All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
Here is a brief list of some HIV-1 CAR products. You can also search for products that interest you by filtering the corresponding keywords.
After human immunodeficiency virus type 1 (HIV-1) infection, combination antiretroviral therapy (ART) can effectively inhibit viral replication. However, because the virus integrates into infected cells and forms a stable reservoir of latent infection, infected individuals can re-erupt in viremia within a short period of time once they stop ART therapy, which constitutes a major obstacle to curing HIV-1 infection. Studies have shown that in ART-treated infected patients, even if the latent infection is successfully activated, the CD8+ T lymphocytes in the body cannot completely eliminate the infected cells due to the lack of an effective immune response to HIV-1. Therefore, in order to better clear the virus reservoir, it is necessary to re-establish a robust immune surveillance function in infected individuals.
Chimeric antigen receptor (CAR) immune cell therapy has become a new way to kill tumor cells due to its high affinity, non-restriction of MHC, and recognition of naïve antigen. CAR can specifically kill tumor cells in patients by expressing CAR molecules that recognize target antigens in the patient's own immune cells and performing adoptive immune reinfusion. CAR-T cell therapies can be used for antiviral treatments, including HIV-1, hepatitis B virus (HBV), and hepatitis C virus (HCV).
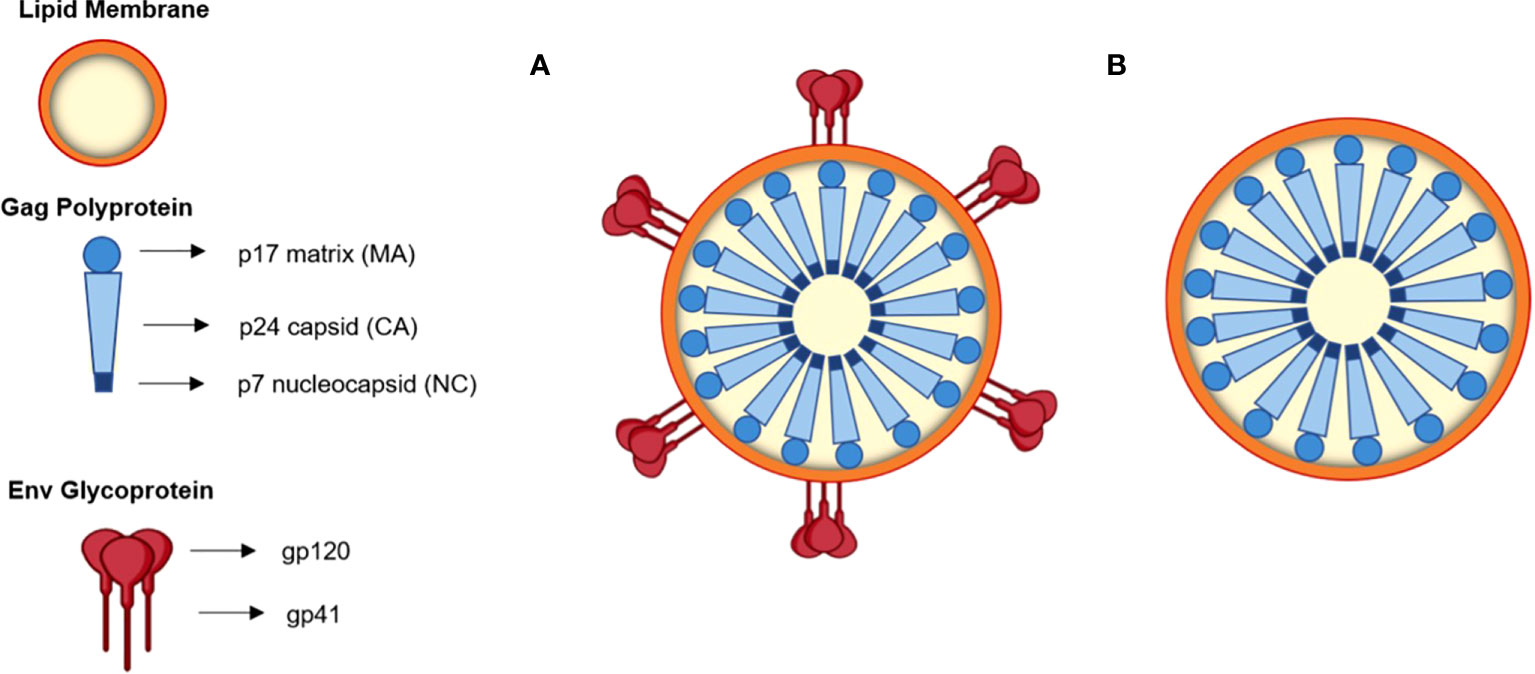 Fig.1 Schematic diagram of HIV-1-based virus-like particles.1
Fig.1 Schematic diagram of HIV-1-based virus-like particles.1
HIV-1 infection
CAR-T cell therapies based on broadly neutralizing antibodies have shown tremendous efficacy and safety in HIV. Creative Biolabs designs HIV-1-specific CAR-T cells using PBMCs and CAR constructs.
