Enzyme Induction Assessment Service
Enzyme induction assays are important in the prediction of drug-drug interactions that may occur in vivo, which can significantly affect the safety and efficacy of drugs. At Creative Biolabs, we utilize physiologically relevant systems like primary human hepatocytes to predict whether a drug may affect the metabolism of other drugs metabolized by key enzymes, leading to potential drug-drug interactions. By measuring the activity of these enzymes, such as CYP450 and UGT enzymes involved in phase I and phase II drug metabolism, we can assess whether the test drugs have the potential to induce changes in enzyme activity.
Introduction What We Can Offer Why Creative Biolabs Customer Reviews FAQs Related Services Contact Us
Enzyme Induction Assays
Enzyme induction assays are essential for predicting and understanding the metabolism-dependent drug-drug interactions that can occur between drugs. To assess enzyme induction assays, hepatocytes are incubated with test drugs suspected of inducing the upregulation of enzymes. By measuring the activity of these enzymes, we assess whether the test compound has the potential to induce changes in enzyme activity. Furthermore, gene expression levels of specific isozymes can be determined using molecular techniques such as quantitative real-time polymerase chain reaction (qRT-PCR).
Discover how we can help - Request a consultation.
What We Can Offer
CYP Induction Assay
The CYP induction assay is a commonly used assay to evaluate the potential of drugs to induce CYP enzymes. In the CYP induction assay, hepatocytes are incubated with the test drugs. By comparing the CYP enzyme activity in hepatocytes incubated with the test drug to that in control hepatocytes, the potential of the test drug to induce CYP enzyme upregulation can be assessed. Additionally, gene expression levels of specific CYP isozymes can be determined using qRT-PCR, providing further insight into the mechanism of enzyme induction.
|
Test System
|
Fresh human hepatocytes or cryopreserved human hepatocytes
|
|
CYP Enzymes
|
CYP1A, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP3A4, etc.
|
|
Concentrations
|
Multiple concentrations of test articles are available. We recommend 6 concentrations.
|
|
Number of Replicates
|
3
|
|
Analysis Method
|
-
Analysis of enzyme catalytic activity of CYP isoforms via LC-MS/MS
-
Analysis of relative mRNA levels by qRT-PCR
|
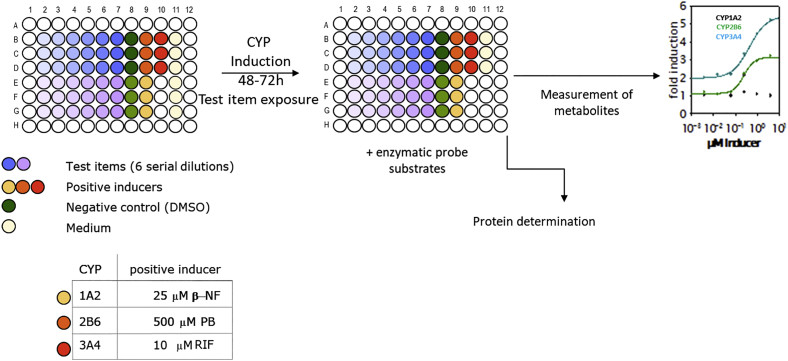 Fig.1 A CYP induction experimental design. 1
Fig.1 A CYP induction experimental design. 1
UGT Induction Assay
In addition to CYP enzymes, UGT enzymes also play a crucial role in drug metabolism. We offer the UGT induction assay as well.
|
Test System
|
Fresh human hepatocytes or cryopreserved human hepatocytes
|
|
UGT enzymes
|
UGT1A1, UGT1A4, UGT1A6, UGT1A9, UGT2B, etc.
|
|
Concentrations
|
Multiple concentrations of test articles are available. We recommend 6 concentrations.
|
|
Number of Replicates
|
3
|
|
Analysis Method
|
-
Analysis of enzyme catalytic activity of UGT isoforms via LC-MS/MS
-
Analysis of relative mRNA levels by qRT-PCR
|
Highlights
Flexible In Vitro Modeling
Studies can be run using batch, fed-batch, or continuous dosing modes to accurately simulate complex clinical exposure profiles, thus optimizing culture conditions to maximize the predictive power and reliability of the induction data.
Quality-by-Design (QbD) Implementation
The entire assessment utilizes a well-established quality system, incorporating QbD principles and process analytical techniques (PAT) to guarantee the procedural quality and integrity of every induction assay run.
High-Standard Quantitative QC
Utilization of advanced quality control tools, notably highly precise LC-MS/MS instrumentation and qRT-PCR, to meticulously quantify and evaluate the induction response and ensure data rigor.
Customized Service Offerings
Creative Biolabs specializes in providing fully tailored induction studies to address unique client challenges, including non-standard test matrices, investigation of novel CYP/UGT targets, or specialized kinetic sampling regimens.
Discover how a partnership with us can streamline your research - Get a quote today.
Customer Reviews
-
High Predictivity
Using Creative Biolabs' service in our research has significantly improved our confidence in our candidate's regulatory acceptance package. Their qRT-PCR data were highly concordant with the final activity changes, giving us a strong mechanistic story for our lead compound, which exhibited complex PXR binding. - L. Ma***y.
-
Operational Efficiency
The final report was incredibly useful - a clear, quantitative summary that streamlined our internal review process. The gold-standard LC-MS/MS with deuterated standards provided a level of analytical rigor that directly addressed international regulatory guidance concerns regarding precision and reproducibility. - P. Aln***r.
FAQs
Q: Why is a dual-endpoint assessment (mRNA and Activity) necessary, as opposed to measuring only enzyme activity?
A: Transcriptional induction is a delayed process. Measuring mRNA via qRT-PCR offers the earliest proof of gene activation. Functional activity via LC-MS/MS confirms this change translates to increased metabolism. This dual approach provides the full, robust mechanistic story required for definitive assessment.
Q: Is the primary human hepatocyte model truly the most accurate model for predicting human risk?
A: Yes, primary human hepatocytes (PHH) are the industry's gold standard. They retain native expression of key nuclear receptors (PXR, CAR) and critical metabolizing enzymes, ensuring the highest level of in vitro-to-in vivo predictability for drug risk modeling.
Related Services
Reaction Phenotyping Assessment
Determine which CYP enzymes metabolize your compound and identify key human metabolites, crucial for understanding potential DDI liabilities and subsequent UGT induction risk.
Learn More →
Inhibition Assays
The counterpart to induction - we provide definitive in vitro assays for time-dependent (TDI) and reversible inhibition using LC-MS/MS.
Learn More →
How to Contact Us
Creative Biolabs combines regulatory expertise, the gold-standard primary human hepatocyte model, and the analytical rigor of dual-endpoint LC-MS/MS to deliver clear, actionable R3 risk assessments. We translate complex biochemistry into definitive safety data, ensuring your program advances without unforeseen DDI liabilities that compromise patient safety and development timelines. If you have a need for enzyme induction assessment, please contact us now.
Reference
-
Bernasconi, Camilla et al. "Validation of in vitro methods for human cytochrome P450 enzyme induction: Outcome of a multi-laboratory study." Toxicology in vitro: an international journal published in association with BIBRA vol. 60 (2019): 212-228. Distributed under an Open Access license CC BY 4.0, without modification. https://doi.org/10.1016/j.tiv.2019.05.019


 Fig.1 A CYP induction experimental design. 1
Fig.1 A CYP induction experimental design. 1

