Enzyme Inhibition Assessment Service
At Creative Biolabs, we provide end-to-end digital excellence, specializing in the strategic planning and tactical execution of modern business solutions. We offer a holistic package that covers consultation, custom development, and post-launch optimization. Clients receive concrete deliverables, including fully documented strategic roadmaps, scalable high-performance applications. Our primary advantages are our rapid deployment cycle, driven by an expert, agile team, and our unwavering focus on delivering solutions that guarantee a clear, measurable return on investment, positioning us as the most efficient partner for your growth goals.
Introduction What We Can Offer Why Creative Biolabs Customer Reviews FAQs Related Services Contact Us
Enzyme Inhibition in Drug-Drug Interactions
Understanding drug-drug interactions is crucial for ensuring safe and effective drug therapy. These interactions can either potentiate or attenuate the therapeutic effect of drugs, leading to either improved or worsened patient outcomes. One of the mechanisms through which drug-drug interactions occur is through enzyme inhibition. Enzymes play a pivotal role in drug metabolism. Co-administration of two drugs may cause one drug or both to inhibit the activity of an enzyme involved in the metabolism of the other drug, thus leading to significant changes in drug concentrations and pharmacological effects.
Discover how we can help - Request a consultation.
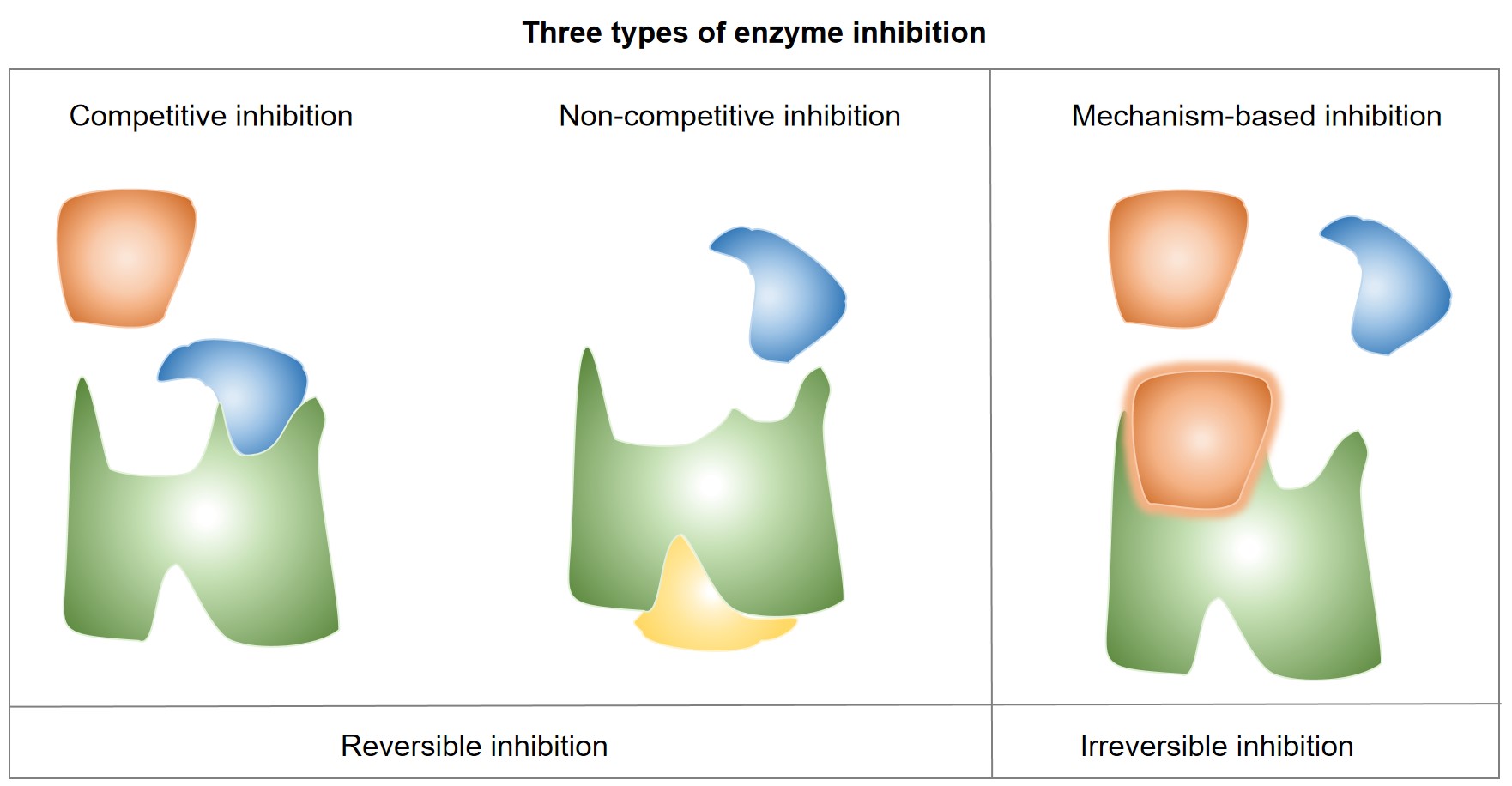 Fig.1 A workflow of predicting DDI by fusing biochemical and structural information.
Fig.1 A workflow of predicting DDI by fusing biochemical and structural information.
What We Can Offer
At Creative Biolabs, we provide a wide range of enzyme inhibition assays to assess drug-drug interactions. Our team of experts has extensive experience in performing cytochrome P450 (CYP) inhibition assays, UGT inhibition assays, and other enzyme inhibition assays. These assays can provide valuable insights into the pharmacokinetic properties of drugs and their potential for interactions with other drugs.
CYP Inhibition Assay
CYP enzymes play a vital role in drug metabolism as they are responsible for the oxidation, reduction, and hydrolysis of numerous compounds. We perform CYP inhibition assays to evaluate the potential of the drugs to inhibit the activity of these enzymes by using several systems, such as hepatocytes, liver microsomes, or recombinant CYP enzymes. The formation of metabolites is assessed using LC/MS/MS analysis.
-
Using liver microsomes, recombinant CYP enzymes, or hepatocytes
-
Various P450 isozymes (e.g., CYP1A, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP3A)
-
Analysis with LC/MS/MS
-
Evaluation of the inhibition constants
-
Evaluation of the half-maximal inhibitory concentration
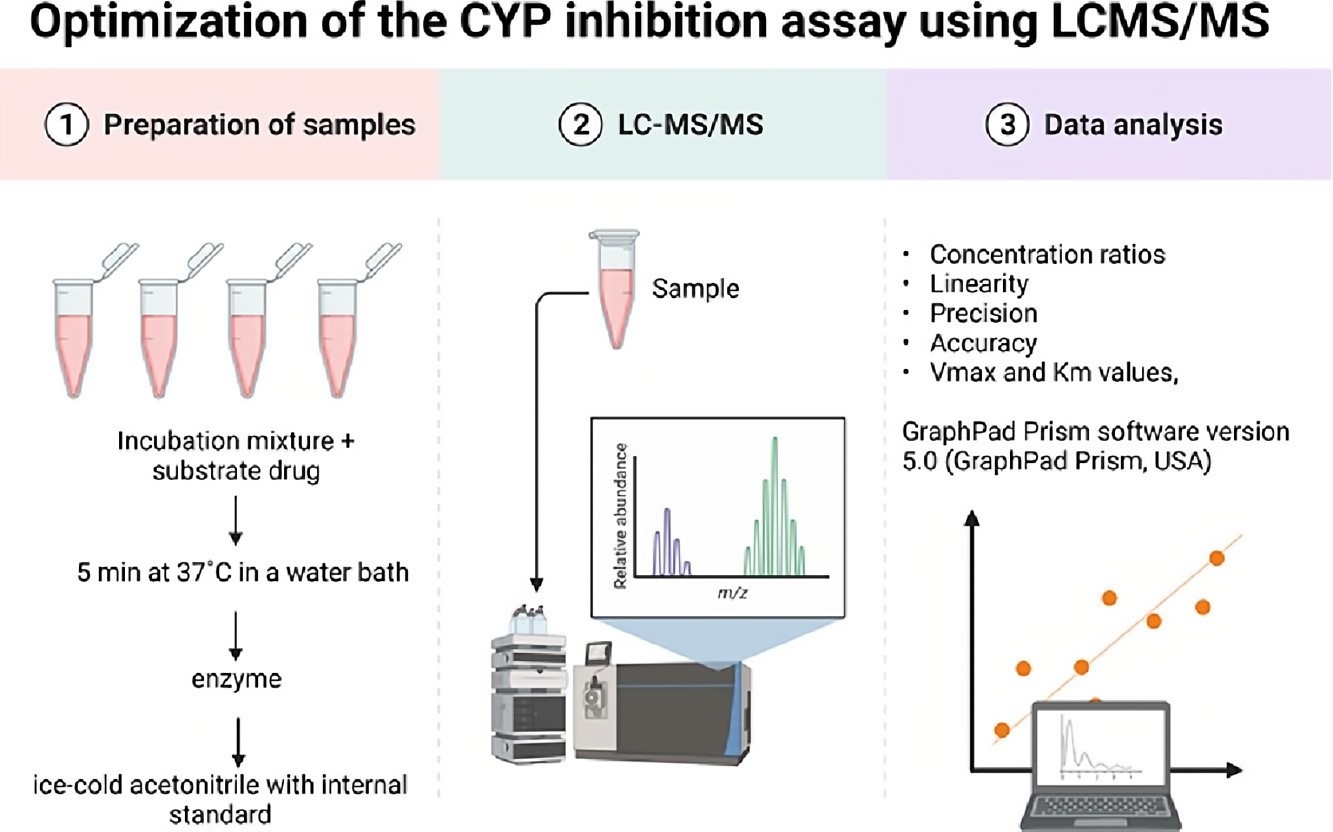 Fig.2 The workflow of the CYP inhibition assay. 1
Fig.2 The workflow of the CYP inhibition assay. 1
UGT Inhibition Assay
Uridine diphosphate glucuronosyltransferases (UGTs) are a family of enzymes that catalyze the conjugation of various compounds, including drugs and endogenous substances, with glucuronic acid. UGT Inhibition assays typically involve incubating the test compound with UGT enzymes and a suitable substrate and then measuring the formation of the glucuronide conjugate to determine the enzyme activity. Our CYP inhibition assays are designed to assess the potential for drugs to inhibit CYP enzymes, which can lead to altered drug metabolism and potentially significant pharmacokinetic interactions.
-
Using recombinant UGT enzyme (e.g., UGT1A1, UGT1A3, UGT1A4, UGT1A9, UGT2B7, and UGT2B15)
-
Analysis with LC-MS/MS
-
Evaluation of the IC50
Other Enzyme Inhibition Assay
In addition to CYP and UGT enzymes, there are many other enzymes involved in drug metabolism and other biological processes that may be targeted for inhibition, such as N-acetyltransferases, sulfotransferases, and carboxylesterases. At Creative Biolabs, we also offer inhibition studies towards these enzymes as part of our drug-drug interaction services.
Highlights
Unmatched Analytical Capacity
High-throughput capabilities utilizing multi-channel LC/MS/MS platforms, ensuring we can process large compound libraries without compromising data quality or turnaround time.
Quality-by-Design (QbD) and PAT
Our studies operate under strict QbD principles with process analytical technology (PAT) applied to enzyme integrity and kinetic parameter measurement, ensuring auditable, regulatory-compliant results for every data point.
Customization in Assay Mode
Flexibility to run inhibition kinetics in batch, fed-batch (time-dependent), or continuous TDI mode to accurately simulate in vivo conditions for both reversible and irreversible inhibitors, depending on the compound's characteristics.
Enzyme Source Stability and Verification
Stringent quality assurance procedures guarantee the stability and activity of all recombinant enzymes and human liver fractions (HLM, hepatocytes) used in the DDI assessment, analogous to rigorous cell bank verification.
Discover how a partnership with us can streamline your research - Get a quote today.
Customer Reviews
-
Exceptional MBI Characterization
Creative Biolabs' service gave us definitive kinact and KI data, differentiating true covalent MBI from tight-binding. This precise mechanistic data justified essential SAR changes, satisfying regulatory requirements. - J. Ha**k.
-
Superior UGT and Non-CYP Profiling
Creative Biolabs' LC/MS/MS provided highly consistent and reliable UGT1A4 and sulfotransferase data. This significantly improved our holistic assessment of non-CYP metabolism risk compared to our prior in-house assays. - K. M*tlin.
FAQs
Q: Can you perform inhibition studies against non-CYP or non-UGT therapeutic targets?
A: Absolutely. Beyond the standard metabolic panel, our service includes bespoke assay development for novel therapeutic enzymes, such as specific kinases or ATPases. We work with your team to determine the substrate Km and Vmax, and then fully characterize the MoA to inform your target engagement strategy.
Q: How does Creative Biolabs ensure the data is accurate, especially for challenging MBI compounds?
A: We use LC/MS/MS, the most sensitive and selective analytical method, to directly measure metabolite depletion, which is superior to less accurate fluorescent endpoints. For MBI studies, we use validated kinetic models and follow industry best practices to accurately calculate the irreversible constants, providing confidence in your DDI risk profile.
Related Services
Enzyme Induction Assessment Service
We provide a comprehensive CYP and UGT induction assessment using human hepatocytes, featuring a dual readout of enzyme activity (LC/MS/MS) and mRNA expression (qRT-PCR) for high-confidence regulatory filings.
Learn More →
Metabolite Profiling Service
Core ADME assay to determine a compound's stability in various matrices (e.g., human liver microsomes, hepatocytes, plasma). We calculate crucial parameters to predict in vivo exposure and guide lead optimization.
Learn More →
How to Contact Us
The Creative Biolabs enzyme inhibition assessment service is your critical ally in pharmaceutical development. We transform uncharacterized leads into de-risked candidates by delivering precise data across the entire ADME landscape. If you have a need for enzyme inhibition assessment, please contact us now.
Reference
-
Abduraman, Muhammad Asyraf et al. "Optimization of the CYP inhibition assay using LC-MS/MS." MethodsX vol. 9 101827. 23 Aug. 2022. Distributed under an Open Access license CC BY 4.0, without modification. https://doi.org/10.1016/j.mex.2022.101827


 Fig.1 A workflow of predicting DDI by fusing biochemical and structural information.
Fig.1 A workflow of predicting DDI by fusing biochemical and structural information.
 Fig.2 The workflow of the CYP inhibition assay. 1
Fig.2 The workflow of the CYP inhibition assay. 1

