All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
The αβ T-cell receptor (TCR) serves as a pan-T cell antigen which is highly expressed in T-cell malignancies. However, targeting the αβ TCR to treat T-cell malignancies would result in T-cell aplasia, which can cause severe immunosuppression. Meanwhile, it is impractical to generate customized drugs for each patient due to the diversity of T-cell clones in patients. Since T cells exclusively express either TRBC1 or TRBC2 in T-cell malignancy patients, the development of specific CAR-T cells targeting T-cell receptor β-chain constant region (TRBCs) may be more feasible for effective treatment of T-cell lymphoma.
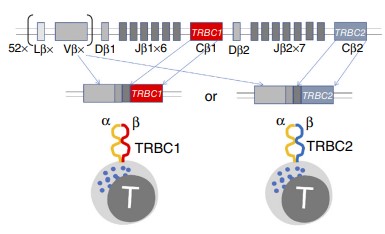 Fig.1 T cells exclusively express either TRBC1 or TRBC2 in T-cell malignancies patients.1
Fig.1 T cells exclusively express either TRBC1 or TRBC2 in T-cell malignancies patients.1
Leverage by our years of experience in the CAR-T field, Creative Biolabs provides novel TRBCs-targeted CAR-T engineering services to assist global customers' T cell malignancy therapy research. As T cell malignancies are highly heterogeneous in terms of oncogenic-driver pathways, we are committed to developing appropriate TRBCs-targeted CAR-T engineering services according to the variable needs of customers. At the same time, we offer a comprehensive range of services, including from CAR-T design & construction, and delivery system construction, to in vitro and in vivo validation to speed the process of customers' research.
Compared to other common CAR-T approaches for T cell malignancies, this strategy has the following advantages:
One-stop CAR-T development to assure our top-ranking service
Highly experienced scientists to assist CellRapeutics™ TRBCs-targeted CAR-T engineering services

Reliable and reputable experiment systems to guarantee high-quality services
Comprehensive customized services to meet the variable needs of global customers
Representative data 1: Anti-TRBC1 CAR-T for immunotherapy of T cell malignancies
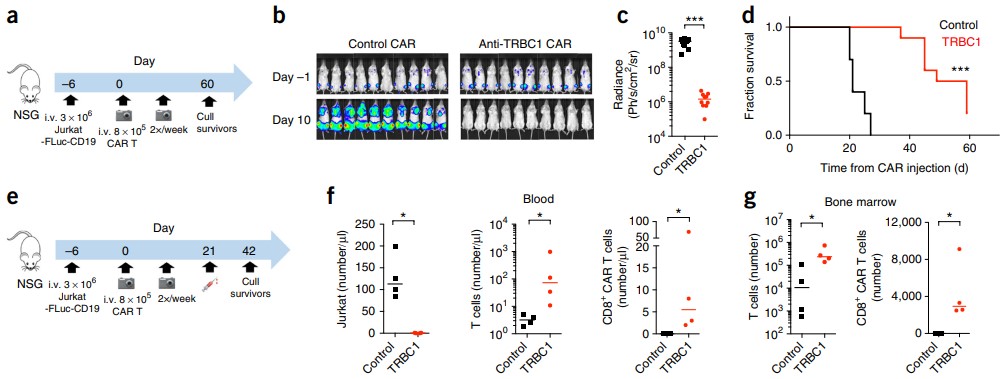 Fig.2 In vivo efficacy and specificity validation of anti-TRBC1 CAR-T cells in T cell malignancy mouse model.1
Fig.2 In vivo efficacy and specificity validation of anti-TRBC1 CAR-T cells in T cell malignancy mouse model.1
Q1: What are the challenges for TRBCs-targeted CAR-T engineering?
A1: In the TRBCs-targeted CAR-T engineering development process, how to ensure high effectiveness while avoiding possible security problems will always be a key consideration. Based on the years of CAR-T research experience, we adopt combination designs to compare and optimize, and at the same time conduct several safety monitoring to investigate the security issues of this strategy from multiple aspects of efficacy and safety evaluations.
Q2: Is it possible to develop a TCRV-targeted CAR-T engineering service?
A2: During T cell development, variable-diversity-joining (VDJ)-domain recombination leads to a diversity of T cell receptors, which results in unique TCR Vβ and Vα features in specific T cell clones. Due to the diversity of TCRV subtypes, developing TCRV-targeted CAR-T may be more diverse and flexible. If our customers need some related service, we are committed to developing custom services.
For more details about our CellRapeutics™ TRBCs-targeted CAR-T engineering services, please feel free to contact us or send us a query.
Reference
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION