Mediator Release Assay Service
Understanding TNF-α-induced PGE2 Release
Tumor necrosis factor-alpha (TNF-α) is a pro-inflammatory cytokine that plays a pivotal role in the body's immune response, inflammation, and cellular apoptosis. One significant pathway influenced by TNF-α is the release of prostaglandin E2 (PGE2), a lipid compound that functions as an inflammatory mediator. The TNF-α-induced PGE2 release is critical in various physiological and pathological processes, including pain sensation, fever, and the modulation of immune responses.
In biomedical research and drug development, modulating the TNF-α pathway through agonists or antagonists presents a promising therapeutic strategy. TNF-α agonists can mimic the cytokine's action and enhance immune responses, beneficial in certain infectious or malignant conditions. Conversely, TNF-α antagonists can attenuate/inhibit excessive inflammatory responses, offering potential relief in chronic inflammatory states such as rheumatoid arthritis, Crohn's disease, and psoriasis. Given the critical interplay between TNF-α and PGE2 in inflammatory mechanisms, efficiently monitoring the TNF-α-induced PGE2 release can substantially advance the screening and characterization of potential therapeutics aimed at modulating inflammatory responses.
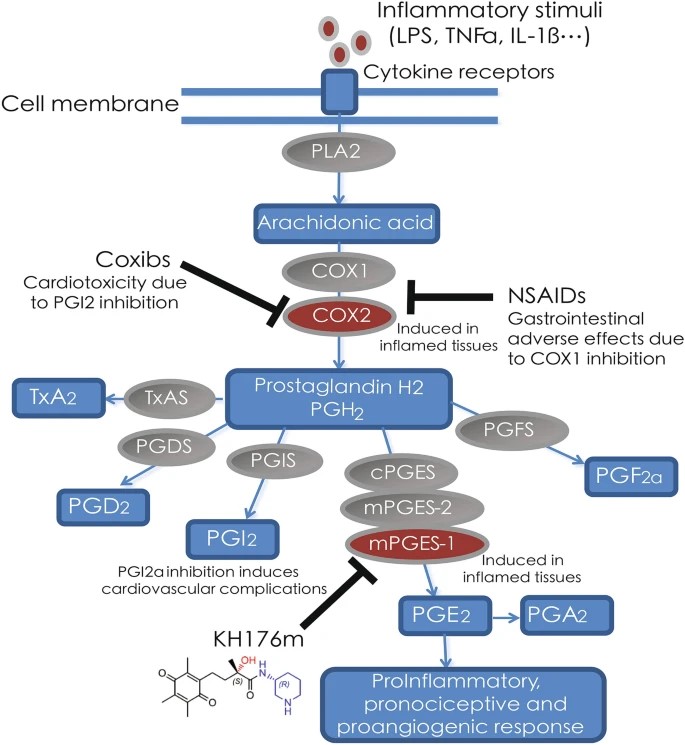 Fig.1 Inflammatory stimuli (e.g. TNF-α, LPS, IL-1β)-induced PGE2 release.1
Fig.1 Inflammatory stimuli (e.g. TNF-α, LPS, IL-1β)-induced PGE2 release.1
Our Specialized Mediator Release Assay Service
Creative Biolabs is proud to offer our specialized mediator release assay service designed to rigorously assess the activity of potential TNF-α agonists or antagonists with high precision and reliability. Our TNF-α-induced PGE2 assay is meticulously optimized to deliver high-quality, reliable data to support drug discovery and development projects. By leveraging our expertise, you can accurately determine the efficacy and potency of your target compounds, opening avenues for therapeutic discoveries and advancements.
Our TNF-α-induced PGE2 assay service is designed to provide comprehensive analysis, enabling our clients to measure the efficacy of test compounds in modulating TNF-α activity. We utilize human epitheloid cervix carcinoma HeLa S3 cells, a trusted cellular model, for our TNF-α-induced PGE2 assay. After incubation of a test compound or control vehicle with the HeLa S3 cells, we assess the release of PGE2 upon stimulation or inhibition using Time-Resolved Fluorescence Resonance Energy Transfer (TR-FRET), offering high sensitivity and accuracy.
Protocol
|
Assay Type:
|
Cell-based PGE2 release
|
|
Detection:
|
PGE2
|
|
Cell Type:
|
HeLa S3
|
|
Functional Mode:
|
Agonist / Antagonist
|
|
Detection Method:
|
TR-FRET
|
|
Measured Response:
|
Fluorescence
|
|
Control Inhibitor:
|
Indomethacin
|
|
Control Activator:
|
TNF-α
|
Features of Our Service
Our assay is versatile, and suitable for various stages of drug development, from initial screening to detailed mechanistic studies. Whether you're evaluating a compound's potential anti-inflammatory properties or investigating its role in other physiological or pathological contexts, this assay offers vital insights.
-
Reliable Controls for Optimized Assay Performance
Our robust experimental setup includes using Indomethacin as the control inhibitor and TNF-α as the control activator, ensuring the reliability and reproducibility of your results.
-
Superior Sensitivity and Specificity with TR-FRET Technology
Creative Biolabs integrates TR-FRET into our assay platforms to ensure that you receive precise and reproducible measurements of PGE2 release. By employing TR-FRET, we overcome many of the limitations and challenges associated with traditional detection methods, such as ELISA, enhancing the assay's robustness and consistency.
For more details about our release assay services please contact us.
Reference
-
Jiang, X., et al. "Mechanism of action and potential applications of selective inhibition of microsomal prostaglandin E synthase-1-mediated PGE2 biosynthesis by sonlicromanol's metabolite KH176m." Scientific Reports 11.1 (2021): 880. Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only | Not For Clinical Use


 Fig.1 Inflammatory stimuli (e.g. TNF-α, LPS, IL-1β)-induced PGE2 release.1
Fig.1 Inflammatory stimuli (e.g. TNF-α, LPS, IL-1β)-induced PGE2 release.1
 Download our brochure
Download our brochure