Reaction Phenotyping Assessment Service
Creative Biolabs provides the definitive evidence required by global regulatory agencies to establish the metabolic fate of your drug candidate. We move beyond simple screening to deliver a quantitative metabolic fingerprint, detailing the fractional contribution of every relevant enzyme. This data allows you to proactively design DDI mitigation strategies, predict potential patient variability due to genetic polymorphism.
Introduction What We Can Offer Protocol Why Creative Biolabs Customer Reviews FAQs Related Services Contact Us
Reaction Phenotyping Assessment in Drug-Drug Interactions
Reaction phenotyping assays are valuable tools in drug development and pharmacology research. These assays aim to evaluate the role of specific enzymes or transporters in the metabolism of a drug. These assays are used to identify and quantify the enzymes responsible for drug metabolism, which can help predict how a drug will be metabolized in the body and its potential interactions with other drugs. The information obtained from reaction phenotyping assays can be crucial in drug development and personalized medicine, as it can contribute to optimizing drug dosages, improving drug efficacy, and minimizing the risk of adverse drug reactions.
Discover how we can help - Request a consultation.
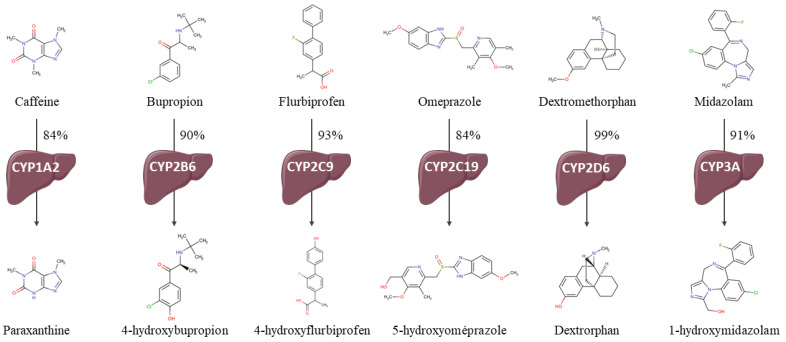 Fig.1 CYP reaction phenotyping via cocktail assay. 1
Fig.1 CYP reaction phenotyping via cocktail assay. 1
What We Can Offer
At Creative Biolabs, we offer reaction phenotyping assessment services to identify the specific enzymes involved in the metabolism of a drug, predict potential drug-drug interactions, and assess the overall metabolic clearance of the drug in humans. Our reaction phenotyping assays involve incubating the drug of interest with recombinant enzymes and/or human liver microsomes. By monitoring the formation of metabolites, we are able to determine which enzymes are responsible for the metabolism of the drug.
Test System
Recombinant enzyme system: Recombinant enzymes are expressed in cell systems and used to study the metabolism of drugs. By incubating the specific enzyme with the drug, researchers can determine the enzyme's specific role in drug metabolism.
Human liver microsome system: Liver microsomes contain the enzymes responsible for drug metabolism. By incubating the microsomes with the drug in the presence of specific enzyme inhibitors and analyzing the metabolites formed, researchers can determine which enzymes are responsible for metabolism.
Enzymes
Creative Biolabs offers CYP and UGT reaction phenotyping assays tailored to the client's specific requirements.
|
CYP isoforms
|
UGT isoforms
|
|
CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP3A
|
UGT1A1, UGT1A3, UGT1A4, UGT1A6, UGT1A9, UGT2B7, and UGT2B15
|
Parent depletion and metabolite formation assays are two common approaches for reaction phenotyping.
Our Protocol
|
Feature
|
ADCC
|
|
Test Article Concentration
|
5 μM (custom concentrations available)
|
|
Test System
|
Recombinant enzymes or human liver microsomes
|
|
Enzymes
|
CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP3A, UGT1A1, UGT1A4, UGT1A6, UGT1A9, UGT2B, etc.
|
|
Time Points
|
0, 5, 15, 30, 45 minutes (custom time points available)
|
|
Number of Replicates
|
3
|
|
Analysis Method
|
LC-MS/MS
|
|
Readout
|
Parent depletion or specific metabolite formation
|
Highlights
Customizable Tiered Assay Design
We offer a one-stop solution, ranging from initial HLM screening to advanced, physiologically relevant hepatocyte co-culture models, allowing us to customize the study scope precisely to your compound's clearance profile.
