Next-IO™ Anti-GPC3 Therapeutic CAR-T Cell Program
About This Program
This program aims to develop anti-GPC3 therapeutic CAR-T cells for immuno-oncology.
Rationale when developing the program:
-
Hepatocellular carcinoma (HCC) is a common form of liver cancer that begins in hepatocytes, i.e the basic liver cell, and it accounts for 90% of primary liver cancers. One data suggests it is one of the most deadly cancers in Asia. To date, the 5-year survival rate for patients with liver cancer remains low at approximately 10%. A new but potent strategy is urgently needed for the field.
-
Glypican-3 (GPC3) is a heparan sulfate proteoglycan expressing in 75% of HCC tissues but not in healthy liver or other normal tissues.
-
GPC3-CAR T cells have been shown to have positive results regarding its elimination activity against HCC cell lines in vivo.
For the reasons mentioned above, we believe GPC3 is a suitable target for CAR T cell therapy.
GPC3
GPC3 belongs to heparin sulfate proteoglycan family, can be immobilized on the cell surface by glycosylphosphatidylinositol anchors. GPC3 can be expressed in a variety of tissues during embryonic development. Beyond that life state, it is a highly tumor-specific antigen in which expression is severely inhibited in most adult tissues.
Highlighted Functions:
-
Elevated GPC3 expression is reported in a variety of tumor types, such as liver, lung, stomach, ovarian, esophageal, and other forms of cancers, indicating GPC3 is an ideal target to be studies in patients with solid tumors.
-
In particular, GPC3 is currently used as a diagnostic biomarker to distinguish HCC from normal liver tissue, benign liver tumors, and other types of metastatic cancer.
-
There is sufficient evidence to suggest that GPC3 is involved in the malignant transformation of HCC.
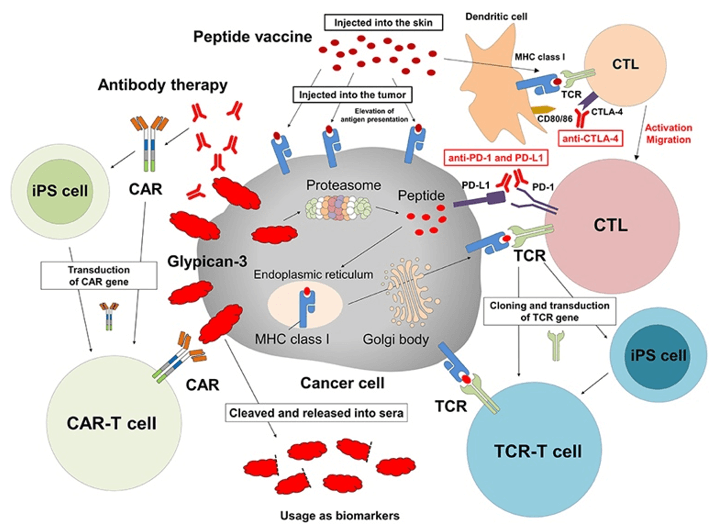 Fig.1 Cancer immunotherapy targeting GPC3. (Shimizu, 2019)
Fig.1 Cancer immunotherapy targeting GPC3. (Shimizu, 2019)
GPC3 in Cancer Studies
Here are some published data about GPC3 working as a potential target for cancer immunotherapy.
-
Anti-GPC3-CAR T cells inhibit tumor cell growth in xenograft tumors from the studies on patients with HCC.
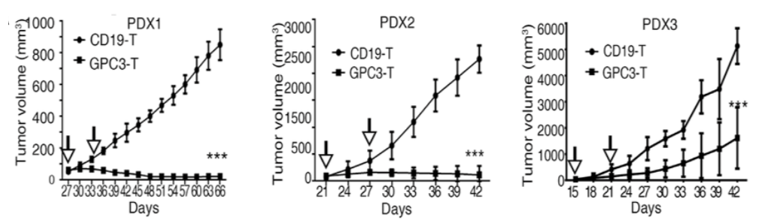 (Jiang, 2019)
(Jiang, 2019)
-
Destruction of PD-1 enhances the anti-tumor activity of GPC3 CAR-T cells against HCC.
