Immunophenotyping based Cell Population Characterization Services
Background Service Highlights FAQs Contact Us
Introduction: Deciphering Cellular Complexity with Precision
In the intricate landscape of cellular biology and therapeutic development, understanding the composition and functional state of heterogeneous cell populations is paramount. At Creative Biolabs, we harness the power of high-dimensional flow cytometry to provide immunophenotyping services that are not merely data points, but functional insights that accelerate drug discovery, clarify mechanisms of action, and support clinical translation. The ability to accurately delineate a rare regulatory T-cell subset, identify a transient pharmacodynamic response in myeloid cells, or confirm target engagement on malignant B-cells can be the pivotal factor that propels a research program toward success. Our philosophy is built on a foundation of deep scientific expertise and a commitment to providing exquisitely detailed and reliable cellular characterization.
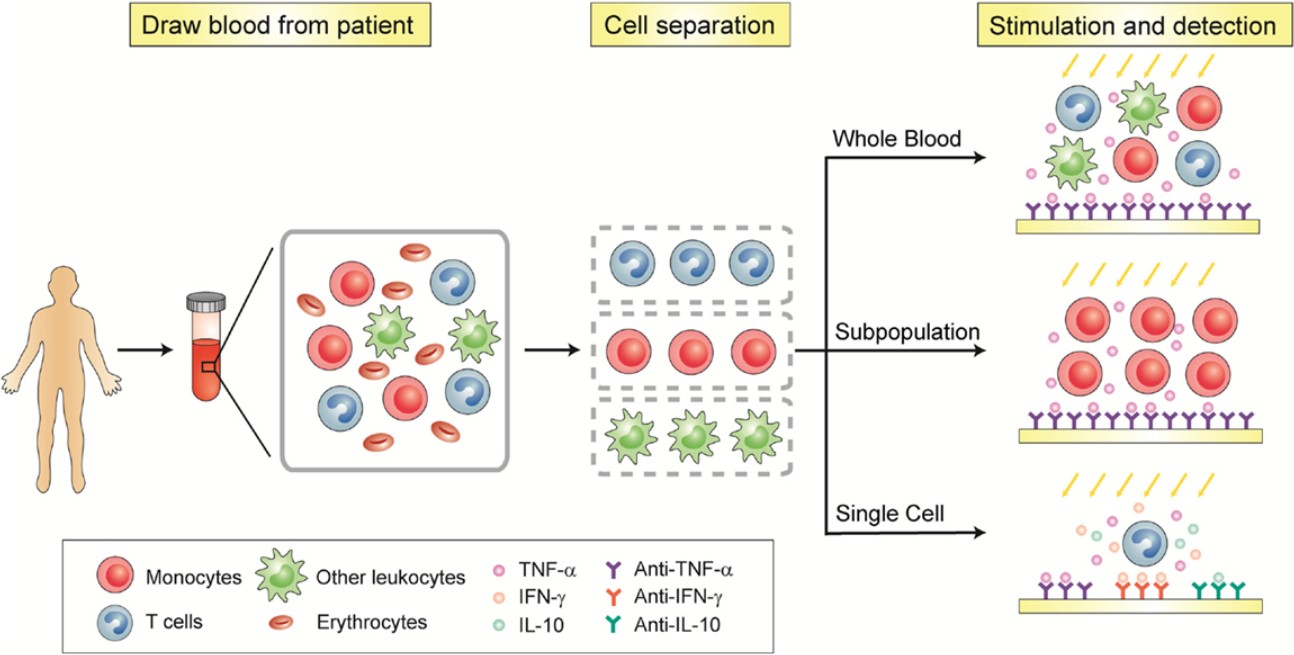 Fig.1 Schematic of immunophenotyping of immune cells.1
Fig.1 Schematic of immunophenotyping of immune cells.1
Our Immunophenotyping based Cell Population Characterization Services
High-dimensional immunophenotyping by flow cytometry is the gold-standard methodology for identifying, quantifying, and functionally characterizing discrete cell populations within a complex biological sample, such as whole blood, peripheral blood mononuclear cells (PBMCs), or dissociated tissues. The technique leverages the exquisite specificity of fluorochrome-conjugated monoclonal antibodies to label cell surface and intracellular protein markers, building a unique "phenotypic signature" for each individual cell.
Comprehensive Service Content
At Creative Biolabs, we offer an end-to-end solution, from initial consultation and experimental design to final, presentation-ready data analysis and reporting.
1. Expert Consultation and Custom Panel Design
Every project begins with a deep-dive consultation with our Ph.D.-level scientists. We recognize that no two studies are identical. Based on your specific research questions, biological matrix, and therapeutic modality, we design fully customized, multi-parameter flow cytometry panels. This process involves:
-
Target Selection: Identifying the most relevant biomarkers to define your populations of interest.
-
Fluorochrome Optimization: Selecting the optimal combination of fluorochromes to minimize spectral overlap and maximize signal resolution, a critical challenge in high-parameter cytometry. We leverage our experience with the latest dye technologies to build robust panels of 10, 15, and even 20+ colors.
-
Validation Strategy: Employing fluorescence-minus controls and other rigorous validation methods to ensure accurate gating and data interpretation.
