Next-IO™ PSCA x CD3 Therapeutic Bispecific Antibody Program
About This Program
This program aims to develop PSCA x CD3 therapeutic bispecific antibody for bladder carcinoma immunotherapy.
Rationale for our program:
-
Prostate cancer (PC) is the most common non-skin malignancy in men, but effective treatment options for patients with metastatic prostate cancer are limited. Therefore, novel treatment options are urgently needed.
-
Prostate stem cell antigen (PSCA) has been identified as a promising target for advanced prostate cancer immunotherapy. PSCA expression was detected in approximately 90% of primary PCs.
-
T-cell engagers are a growing class of biotherapeutics. Emerging clinical data reveals the importance of improving the safety of T-cell engagers.
PSCA x CD3
PSCA is a cell surface protein that binds to the surface of the membrane via a glycosylphosphatidylinositol (GPI)-anchor. PSCA is upregulated in several refractory cancers, including prostate, bladder and pancreatic cancer.
Highlight functions:
-
The expression of PSCA is positively associated with advanced clinical stage and metastasis of prostate cancer and is also associated with malignant progression of pre-clinical lesions.
-
PSCA has been proposed as a biomarker for tumor diagnosis and prognosis, as well as therapeutic targets for these cancers.
-
PSCA has also shown clinical potential as a prostate-specific antigen in immunotherapy, which, when presented by dendritic cells, can elicit strong tumor-specific immunity.
Prostate Cancer
-
Prostate cancer is the third leading cause of cancer-related deaths in the United States. Effective treatment options for patients with prostate cancer are limited.
-
The rise in global disease prevalence is driving the need for prostate cancer diagnosis. According to the National Cancer Institute, in 2018, an estimated 64,690 new cases were diagnosed.
-
The global prostate cancer diagnosis market is valued at US$2.5 billion in 2018 and is expected to grow at a compound annual growth rate of 12.3% during the forecast period.
Ongoing Clinical Trials
-
Currently, there are still NO PSCA x CD3 BiAbs entering the clinical stage.
-
We believe that this dual targeting strategy will provide insights into the tumor immunotherapy, especially in the treatment of BC. In an effort to optimally leverage PSCA x CD3-mediated immune response, our next-IO™ PSCA x CD3 targeted antibody program attempts to explore the optimal combination strategy by involving other immunomodulatory agents.
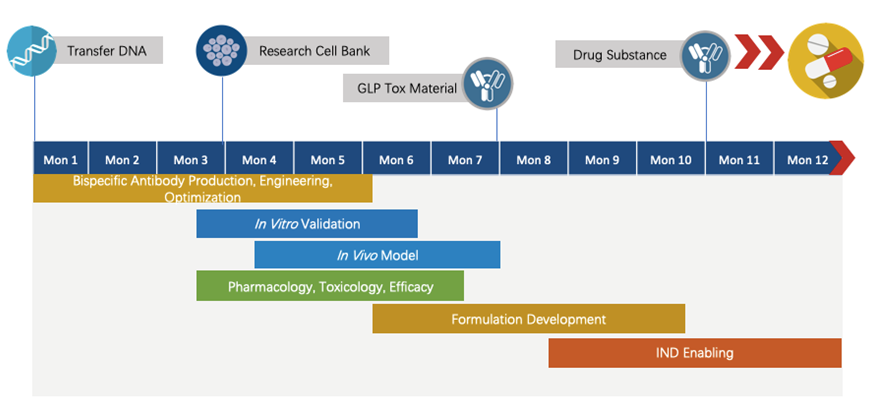
Program Plan
Creative Biolabs has extensive knowledge of end-to-end program development. For each program, we are committed to delivering the final complete program to our clients within 1.5 years before entering the IND stage.
Cooperation
Creative Biolabs is looking for potential partners (include but not limit to major pharma or biotech firms) to develop PSCA x CD3 therapeutic bispecific antibody program together. Our scientists are dedicated to bringing years of valuable experience to our partner and achieve a meaningful partnership together. For any partners interested in our Next-IO™ programs, Creative Biolabs welcomes collaboration.
Here are two ways for your choice, and please contact us for more details.
1) Collaborate with us and co-develop the programs from the discovery phase to IND enabling. Costs will be shared.
2) Become a licensed candidate for our programs.
With our quality control protocol and knowledge of global regulatory requirements, we can help our partners further their programs with more chances to succeed. Look forward to cooperating with you in the near future.
For Research Use Only | Not For Clinical Use



 Download our brochure
Download our brochure