All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
CAR-T-based cell has been demonstrated to be a promising therapy in the treatment of many different tumor types, especially in the treatment of relapsed or refractory B-cell malignancies. However, the administration of CAR-T cells is associated with serious adverse events, which can be fatal in some cases. Fatal off-target tumor toxicity and fatal cytokine release syndrome (CRS) have been reported in clinical trials. The combination of endogenous safety switches represents a reasonable strategy to control toxicity after adoptive T cell therapy. An ideal safety switch should be able to deplete transferred T cells with minimal damage to normal tissues. To minimize the toxicity to healthy tissues, foreign epitopes can be introduced into CAR-T cells for selective ADC targeting.
The 806 epitope of cryptic is one such candidate site because it is only exposed due to overexpression of EGFR or truncation of the extracellular domain. Creative Biolabs has developed a novel switch CAR - the fusion receptor by engineering the 806 recessive epitope of human epidermal growth factor receptor (EGFR) into the N-terminus of full-length human folate receptor 1 (FOLR1) (called FR806). The expression of FR806 allows the transduced T cells to be targeted by CH12, which is a monoclonal antibody that recognizes the 806 epitope in healthy tissues but cannot recognize wild-type EGFR. Therefore, FR806 constitutes a specific cell surface marker that eliminates transduced T cells.
FOLR1 is a glycosylphosphatidylinositol (GPI)-linked membrane glycoprotein that mediates the uptake of folate into cells. Its effective endocytosis makes FOLR1 an important target for drug delivery to FOLR1-positive tumor cells. Data have shown that FR806 epitope and FOLR1 generated fusion receptor - FR806, which can mediate the internalization of ADC to eliminate FR806-expressing T cells. The selectivity of the 806 epitope and the internalization ability of FOLR1 make FR806 an efficient safety switch, which can be additionally used as a biomarker for detection and purification of human T cell immunotherapy.
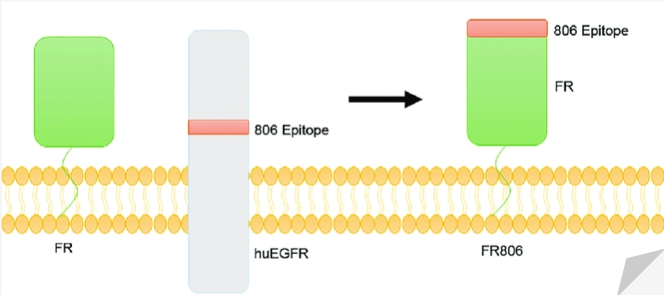

Fig.1 Construction of FR806 and CAR FR806.
As shown in Figure 1, the 806 epitope of EGFR was directly fused to the N-terminus of the full-length human FOLR1 gene. For easy detection, FR806 was co-expressed with EGFP via self-cleavage/ribosome skipping F2A peptide.
With state-of-art CAR development platform and advanced technologies, Creative Biolabs is capable of offering CAR-T-cell development service. For the regular or custom next-generation CAR construction, please refer to our related services: CAR Design & Construction. To further assess your switch CAR biological efficacy (e.g., cytokine production, tumor killing, and CAR-T cell proliferation), our scientists can also provide comprehensive downstream services to complete your whole research. Please click: CAR Cell In Vitro Assay Service, CAR-T Preclinical In Vivo Assay for more details.
For more detailed information, please feel free to contact us or directly sent us an inquiry.
Reference
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION