Phosphatase Assay Service
Background Service Workflow Highlights FAQs Contact Us
Creative Biolabs' specialized Phosphatase Assay Service is engineered to measure enzyme activity, selectivity, and inhibitor potency with high sensitivity. Our platform supports immune-focused targets implicated in checkpoint biology and inflammatory control, aiding discovery efforts that aim to reshape tumor–immune interactions.
Background
Protein phosphatases, including serine/threonine (e.g. PP2A, PP1) and tyrosine phosphatases (e.g. SHP‑1/2, PTPN22), control deactivation of kinases in signaling cascades central to T cell activation, macrophage polarization, and cytokine-mediated communication. In tumor immunology, dysregulated phosphatase activity may suppress checkpoint signaling or alter tumor antigen presentation. Understanding phosphatase kinetics and regulation is therefore essential to uncover immune modulators and restore antitumor immunity.
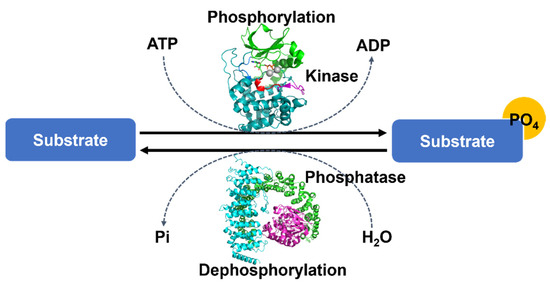 Fig.1 Protein phosphorylation overarching mechanism.1
Fig.1 Protein phosphorylation overarching mechanism.1
Featured Services at Creative Biolabs
Fluorogenic peptide assays
Using substrates such as DiFMUP/O-methylfluorescein phosphate to monitor PP2A, PP1 activity.
Colorimetric pNPP assays
Robust high-throughput screening of tyrosine phosphatases with simple absorbance detection.
Peptide libraries based on immune regulatory motifs
Derived from PD‑1, CTLA‑4, STAT1, JAK1 activation loops for selectivity profiling.
Isoform panel screening
Evaluate SHP‑1, SHP‑2, PTPN22, CDC25A among immune-relevant phosphatases.
Kinetic and inhibition assays
Real-time measurement of Km, Vmax, IC₅₀, and Ki using optimized buffers and enzyme preparations.
Assay formats include biochemical and cell-extract-based setups and are fully adaptable for HTS or focused SAR studies.
Workflow

Highlights
-
Immune-Specific Substrate Panels – Peptides designed from immune checkpoint signaling domains increase biological relevance.
-
Dual Detection Formats – Supports both fluorescence and colorimetric readouts for flexibility and throughput balance.
-
Isoform–Selective Profiling – Simultaneous assessment of multiple phosphatases across immune pathways.
-
Quantitative Kinetic Analysis – High-resolution measurement of enzymatic parameters for SAR and biocharacterization.
-
Robust Assay Performance – High Z' (>0.75) and signal-to-background ratios across assay formats.
FAQs
Q1: Can you tailor the assay to dephosphorylate specific immune-related proteins?
A1: Yes, we offer custom peptide substrates representing activation loops or docking sites from proteins such as PD‑1, STATs, or JAK kinases.
Q2: Is both biochemical and cell-lysate format supported?
A2: Absolutely. We can work with purified enzymes, immune‑cell extracts, or co‑culture systems to evaluate real-world dephosphorylation.
Q3: Do you support kinetic modeling for Km and Ki?
A3: Yes. We provide comprehensive kinetics including initial velocity plots, IC₅₀, Ki, and enzyme-substrate dynamics.
Q4: What throughput is available for screening?
A4: Our workflows support 96‑ and 384‑well formats with consistent Z' >0.7, ideal for compound library profiling or SAR studies.
Engage with Phosphatase Assay Experts
Creative Biolabs' enzymology and immune signaling teams are available to design tailored phosphatase assay workflows aligned with your research goals. Whether you aim to investigate checkpoint phosphatases or immune-associated kinases, we provide technical consultation, assay customization, and data delivery with exceptional clarity.
Reference
-
Seok, Seung-Hyeon. "Structural Insights into Protein Regulation by Phosphorylation and Substrate Recognition of Protein Kinases/Phosphatases." Life (Basel, Switzerland) vol. 11,9 957. DOI: 10.3390/life11090957. Distributed under Open Access license CC BY 4.0, without modification.
For Research Use Only | Not For Clinical Use


 Fig.1 Protein phosphorylation overarching mechanism.1
Fig.1 Protein phosphorylation overarching mechanism.1