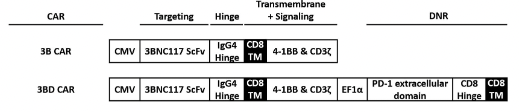 Fig.2 Schematic diagram of the HIV-1 CAR construction.2
Fig.2 Schematic diagram of the HIV-1 CAR construction.2
Anti-HIV-1 CAR Cell Proliferation Test
Creative Biolabs offers a variety of cell proliferation assays to help customers evaluate the ability of HIV-1 CAR-T cells to grow in vitro. Services we offer include but are not limited to Cell Counting Kit-8 (CCK-8), MTT, MTS, 5,6-Carboxyfluorescein Diacetate Succinimidyl Ester (CFSE), [3H] thymidine incorporation.
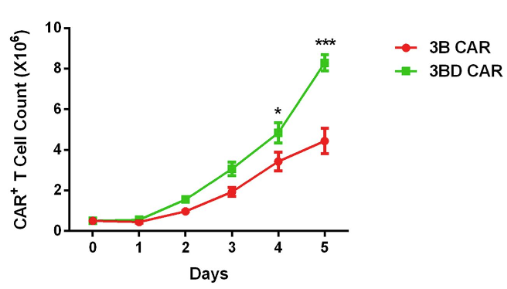 Fig.3 Proliferation of anti-HIV-1 CAR-T cells (3BD CAR-T) in vitro. The results showed enhanced accumulation of anti-HIV-1 CAR-T cells.2
Fig.3 Proliferation of anti-HIV-1 CAR-T cells (3BD CAR-T) in vitro. The results showed enhanced accumulation of anti-HIV-1 CAR-T cells.2
Anti-HIV-1 CAR-T Cytokine Release Test
The multiplex cytokine detection kit developed by Creative Biolabs based on liquid microarray technology has the characteristics of high sensitivity, high detection throughput, and short detection time, and is used for the quantitative detection of interleukin, interferon, tumor necrosis factor and other cytokines in specimens in vitro.
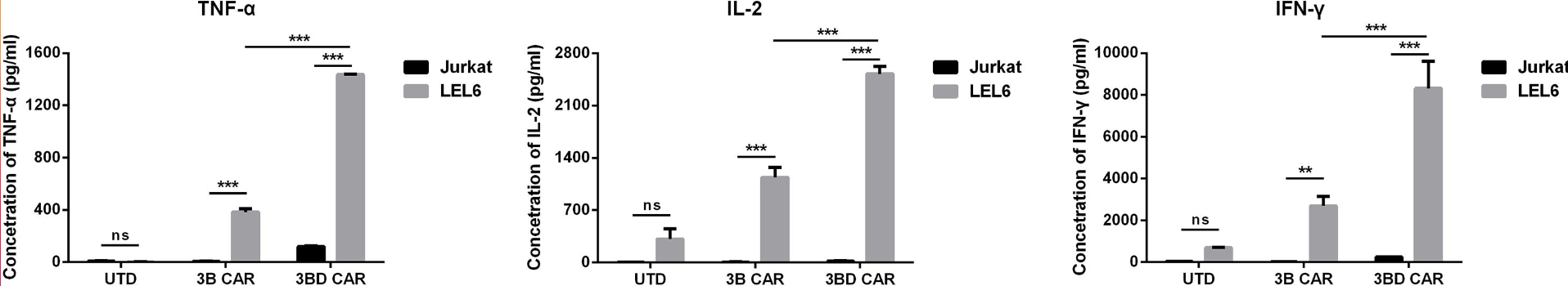 Fig.4 TNF-α, IL-2 and IFN-r were detected by ELISA. The results showed that anti-HIV-1 CAR-T cells exhibited enhanced cytokine secretion.2
Fig.4 TNF-α, IL-2 and IFN-r were detected by ELISA. The results showed that anti-HIV-1 CAR-T cells exhibited enhanced cytokine secretion.2
Anti-HIV-1 CAR-T In Vitro Cytotoxicity Assay
Creative Biolabs offers in vitro toxicity testing for HIV-1 CAR-T cells. We reflect the killing effect of CAR-T cells by mixing CAR-T cells with tumor cells or constructing antigen-overexpressing cell models to detect the death of tumor cells or antigen-overexpressing cell models. Among them, flow cytometry can detect the apoptosis of tumor cells.
