Protein Binding Analysis Service
At Creative Biolabs, we have extensive experience in protein binding studies and can offer advice and support throughout the entire process, from sample preparation to data analysis and interpretation. We offer several protein-binding assays. Our team of experts will carefully evaluate your specific needs to recommend the most suitable approach. Our protein binding assay services are designed to provide accurate and reproducible results, ensuring that our clients have the necessary data to make informed decisions about their drug candidates.
Introduction What We Can Offer How We Can Help Why Creative Biolabs Customer Reviews FAQs Related Services Contact Us
Protein Binding Analysis for Screening Drug Candidates
The in vitro protein binding assay is a key tool in the early stages of drug discovery and development to characterize the drug distribution. The results of the protein binding assay provide critical information about the drug's affinity for plasma, serum, whole blood, microsomes, and tissue homogenates. For example, a high affinity for plasma proteins indicates that a significant portion of the drug will be bound and, therefore, less available to exert its therapeutic effects; Conversely, a low affinity for plasma proteins suggests that a larger proportion of the drug will remain unbound, potentially leading to higher concentrations at the therapeutic site and more pronounced therapeutic effects. Therefore, understanding the extent of protein binding can provide valuable insights into a drug's pharmacokinetic profile.
Discover how we can help - Initiate consultation for project assessment.
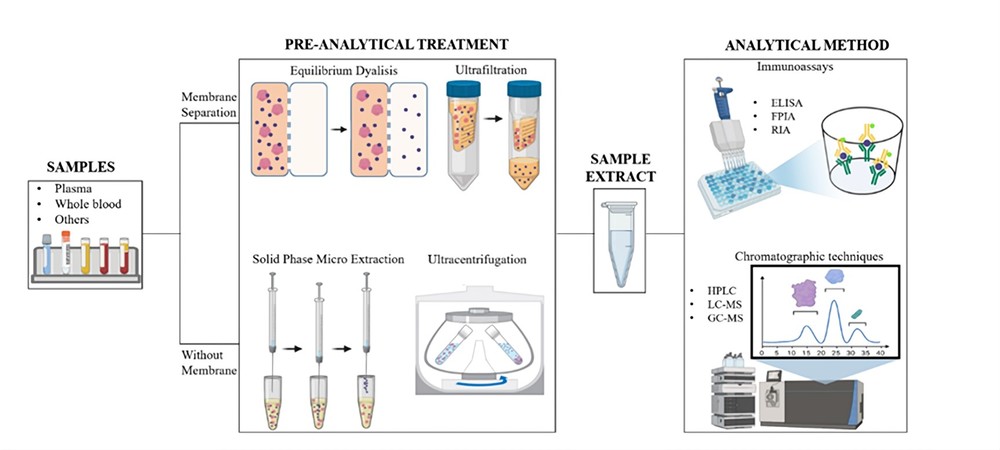 Fig.1 A workflow of protein binding assay. 1
Fig.1 A workflow of protein binding assay. 1
What We Can Offer
During the protein binding assay, the drug is added to tissues such as human plasma, brain homogenate, etc., and the mixture is incubated under controlled conditions. The incubation period allows the drug to interact with the proteins present in the sample. Subsequently, the unbound drug is separated from the protein-bound drug using suitable techniques such as centrifugation or ultrafiltration. The concentration of the unbound drug is then determined using analytical methods such as HPLC or LC-MS.
|
Available Assays
|
Techniques
|
-
Plasma Protein Binding Assay
-
Whole Blood Binding Assay
-
Brain Tissue Binding Assay
-
Hepatocyte Binding Assay
-
Microsomal Binding Assay
|
-
Equilibrium dialysis
-
Ultrafiltration
-
Ultracentrifugation
-
Flux dialysis
|
Our Protein Binding Assays
Highlights
Customized Assay Design
Every project starts with a deep consultation to custom-tailor the assay methodology (ED, UF, SPR, ITC, etc.) based precisely on your molecule's chemical properties, target complexity, and specific regulatory phase requirements.
Integrated Multi-Modal Platform
We offer a seamless, one-stop solution integrating advanced biophysical tools (SPR/ITC for kinetics) with quantitative ADME/PK separation techniques and high-sensitivity LC-MS/MS analysis, ensuring robust and consistent data across all pillars.
Kinetic & Mechanistic Insight
Move beyond static affinity by accessing real-time kinetic data, enabling the calculation of drug residence time - the critical predictor of long-lasting in vivo efficacy.
Complex Target Expertise
Specialized protocols for successfully handling challenging compounds, including highly lipophilic drugs, unstable molecules, and complex membrane protein targets, eliminating non-specific binding artifacts.
For more information regarding service integration and pricing, please request a formal quotation.
Customer Reviews
-
Optimized Drug Residence Time
Using Creative Biolabs' service in our research has significantly improved our understanding of true in vivo efficacy. The precise Kd measurements via SPR allowed us to confirm the required long residence time for our oncology target, which was key to moving the candidate forward. - J**e Williams.
-
Complex Interaction Mapping
Using Creative Biolabs' service in our research has significantly improved our cellular target validation. The combination of Co-IP and SILAC-MS helped us confidently map the native protein complex that interacts with our small molecule, ruling out several false-positive interactors identified in our initial screening. - M**a Lopez.
FAQs
Q: Can Creative Biolabs handle challenging targets like membrane proteins or highly lipophilic compounds?
A: Absolutely. Our platform is specifically equipped for challenging targets. For membrane proteins, we utilize detergent-solubilized systems coupled with SPR or employ solution-based methods. For highly lipophilic compounds, we ensure accuracy by assessing non-specific binding to the apparatus and providing specialized blood partitioning assays.
Q: What is the most common assay for protein binding analysis?
A: Equilibrium dialysis is considered the gold standard for investigating protein binding over other methods, such as filtration. Equilibrium dialysis is a relatively simple technique that can be performed with a wide range of samples, including plasma, serum, and other biological fluids. It offers a high degree of precision and accuracy in determining the binding affinity of drugs or other ligands to plasma proteins. While other methods, such as filtration, may be less labor-intensive, they often lack the sensitivity and specificity required for accurate protein binding analysis.
Related Services
Small-Molecule Drug Bioanalysis Service
Dedicated quantitative analysis of your small molecule drug in complex biological matrices (plasma, urine, tissue) using our high-resolution LC-MS/MS platforms.
Learn More →
Antibody and Protein Production
Full-service development and production of high-purity, active recombinant proteins and therapeutic antibodies, utilizing mammalian, insect, or microbial expression systems to ensure high-quality material for binding analysis.
Learn More →
How to Contact Us
To facilitate the acceleration of your drug discovery program utilizing verifiable, high-quality binding data, please initiate contact with our specialized scientific team for the acquisition of detailed documentation and a comprehensive project consultation. The development of a collaborative partnership is highly valued by Creative Biolabs. If you have a need for protein-binding analysis, please contact us now.
Reference
-
Charlier, Bruno et al. "The Effect of Plasma Protein Binding on the Therapeutic Monitoring of Antiseizure Medications." Pharmaceutics vol. 13,8 1208. 5 Aug. 2021. Distributed under an Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/pharmaceutics13081208


 Fig.1 A workflow of protein binding assay. 1
Fig.1 A workflow of protein binding assay. 1