2. Targeted Immunophenotyping Services by Cell Type
We provide specialized expertise in isolating and characterizing a diverse array of cell types from various tissues, each requiring a unique technical approach.
Our core expertise. We perform deep profiling of all leukocyte subsets, including rare and nuanced populations like T-cells (naïve, memory, effector, regulatory), B-cells (transitional, memory, plasma cells), NK cells, monocytes, and dendritic cells (pDCs, cDC1, cDC2).
We accurately identify and quantify hematopoietic (HSCs), mesenchymal (MSCs), and pluripotent (PSCs) stem cells using canonical markers such as CD34, CD90, and SSEA-4, ensuring precise characterization for regenerative medicine and oncology research.
We dissect complex neural cultures and dissociated brain tissue to distinguish between neuronal populations (e.g., NeuN+), astrocytes (GFAP+), microglia (Iba1+), and oligodendrocyte precursors, providing key insights for neuro-inflammatory and neurodegenerative disease models.
We characterize adipocyte populations and their associated immune cells from fat tissue, a critical service for metabolic disease research, focusing on markers related to inflammation and metabolic state.
We phenotype distinct cardiac cell populations, including cardiomyocytes (cTnT+), fibroblasts, and infiltrating immune cells from heart tissue to support research in cardiovascular disease, fibrosis, and myocarditis.
We identify and characterize endothelial cells (e.g., CD31+, VE-Cadherin+) and their activation states, crucial for studies in angiogenesis, vascular inflammation, and tumor vascularization.
We analyze skin cell suspensions to phenotype keratinocytes, melanocytes, and resident immune cells like Langerhans cells, supporting dermatological and cosmetic research.
We distinguish various epithelial cell subsets from different organs using markers like EpCAM and cytokeratins, essential for research in cancer, infectious disease, and wound healing.
We perform phenotypic analysis of primary hepatocytes and other non-parenchymal liver cells (e.g., Kupffer cells, stellate cells) to advance studies in liver disease, toxicology, and metabolism.
We characterize smooth muscle cells from vascular and other tissues using markers like alpha-smooth muscle actin (α-SMA), supporting research in atherosclerosis, hypertension, and asthma.
3. Functional Cellular Assays
Beyond surface marker expression, we measure what your cells are doing.
-
Cell Viability and Apoptosis Analysis: Quantifying programmed cell death using established methods like Annexin V and 7-AAD staining to assess compound cytotoxicity.
-
Cell Proliferation and Cycle Analysis: Measuring cell division using dye dilution assays or DNA content analysis to evaluate the cytostatic or cytotoxic effects of novel therapeutics.
-
Receptor Occupancy (RO) Assays: Quantifying the binding of a therapeutic antibody to its cell-surface target, providing direct evidence of target engagement.
-
Intracellular Cytokine Staining (ICS): Measuring the functional capacity of cells by detecting cytokine production (e.g., IFN-γ, TNF-α, IL-2) at the single-cell level following stimulation.
4. Advanced Data Analysis and Bioinformatics
Raw flow cytometry data is only the beginning. Our bioinformatics team utilizes advanced analytical tools, including dimensionality reduction and unsupervised clustering algorithms, to reveal subtle but significant patterns in complex datasets. This moves beyond manual gating to provide an unbiased, comprehensive view of the cellular landscape and treatment-induced changes.
Our Advantages
In a competitive landscape of both global CROs and specialized boutiques, Creative Biolabs differentiates itself through three core pillars: scientific rigor, technological excellence, and a truly collaborative partnership model.
-
Unmatched Expertise
-
State-of-the-Art Technology
-
Regulatory-Aware Environment
FAQs
Q1: What sample types can you analyze?
A1: We routinely work with a wide range of sample types, including fresh or cryopreserved whole blood, PBMCs, bone marrow aspirates, and cells isolated from dissociated tissues (e.g., spleen, lymph nodes, tumors). Please contact us to discuss the specifics of your model system.
Q2: How do you handle panel design for a novel target?
A2: For novel targets or unique cell populations, we initiate a comprehensive panel development project. This involves sourcing and testing multiple antibody clones, optimizing titration, and establishing a robust staining and gating strategy before analyzing any precious study samples.
Q3: Can you help with the data analysis and interpretation?
A3: Absolutely. All our service packages include comprehensive data analysis, a detailed final report with high-resolution graphics, and a consultation with one of our scientists to review the findings and discuss next steps.
Contact Us
To discover how our advanced cell population characterization services can bring clarity and insight to your drug discovery and development programs, please contact our scientific liaison team. We are ready to discuss your unique project needs and design a solution that delivers precise, actionable data. Connect with a Creative Biolabs expert today.
Reference
-
Chen, Weiqiang et al. "Emerging microfluidic tools for functional cellular immunophenotyping: a new potential paradigm for immune status characterization." Frontiers in oncology vol. 3 98. 22 Apr. 2013. DOI: 10.3389/fonc.2013.00098. Distributed under Open Access License CC BY 4.0, without modification.


 Fig.1 Schematic of immunophenotyping of immune cells.1
Fig.1 Schematic of immunophenotyping of immune cells.1
