All products and services are For Research Use Only and CANNOT be used in the treatment or diagnosis of disease.
As a recognized service provider, Creative Biolabs'highly skilled team can provide CellRapeutics™ first generation of CARs products to meet your specific needs. Creative Biolabs has built up a unique and unparalleled platform for producing a variety of CARs engineered immune cells, which can greatly help and accelerate your research progress.
Donald R. Shaffer, Penghui Zhou and Stephen Gottschalk. Foreign or Domestic CARs: Receptor Ligands as Antigen-Binding Domains. Med. Sci. 2014, 2(1), 23-36.
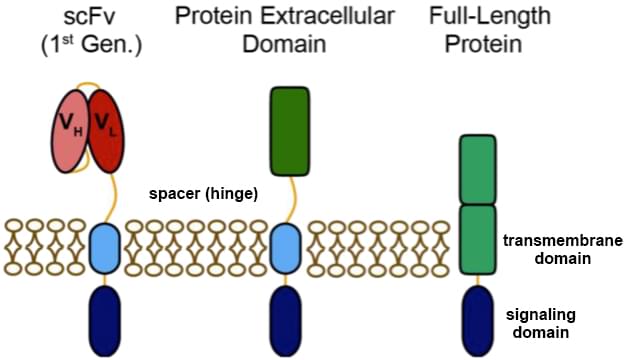
The design of the first generation CARs includes a scFv targeting tumor associated antigen (TAA), a space (hinge), a transmembrane region (TM) and an intracellular signaling domain. Clinical trials have shown that the first generation CAR-engrafted T cells are a very feasible concept in the treatment for some cancer patients. The application of CAR-engineered T cells have more advantages than the traditional T cell immunotherapy, which includes major histocompatibility complex (MHC)-independent antigen recognition in treating viral infection as well as cancer. Recent reports show that the first generation CAR modified T cells with anti-CD19 specificity has entered phrase I clinical trial for B cell malignances.
The scFv of the first generation CAR is derived from a monoclonal antibody specific to TAAs which is presented on tumor cell surface in various malignancies. Our seasoned scientists can provide a broad range of highly customized service for scFvs design to meet the growing requirements from our clients. An intracellular signaling domain is derived from the CD3ζ chain of the T cell receptor (TCR) complex, which can activate T cells. In addition to CD3ζ, FcεRIγ can also be used as an intracellular signaling domain.
Creative Biolabs offers you sophisticated strategy and the best-in-class CellRapeutics™ first generation CARs products, which can meet and exceed your original expectations.
For any technical issues or product/service related questions, please leave your information below. Our team will contact you soon.
 NEWSLETTER
NEWSLETTER
The latest newsletter to introduce the latest breaking information, our site updates, field and other scientific news, important events, and insights from industry leaders
LEARN MORE NEWSLETTER NEW SOLUTION
NEW SOLUTION
CellRapeutics™ In Vivo Cell Engineering: One-stop in vivo T/B/NK cell and macrophage engineering services covering vectors construction to function verification.
LEARN MORE SOLUTION NOVEL TECHNOLOGY
NOVEL TECHNOLOGY
Silence™ CAR-T Cell: A novel platform to enhance CAR-T cell immunotherapy by combining RNAi technology to suppress genes that may impede CAR functionality.
LEARN MORE NOVEL TECHNOLOGY NEW SOLUTION
NEW SOLUTION
Canine CAR-T Therapy Development: From early target discovery, CAR design and construction, cell culture, and transfection, to in vitro and in vivo function validation.
LEARN MORE SOLUTION