All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
With the development of tumor immunotherapy, a great number of markers have been found to predict immunotherapy response. More and more evidence highlights that T cells infiltrating in the tumor microenvironment play an important role in the efficacy of cancer immunotherapy. However, the level of distinct T cell subset infiltration and the signals that attract them to tumor sites are still rarely characterized in different cancer types. Therefore, there is an urgent need to develop reliable T cell-related biomarkers profiling for immunotherapy response and prognosis.
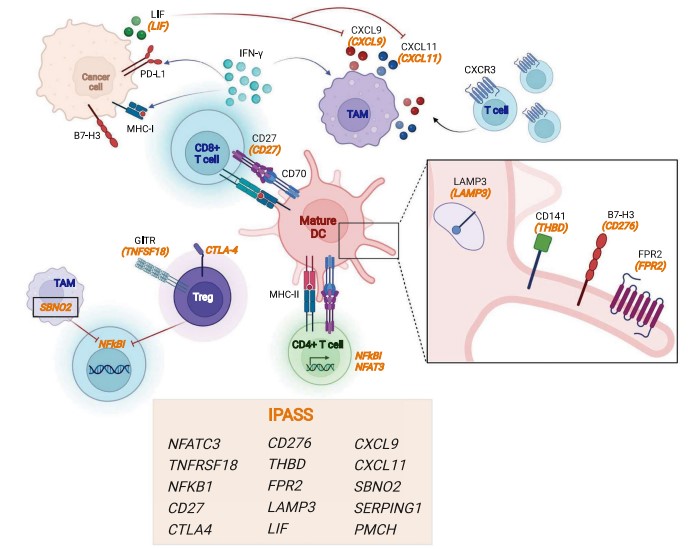 Fig.1 The IPASS gene signature description of a complex immune network in pediatric cancers.1
Fig.1 The IPASS gene signature description of a complex immune network in pediatric cancers.1
Together with our seasoned and expert research teams, Creative Biolabs specializes in providing comprehensive T-cell signature profile detection services for global customers to speed up immunotherapy projects. Our service performed and validated a new T-cell-related prognostic signature profiling by integrating a variety of analysis, such as single-cell sequencing, and multiple RNA sequencing. The combined service will offer new insights into the function of immune cell marker genes in the prognosis and immunotherapy response. Powered by our advanced technology platform, we offer one-stop services from sample treatment to data visualization to meet global customers’ diverse needs.
Here are some results displayed that refer to the drawings of T cell signature profiling articles.
| 1. Validation of T cell-related signature for prognosis and immunotherapy response in HCC. | |
|
|
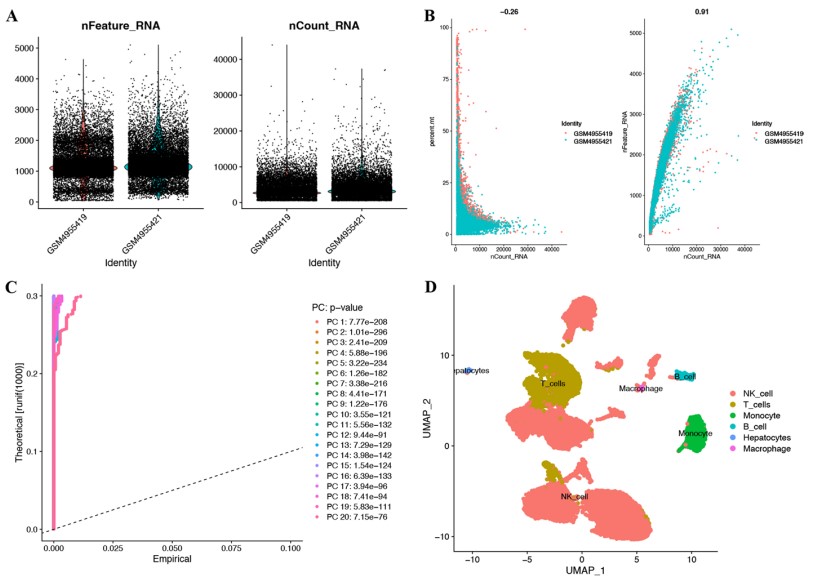
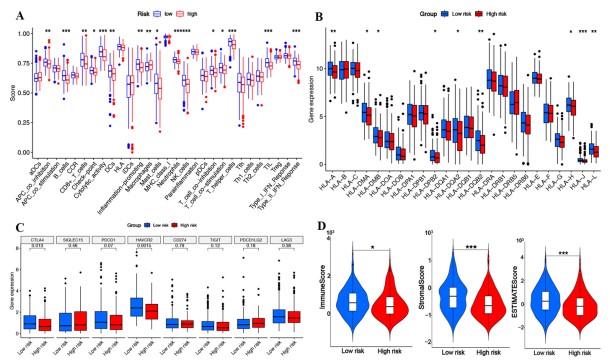
| Fig.2 The single-cell RNA sequencing analysis from two HCC samples.2 | Fig.3 Immune microenvironment landscape and functional enrichment analysis between high- and low-risk groups.2 |
| 1. Identification and validation of T-cell infiltration in high-risk pediatric cancer | |

|
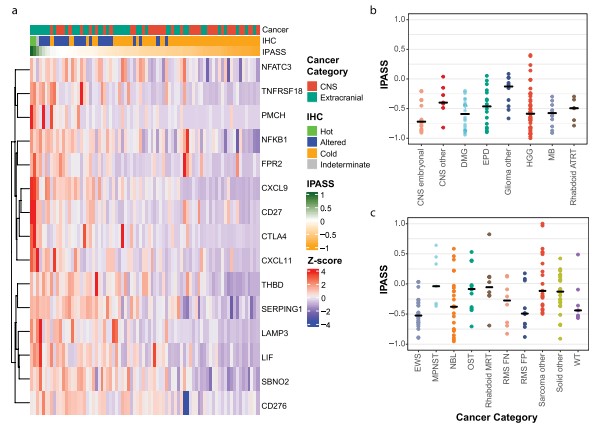
|
| Fig.4 Bulk RNA sequencing deconvolution in pediatric cancer.1 | Fig.5 Novel pediatric immune signature analysis to predict T-cell infiltrated tumors.1 |
Q1: What are the limitations that should be considered in T cell signature profiling?
A1: In the analysis, it is necessary to consider multiple related factors, such as sample quantity, if the sample to low which may cause the data to be not very explanatory. In addition, it is necessary to consider the age and gender of the sample donor, as well as the tissue site chosen for the sample.
For more details about our CellRapeutics™ T cell signature profile detection service, please feel free to contact us. We will be pleased to receive your inquiry. Sincerely look forward to cooperating with global customers.
References
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION