Drug Distribution Analysis Service
At Creative Biolabs, we provide comprehensive distribution analysis services and the quantitative data required to move beyond the apparent volume of distribution to help our clients understand the pharmacokinetic behavior of their drugs. Our team of experienced scientists has extensive knowledge in drug distribution analysis and offers consulting services to help you tailor drug distribution assay solutions and interpret the results of your distribution assays.
Introduction What We Can Offer How We Can Help Why Creative Biolabs Customer Reviews FAQs Related Services Contact Us
Drug Distribution Analysis for Pharmacokinetic Properties
Drug distribution specifically refers to that a drug is spread and transport throughout the various tissues and organs in the body after it is absorbed into the bloodstream. Drug distribution analysis in vitro is a crucial step in the drug development process. The distribution assays help researchers predict how a drug will behave in vivo, or in a living organism. It is crucial to understand the pharmacokinetic properties of a drug and determine the effective dose of a drug, its safety profile, and its potential therapeutic efficacy.
Discover how we can advance your program - Request a consultation.
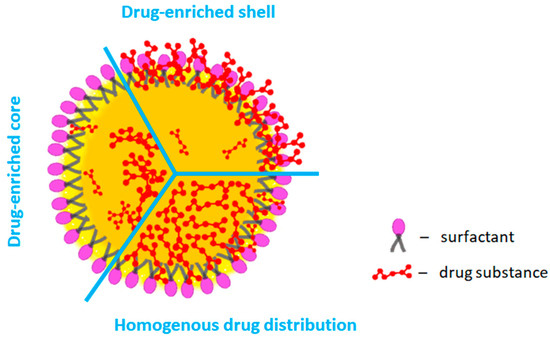 Fig.1 Models of probable drug substance distribution patterns in SLMs. 1
Fig.1 Models of probable drug substance distribution patterns in SLMs. 1
What We Can Offer
At Creative Biolabs, our drug distribution analysis services are designed to simulate the physiological conditions of the human body, providing insights into how drugs interact with tissues and organs. Using state-of-the-art instrumentation and validated methods, we analyze the distribution of drugs in vitro, allowing for predictions of in vivo behavior. Several in vitro drug distribution assays are available at our lab, including:
|
Protein Binding Assays
|
Red Blood Cell (RBC) Partitioning Assay
|
Protein binding can affect the distribution of a drug in the body. At Creative Biolabs, we typically use blood, microsomes, tissue homogenates, and animal or human cells to study the distribution of a drug in specific cell types or tissues. To determine the drug distribution, we offer the following protein binding assays:
-
Plasma Protein Binding Assay
-
Whole Blood Binding Assay
-
Brain Tissue Binding Assay
-
Hepatocyte Binding Assay
-
Microsomal Binding Assay
|
Red blood cells can serve as a reservoir for certain drugs, affecting their overall distribution and pharmacokinetics in the body. The RBC partitioning assay provides valuable insights into the distribution of drugs within the blood compartment, specifically the ratio of whole blood to plasma. It can help in understanding the extent to which a drug binds to red blood cells, which may affect its distribution to different organs and tissues.
|
How Creative Biolabs' Drug Distribution Analysis Service Can Assist Your Project
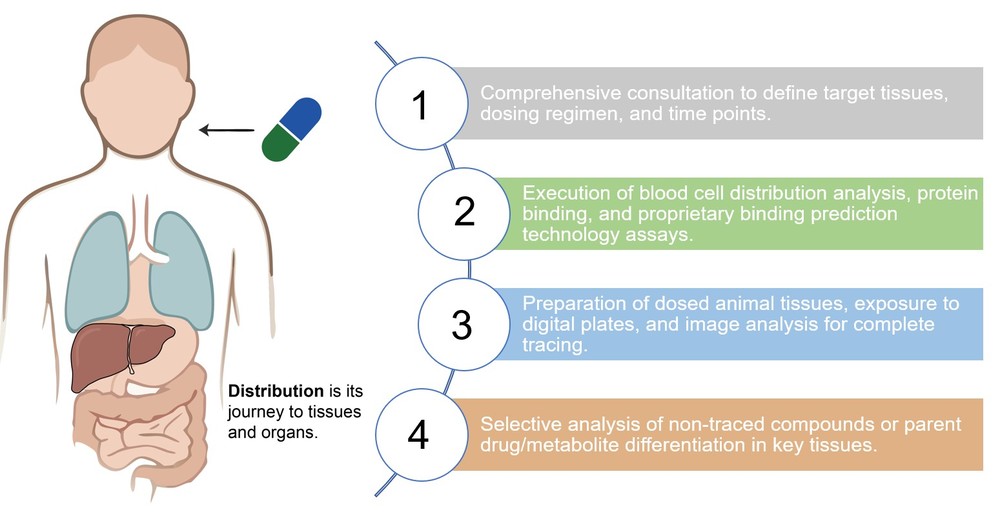
Highlights
One-Stop Distribution Mapping
We offer an integrated, full-spectrum service from foundational in vitro binding profiles to in vivo tissue mapping, ensuring a seamless, high-quality data chain for building robust preclinical data packages.
Unmatched Resolution for Highly Bound Compounds
We utilize our proprietary binding prediction technology to reliably differentiate between molecules that exhibit extremely high plasma binding, providing the necessary resolution where conventional methods often fail.

Custom Study Design & Flexibility
Every project is managed by dedicated ADME experts who collaborate with you to define highly customized PK/PD study designs, perfectly tailored to your molecule's unique profile and specific research objectives.
Targeted Tissue Analysis & Specificity
We maintain the capability for high-magnification analysis, including cellular-level visualization, to deliver precise, sub-organ distribution data for anatomically restricted or specialized areas, such as ocular tissue or specific CNS regions.
Experience the Creative Biolabs advantage - Secure a formal quotation today.
Customer Reviews
-
Critical Data
Using Creative Biolabs' drug distribution analysis service in our research has significantly improved/facilitated accurate CNS exposure measurement for our small molecule library, allowing us to confidently screen out compounds with insufficient delivery across the CNS barrier. - M***s R.
-
High-Level Insight
Using Creative Biolabs' drug distribution analysis service in our research has significantly improved/facilitated justifying a lower, safer PD-driven human dose by confirming sufficient target tissue concentration, preventing the toxicity associated with high Vd storage. - A***a L.
FAQs
Q: What species do your blood/tissue samples come from?
A: At Creative Biolabs, our blood/tissues are from several species, including human, rodent, and non-rodent species such as human, mouse, rat, rabbit, and monkey.
Q: What method do you use to analyze Plasma Protein Binding?
A: Plasma protein binding of compounds is frequently evaluated using a range of techniques, such as equilibrium dialysis, ultrafiltration, ultracentrifugation, and flux dialysis. Equilibrium dialysis is the most common method among them. The choice of method depends on the specific drug properties and the needs of your projects.
Related Services
In Vitro ADME Screening Service
Assess metabolic stability, drug-metabolizing enzyme interaction, and identify major metabolites to complement the distribution data.
Learn More →
Drug Metabolic Stability Analysis Service
Detailed LC-MS and NMR-based services for the structural elucidation and quantification of circulating and tissue-bound metabolites, which are critical for safety assessment and understanding drug pathways.
Learn More →
How to Contact Us
To cease reliance on empirical assumptions and commence the rigorous quantification of a compound's actual tissue distribution, the ADME specialists at Creative Biolabs stand prepared to offer comprehensive guidance and bespoke study design, ensuring the fulfillment of all necessary regulatory and developmental requirements. If you require drug distribution analysis, please contact us now.
Reference
-
Wolska, Eliza, and Małgorzata Sznitowska. "Modeling the Analysis Process of a Lipid-Based, Multi-Compartment Drug Delivery System." Processes 13.2 (2025): 460. Distributed under an Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/pr13020460
For Research Use Only | Not For Clinical Use


 Fig.1 Models of probable drug substance distribution patterns in SLMs. 1
Fig.1 Models of probable drug substance distribution patterns in SLMs. 1