Unrivaled Enzyme Breadth
Our service provides definitive fm quantification for a large-scale industrial-level panel. This includes the major CYPs, key UGTs, and essential non-CYP DMEs, ensuring comprehensive metabolic clarity with no blind spots.

Superior Quality Control and Traceability
Our well-established quality system ensures the documentation and activity of all biological matrices - from pooled HLMs to recombinant enzymes - are assessed and approved by a qualified quality assurance service, guaranteeing data reliability.
Analytical Excellence
High-standard quality control tools, specifically our high-resolution LC-MS/MS platform, are used to quantify products, ensuring the sensitivity and analytical precision needed for challenging, low-turnover compounds.
Initiate project engagement: request a detailed service proposal.
Customer Reviews
-
Definitive UGT Contribution
Using Creative Biolabs' service in our research has significantly improved our ability to distinguish between UGT and CYP metabolism. We definitely quantified UGT1A1's fm for our lead compound, which was critical since HLMs alone were inconclusive. The eCTD-ready report saved us weeks of internal documentation effort. - Marn A*s.
-
Broad Coverage
Creative Biolabs' reaction phenotyping assessment service, covering the full panel of secondary CYPs (CYP2J2), was far more efficient than building internal assays. The data provided a complete metabolic fingerprint, allowing us to confidently rule out victim DDI for CYP pathways. - Jesa L*z.
FAQs
Q1: When should I perform reaction phenotyping relative to other ADME studies?
A1: Reaction phenotyping is typically performed after you have established basic metabolic stability and determined that your compound is cleared by metabolism. Knowing which enzymes are involved is the logical next step before moving into definitive CYP/UGT inhibition and induction studies. Starting early ensures your DDI risk assessment is built on a solid foundation.
Q2: Why do I need Tier 2's advanced hepatocyte model if HLMs are regulatory compliant?
A2: While HLMs are compliant, they lack the full complement of non-CYP enzymes and endogenous cofactors found in primary hepatocytes, and they struggle with low-clearance compounds. Tier 2 is essential if major CYPs are not involved, or if you require an accurate intrinsic clearance value from a highly physiological system, significantly reducing uncertainty for your most critical assets.
Related Services
Metabolite Identification Services
Focus on identifying and confirming potentially toxic, electrophilic metabolites using trapping agents to mitigate safety and off-target toxicity risks.
Learn More →
Enzyme Inhibition Assessment
Rigorous determination of inhibition constants and time-dependent inhibition for all major CYP and UGT enzymes to provide the quantitative parameters necessary for clinically relevant DDI prediction models.
Learn More →
How to Contact Us
Our services are designed to provide comprehensive data on drug metabolism, enabling you to make informed decisions about drug development and dosing. Our reaction phenotyping assays are highly sensitive and specific, ensuring accurate results. Additionally, our team of experienced scientists is dedicated to providing you with prompt and reliable support throughout the entire process. Whether you need assistance with assay design, data interpretation, or any other aspect of reaction phenotyping, we are here to help. Contact us today to learn more about our reaction phenotyping assessment services.
Reference
-
Darnaud, Léa et al. "Phenotyping Indices of CYP450 and P-Glycoprotein in Human Volunteers and in Patients Treated with Painkillers or Psychotropic Drugs." Pharmaceutics vol. 15,3 979. 18 Mar. 2023. Distributed under an Open Access license CC BY 4.0, without modification. https://doi.org/10.3390/pharmaceutics15030979


 Fig.1 CYP reaction phenotyping via cocktail assay. 1
Fig.1 CYP reaction phenotyping via cocktail assay. 1