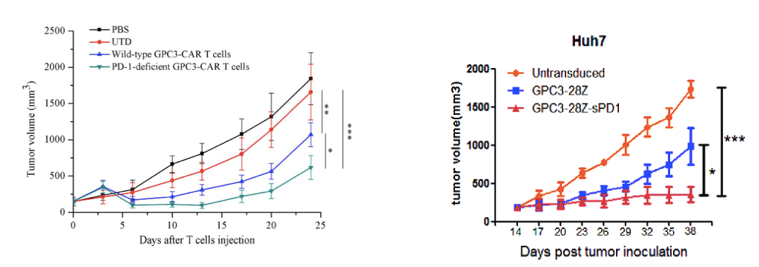 (Guo, 2018)
(Guo, 2018)
Ongoing Clinical Trials
Program Planning and Management
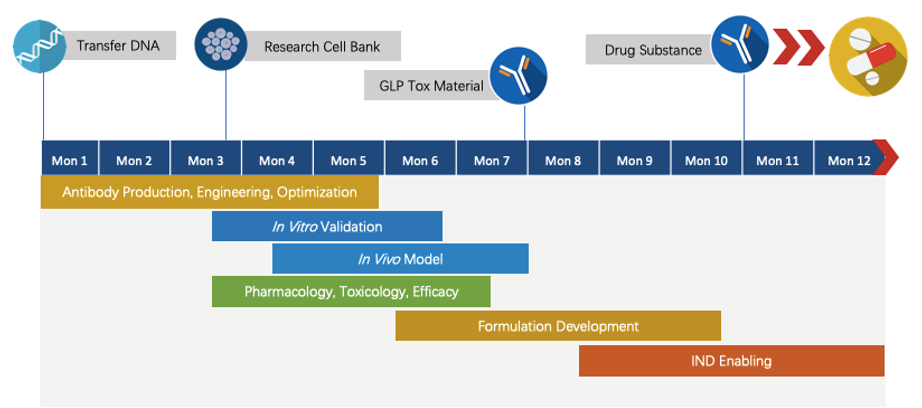
Creative Biolabs has extensive knowledge of end-to-end program development. For each program, we are committed to delivering the final complete program to our clients within 1.5 years before entering the IND stage.
Cooperation
Creative Biolabs is looking for potential partners (include but not limit to major pharma or biotech firms) to develop anti-GPC3 therapeutic CAR-T program together. Our scientists are dedicated to bringing years of valuable experience to our partner and achieve a meaningful partnership together. For any partners interested in our Next-IO™ programs, Creative Biolabs welcomes collaboration.
Here are two ways for your choice, and please contact us for more details.
1) Collaborate with us and co-develop the programs from the discovery phase to IND enabling. Costs will be shared.
2) Become a licensed candidate for our programs.
With our quality control protocol and knowledge of global regulatory requirements, we can help our partners to advance their programs with more chance to succeed. Look forward to cooperating with you in the near future.
References
-
Shimizu, Y.; et al. Next-generation cancer immunotherapy targeting glypican-3. Frontiers in oncology. 2019, 9: 248.
-
Jiang, Z.; et al. Anti-GPC3-CAR T cells suppress the growth of tumor cells in patient-derived xenografts of hepatocellular carcinoma. Frontiers in immunology. 2017: 690.
-
Guo, X.; et al. Disruption of PD-1 enhanced the anti-tumor activity of chimeric antigen receptor T cells against hepatocellular carcinoma. Frontiers in pharmacology. 2018, 9: 1118.
For Research Use Only | Not For Clinical Use


 Fig.1 Cancer immunotherapy targeting GPC3. (Shimizu, 2019)
Fig.1 Cancer immunotherapy targeting GPC3. (Shimizu, 2019)
 (Jiang, 2019)
(Jiang, 2019)
 (Guo, 2018)
(Guo, 2018)

 Download our brochure
Download our brochure