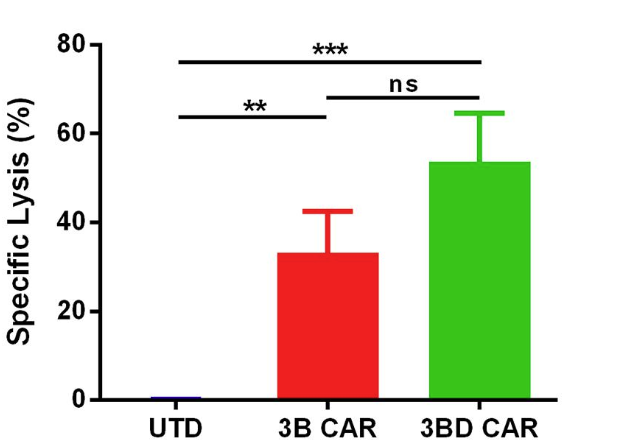 Fig.5 The ability of anti-HIV-1 CAR-T cells to kill latently infected cells.2
Fig.5 The ability of anti-HIV-1 CAR-T cells to kill latently infected cells.2
Anti-HIV-1 CAR-T Cell Therapy Animal Models
In addition to in vitro cell models, Creative Biolabs provides suitable animal models and comprehensive efficacy and safety evaluation protocols for your HIV-1 CAR-T research.
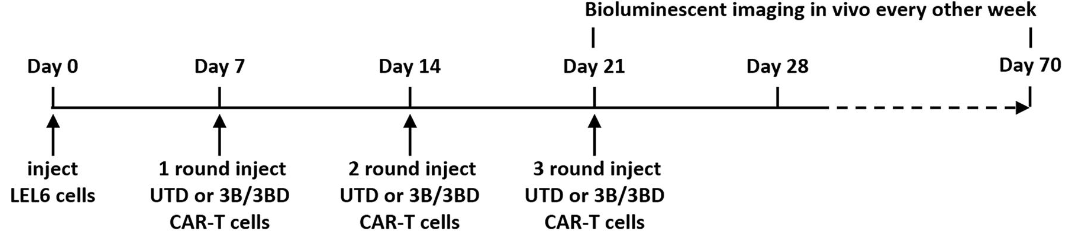 Fig.6 Schematic diagram of the experimental design of anti-HIV-1 CAR-T cell in vivo therapy.2
Fig.6 Schematic diagram of the experimental design of anti-HIV-1 CAR-T cell in vivo therapy.2
Efficacy Test of Anti-HIV-1 CAR-T
Our team of experts offers a comprehensive approach to in vivo pharmacodynamics studies to provide authentic and reliable preclinical data for your HIV-1 CAR-T therapy.
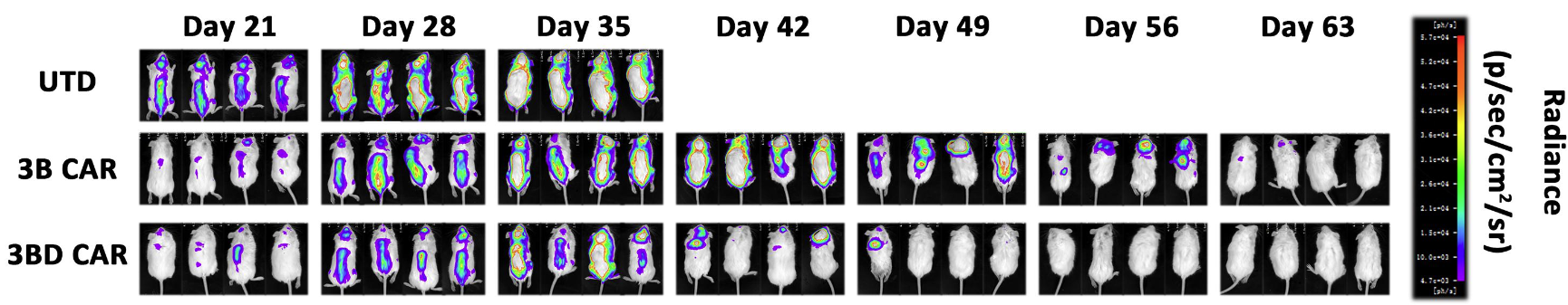 Fig.7 Serial bioluminescence imaging to evaluate the in vivo efficacy of anti-HIV-1 CAR-T cells.2
Fig.7 Serial bioluminescence imaging to evaluate the in vivo efficacy of anti-HIV-1 CAR-T cells.2
References
 Loading...
