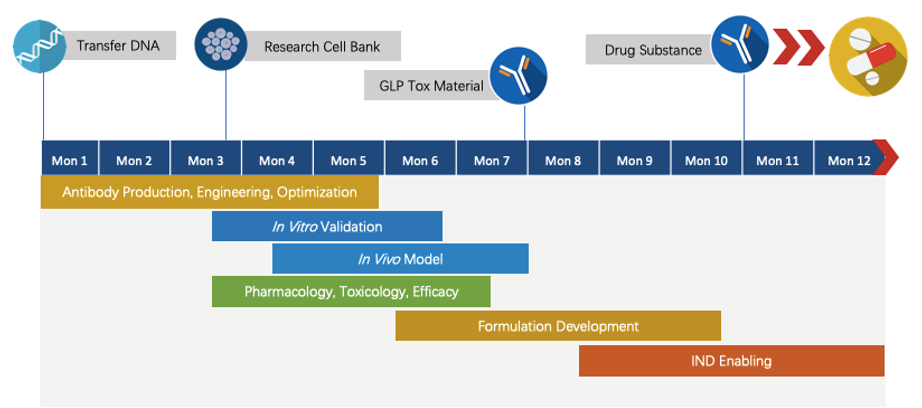
Program Plan
We have extensive knowledge of end-to-end program development. For each program, we are committed to delivering the final complete program to our clients within 1.5 years prior to entering the IND stage.
Cooperation
Creative Biolabs is looking for potential partners (include but not limit to pharma or biotech firms) to develop anti-CD73 therapeutic monoclonal antibody program together. Our scientists are dedicated to bringing together years of valuable experience to our partner and achieve a meaningful partnership. By doing so, we wish to help both parties to proceed with IND and many stages of clinical trials beyond.
If you are interested, please feel free to contact us so that we can discuss the program and other possible opportunities for cooperation. Look forward to working with you in the near future.
References
-
Ghalamfarsa, G.; et al. CD73 as a potential opportunity for cancer immunotherapy. Expert opinion on therapeutic targets. 2019, 23(2): 127-142.
-
Li, H.; et al. Blockade pf CD73/adenosine axis improves the therapeutic efficacy of docetaxel in epithelial ovarian cancer. Archives of Gynecology and Obstetrics. 2019, 299(6): 1737-1746.8.
-
Chen, S.; et al. CD73 expression on effector T cells sustained by TGF-β facilitates tumor resistance to anti-4-1BB/CD137 therapy. Nat Commun. 2019, 10(1): 150.
For Research Use Only | Not For Clinical Use


 Fig.1 CD73 in the tumor microenvironment. (Ghalamfarsa, 2019)
Fig.1 CD73 in the tumor microenvironment. (Ghalamfarsa, 2019)
 (Li, 2019)
(Li, 2019)
 (Chen, 2019)
(Chen, 2019)
 Fig.2 The role of CD73 in tumor development and its underlying molecular mechanisms. (Ghalamfarsa, 2019)
Fig.2 The role of CD73 in tumor development and its underlying molecular mechanisms. (Ghalamfarsa, 2019)

 Download our brochure
Download our brochure