Our Anti-FGL1 monoclonal antibody program aims to develop therapeutic monoclonal antibodies that could recognize and potentially, target against FGL1 (Fibrinogen-like Protein 1). The highlight feature of this program, FGL1, was only identified in 2019. We believe our program will be fairly unique and is a leading program in current bio-pharmaceutical market.
Fibrinogen-like Protein 1 (FGL1)
FGL-1 belongs to fibrinogen family since it’s structurally related to fibrinogen. And in late 2019, FGL-1
was identified as a major immune inhibitory ligand of Lymphocyte Activation Gene 3 protein (LAG-3, see
Fig.1).
-
Being independent of MHC-II, FGL1 is a major ligand of LAG-3 that medicates T cell suppression;
-
FGL1 is normally released at a very low level in the liver. Whereas the number will be higher at cancer patients;
-
Blocking FGL1-LAG-3 interaction proposes anti-tumor immunity;
-
FGL1-LAG3 is completely independent of PD1-PDL1 pathway.
 Fig.1 Mechanism of action for FGL1-LAG3 in T cell suppression.1
Fig.1 Mechanism of action for FGL1-LAG3 in T cell suppression.1
Published Data
• In NSCLC and Melanoma, high level of FGL1 is related with poor prognosis/survival rate..
 Fig.2 High level of FGL1 is related with poor prognosis/survival rate.1
Fig.2 High level of FGL1 is related with poor prognosis/survival rate.1
• The expression value of FGL1 is significantly higher, compared to normal tissue, among various type of
cancer.
 Fig.3 The expression value of FGL1 is significantly higher among various type of cancer.1
Fig.3 The expression value of FGL1 is significantly higher among various type of cancer.1
• Treatment with anti-FGL1 antibody shows higher survival rate compared to the control group, and the efficacy of anti-FGL1 plus anti-B7-H1 therapy is better in other treatments. These findings may indicate that anti-FGL1 has anti-tumor efficacy.
 Fig.4 The anti-tumor efficacy of anti-FGL1 antibody.1
Fig.4 The anti-tumor efficacy of anti-FGL1 antibody.1
Non-Small Cell Lung Cancer (NSCLC)
Lung cancer, characterized by uncontrolled cell growth in lung tissues, is the most common type of cancer
for several decades. About 80% to 85% lung cancers are non-small cell lung cancer (NSCLC). To date, several therapeutic antibodies on market can be used to treat NSCLC. And scientists are trying to find
more therapeutic targets with a better anti-tumor efficacy.
• Based on Cancer Statistics, 2017, lung cancer is the second most common cancer in the United States and is
one of the leading causes of cancer death.
• Preliminary Data of Anti-FGL1 Efficacy to Treat NSCLC
• FGL1 is highly expressed in patients with NSCLC (left), and high level FGL1 in NSCLC is related with poor
prognosis(right).
 Fig.5 The expression level of FGL1 in NSCLC.1
Fig.5 The expression level of FGL1 in NSCLC.1
• Treatment with anti-FGL1 shows better anti-NSCLC efficacy, especially when used in combination with anti-B7-H1.
 Fig.6 Efficacy of the combination of anti-FGL1 and anti-B7-H1.1
Fig.6 Efficacy of the combination of anti-FGL1 and anti-B7-H1.1
Clinical Trials under Progress
• There are NO clinical trials examining FGL1 as the target. Our program will be a pioneer in this field.
• FGL1 is a novel target among all the other immune checkpoint inhibitors. It is believed to be a shining star and the next marketing prospect in the field. From our standpoint, since the combination efficacy with anti-B7-H1 (PD-L1) is more promising as shown by the preliminary data, our therapeutic anti-FGL1 discovery will focus on combination therapy with anti-B7-H1 (PD-L1).



 Fig.1 Mechanism of action for FGL1-LAG3 in T cell suppression.1
Fig.1 Mechanism of action for FGL1-LAG3 in T cell suppression.1
 Fig.2 High level of FGL1 is related with poor prognosis/survival rate.1
Fig.2 High level of FGL1 is related with poor prognosis/survival rate.1
 Fig.3 The expression value of FGL1 is significantly higher among various type of cancer.1
Fig.3 The expression value of FGL1 is significantly higher among various type of cancer.1
 Fig.4 The anti-tumor efficacy of anti-FGL1 antibody.1
Fig.4 The anti-tumor efficacy of anti-FGL1 antibody.1
 Fig.5 The expression level of FGL1 in NSCLC.1
Fig.5 The expression level of FGL1 in NSCLC.1
 Fig.6 Efficacy of the combination of anti-FGL1 and anti-B7-H1.1
Fig.6 Efficacy of the combination of anti-FGL1 and anti-B7-H1.1

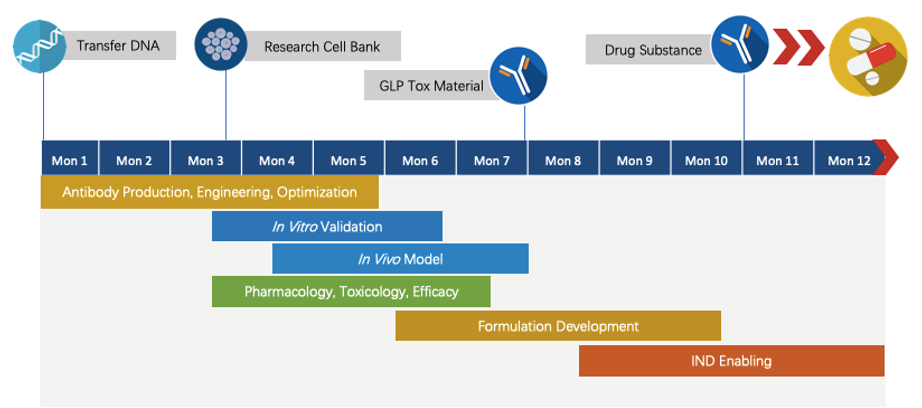 Fig.7 The timeline of Next-IO™ programs.
Fig.7 The timeline of Next-IO™ programs.

 Download our brochure
Download our brochure