Loading...
| CAT | Product Name | Target Species | Antibody Clone | Antibody Host | Receptor Construction | Vector Type | Targeting Cell Type | CAR Vector Type | Inquiry & Datasheet |
| CAR-MZ210 | Anti-HIV-1 (10E8) h(CD3ζ) CAR, pCDCAR1 | Virus | 10E8 | Human | scFv-CD3ζ | Lentiviral | T cell | ||
| CAR-MZ211 | Anti-HIV-1 (3BNC117) h(CD3ζ) CAR, pCDCAR1 | Virus | 3BNC117 | Human | scFv-CD3ζ | Lentiviral | T cell | ||
| CAR-MZ212 | Anti-HIV-1 (PG9) h(CD3ζ) CAR, pCDCAR1 | Virus | PG9 | Human | scFv-CD3ζ | Lentiviral | T cell | ||
| CAR-YF159 | Anti-HIV-1 (PG9) h(4-1BB-CD3ζ) CAR, pCDCAR1 | HIV-1 | PG9 | Human | scFv-4-1BB-CD3ζ | Lentiviral vector | T cell | ||
| CAR-YF160 | Anti-HIV-1 (PG9) h(CD28-CD3ζ) CAR, pCDCAR1 | HIV-1 | PG9 | Human | scFv-CD28-CD3ζ | Lentiviral vector | T cell | ||
| CAR-ZP1784 | Anti-HIV-1 (PG16) h(CD28-CD3ζ) CAR, pCDCAR1 | Human | PG16 | Human | scFv-CD28-CD3ζ | Lentiviral vector | T cell | ||
| CAR-ZP1785 | Anti-HIV-1 (PG16) h(41B-CD3ζ) CAR, pCDCAR1 | Human | PG16 | Human | scFv-41BB-CD3ζ | Lentiviral vector | T cell | ||
| CAR-ZP1786 | Anti-HIV-1 (447-52D) h(CD28-CD3ζ) CAR, pCDCAR1 | Human | 447-52D | Human | scFv-CD28-CD3ζ | Lentiviral vector | T cell | ||
| CAR-ZP1787 | Anti-HIV-1 (447-52D) h(41BB-CD3ζ) CAR, pCDCAR1 | Human | 447-52D | Human | scFv-41BB-CD3ζ | Lentiviral vector | T cell | ||
| CAR-ZP1788 | Anti-HIV-1 (PGT145) h(CD28-CD3ζ) CAR, pCDCAR1 | Human | PGT145 | Human | scFv-CD28-CD3ζ | Lentiviral vector | T cell | ||
| CAR-ZP1789 | Anti-HIV-1 (PGT145) h(41BB-CD3ζ) CAR, pCDCAR1 | Human | PGT145 | Human | scFv-41BB-CD3ζ | Lentiviral vector | T cell | ||
| CAR-ZP1790 | Anti-HIV-1 (H27-14) h(CD28-CD3ζ) CAR, pCDCAR1 | Human | H27-14 | Human | scFv-CD28-CD3ζ | Lentiviral vector | T cell | ||
| CAR-ZP1791 | Anti-HIV-1 (H27-14) h(41BB-CD3ζ) CAR, pCDCAR1 | Human | H27-14 | Human | scFv-41BB-CD3ζ | Lentiviral vector | T cell | ||
| CAR-ZP1792 | Anti-HIV-1 (3BNC60) h(CD28-CD3ζ) CAR, pCDCAR1 | Human | 3BNC60 | Human | scFv-CD28-CD3ζ | Lentiviral vector | T cell | ||
| CAR-ZP1793 | Anti-HIV-1 (3BNC60) h(41BB-CD3ζ) CAR, pCDCAR1 | Human | 3BNC60 | Human | scFv-41BB-CD3ζ | Lentiviral vector | T cell | ||
| XS-0622-ZP3320 | Anti-HIV-1 h(VHH1-VHH2-CD28-CD3ζ) Biepitopic CAR, pCDCAR1 | Human | VHH1-VHH2-CD28-CD3ζ | Lentiviral vector | T Cell | ||||
| XS-0622-ZP3492 | Anti-HIV-1 h(VHH1-VHH2-4-1BB-CD3ζ) Biepitopic CAR, pCDCAR1 | VHH1-VHH2-4-1BB-CD3ζ | Lentiviral vector | T Cell | |||||
| XS-0822-YF511 | Anti-Human HIV-1 (XW-511) h(41BB-CD3ζ) CAR IVT Plasmid, pCARIVT | Human | XW-511 | Mouse | scFv-41BB-CD3ζ | In Vitro Transcription (IVT) Vector | |||
| XS-0822-YF1431 | Anti-Human HIV-1 (XW-511) h(CD28-CD3ζ) CAR IVT Plasmid, pCARIVT | Human | XW-511 | Mouse | scFv-CD28-CD3ζ | In Vitro Transcription (IVT) Vector | |||
| XS-0822-YF2351 | Anti-Human HIV-1 (XW-511) h(CD28-41BB-CD3ζ) CAR IVT Plasmid, pCARIVT | Human | XW-511 | Mouse | scFv-CD28-41BB-CD3ζ | In Vitro Transcription (IVT) Vector |
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION