Human Breast Cell MDA-MB-468 based Proliferation Assay Service
Creative Biolabs offers a definitive, high-confidence solution for efficacy testing against the highly aggressive triple-negative breast cancer (TNBC) subtype. We leverage the established biology of the MDA-MB-468 cell line to move beyond simple IC50 measurements. Our service provides a complete package for lead candidate selection, including detailed mechanistic validation to understand the drug's mode of action. We also help develop strategies for drug resistance circumvention, ensuring your therapeutic candidates have the highest chance of success against this challenging cancer.
Background What We Can Offer Workflow Publication Why Choose Us FAQs Customer Review Related Services Contact Us
The MDA-MB-468 Model: Your Gold Standard for TNBC Efficacy Testing
The MDA-MB-468 cell line is the most recognized in vitro model for TNBC, making it invaluable for drug screening. Its features, including triple-negative status (lacking ER, PR, HER2), make it ideal for screening novel, non-hormonal agents. It exhibits high EGFR expression, allowing perfect testing of EGFR inhibitors, and PD-L1 Expression, which is essential for modeling T-cell exclusion and testing checkpoint inhibitors. As a Basal-like 2 (BL2) subtype, it represents a highly aggressive phenotype, ensuring preclinical findings are highly translational.
Creative Biolabs’ Technical Superiority: High Sensitivity and Customization
Our standardized protocol minimizes inter-assay variability while maximizing data quality. We employ state-of-the-art detection technology to deliver results that are reliable, reproducible, and ready for regulatory submission:
High-Sensitivity Luminescent Detection
Provides a wider dynamic range and enhanced sensitivity compared to colorimetric methods, yielding a superior signal-to-noise ratio for unambiguous, clean data.
Tailored Protocols and Comprehensive Secondary Validation
We offer fully customizable protocols and specialize in immuno-oncology (IO) co-culture models for measuring immunotherapeutic efficacy. We seamlessly integrate secondary validation assays, including flow cytometry (for cell cycle/apoptosis) and western blotting (for markers like EGFR and PD-L1).
Comprehensive Data Analysis
Full-service reports include dose-response curve plotting, IC50 calculation, comparison with controls, and expert interpretation by senior oncology specialists.
Contact us today for a confidential consultation and customized quote.
Workflow: Comprehensive Path from Compound to Conclusion
We follow a meticulously detailed, multi-step process designed for maximum clarity and data integrity, ensuring every step of your TNBC drug screening campaign is optimized for rigor and reliability.
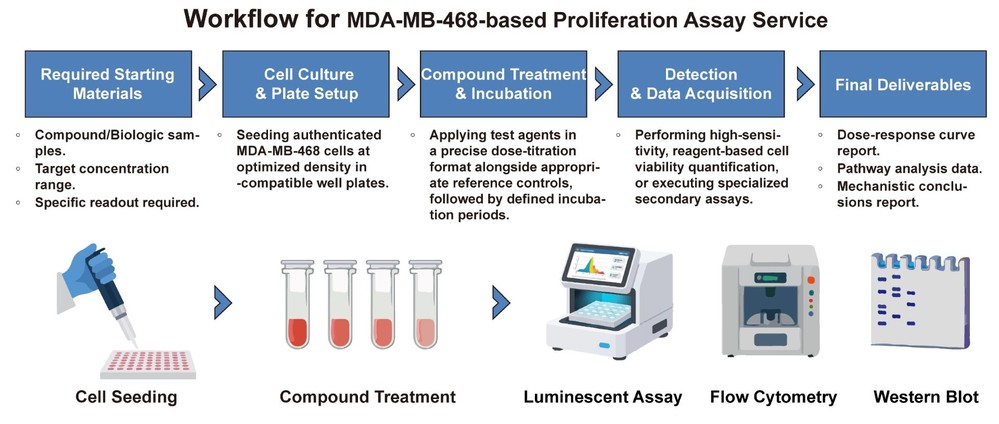
Publication
This study investigates combining the glycolytic inhibitor 2-deoxyglucose (2DG) with metformin for TNBC, which heavily relies on glucose metabolism. Using two TNBC lines under high-glucose conditions, the dual treatment more effectively reduced cell proliferation, viability, and mTOR signaling than monotherapy. The efficacy was influenced by metabolic heterogeneity between TNBC subtypes, highlighting the need to personalize treatment strategies based on a tumor's metabolic profile.
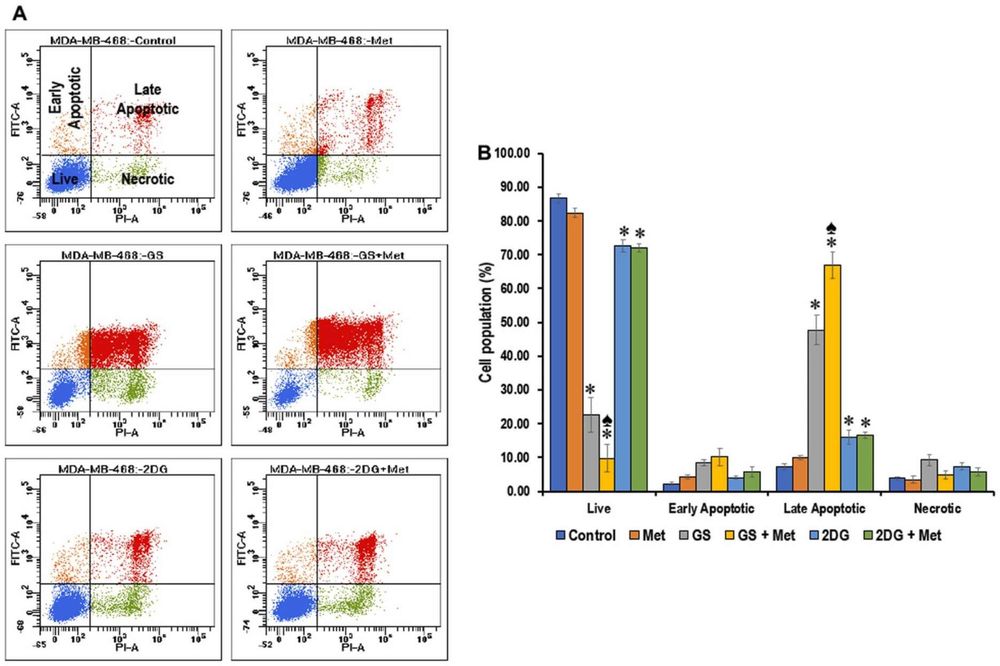 Fig.1 Metformin enhances apoptosis in glucose-starved or 2DG-exposed MDA-MB-468 cells. 1
Fig.1 Metformin enhances apoptosis in glucose-starved or 2DG-exposed MDA-MB-468 cells. 1
Why Choose Us
Creative Biolabs sets the gold standard for TNBC drug screening by moving beyond standard assays to provide deep mechanistic insights. Our expertise utilizes the aggressive MDA-MB-468 cell line, characterized by high EGFR and PD-L1 expression. This specialization gives your project a decisive advantage in identifying highly effective lead candidates by modeling clinically relevant resistance mechanisms. We combine a robust technical platform with scientific guidance, ensuring your preclinical findings are highly translational and significantly accelerating your drug development program.
Experience the Creative Biolabs Advantage - Get a Quote Today
FAQs
Q1: Why should I choose the luminescent assay over the cheaper, traditional MTT or SRB methods?
A1: Our high-sensitivity luminescent detection offers up to 100x greater sensitivity and a far wider dynamic range than colorimetric methods like MTT. This is critical for accurately quantifying the effects of rare or low-concentration compounds and reducing the variability that can compromise lead selection.
Q2: What specific immune cells can be integrated into the IO co-culture protocols?
A2: We routinely integrate peripheral blood mononuclear cells, isolated natural killer cells, and engineered T cells to leverage the MDA-MB-468 line's high PD-L1 expression. This provides a direct measure of immunotherapeutic efficacy.
Q3: Can your assay analyze drug efficacy under conditions relevant to the tumor microenvironment (e.g., hypoxia or metabolic stress)?
A3: Absolutely. Our protocols are highly customizable. We specialize in modeling metabolic vulnerabilities by adjusting glucose/glutamine media concentrations or simulating hypoxia to test compounds that specifically target the unique energy needs of aggressive TNBC cells.
Customer Review
-
Precision IO Co-culture Data
Using Creative Biolabs' service in our CAR-T research has significantly facilitated the accurate measurement of T-cell-mediated killing. Their proprietary co-culture protocol provided precise kinetic data that our in-house assay struggled to replicate, directly supporting our studies. - Dr. J**sW
-
Accelerated Mechanistic Validation
Using Creative Biolabs' service in our research has significantly improved the speed of target validation. We confirmed our compound's inhibition of the PI3K/Akt pathway via their high-quality western blot data. - Dr. D**lR
Related Services
To further accelerate your TNBC therapeutic development, Creative Biolabs highly recommends integrating our MDA-MB-468 proliferation assay with these complementary services:
Cell Cycle Progression Assessment Service
Creative Biolabs provides a precise cell cycle progression assessment service using advanced flow cytometry techniques. This service offers a detailed, cell-by-cell analysis of cell cycle phases to understand cell dynamics and the impact of therapeutic interventions.
Learn More →
Cell based Checkpoint Inhibitor Assay Service
Creative Biolabs' cell based checkpoint inhibitor assay precisely evaluates immune checkpoint interactions in a biologically relevant environment. We assess immune cell activation, cytokine production, and tumor cell apoptosis.
Learn More →
Contact Us
Creative Biolabs is your committed partner in conquering TNBC. Our human breast cell MDA-MB-468 based proliferation assay service delivers high-sensitivity data, unparalleled mechanistic insight into PI3K/Akt and metabolic pathways, and critical IO modeling capabilities. Trust our two decades of expertise to validate your lead candidates and drive your drug discovery forward.
Our specialists are available now to design a custom protocol tailored to your exact research goals. Contact Our Team for More Information and to Discuss Your Project
Reference
-
Samuel, Samson Mathews, et al. "Metabolic heterogeneity in TNBCs: A potential determinant of therapeutic efficacy of 2-deoxyglucose and metformin combinatory therapy." Biomedicine & Pharmacotherapy 164 (2023): 114911. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.1016/j.biopha.2023.114911



 Fig.1 Metformin enhances apoptosis in glucose-starved or 2DG-exposed MDA-MB-468 cells. 1
Fig.1 Metformin enhances apoptosis in glucose-starved or 2DG-exposed MDA-MB-468 cells. 1
