Human Colon Cell DLD-1 based Proliferation Assay Service
The DLD-1 cell line is a highly validated and essential model for predictable drug efficacy screening in colorectal cancer (CRC). Creative Biolabs provides comprehensive in vitro solutions that move beyond simple cell death checks. Our platform offers profound insights into therapeutic action by analyzing key molecular endpoints, including cell proliferation, apoptosis, IC50 determination, and cell cycle arrest patterns. We ensure the generated data package is robust, quantitative, and directly relevant to clinical translation, thereby accelerating your CRC drug development pipeline.
Background What We Can Offer Workflow Why Choose Us FAQs Customer Review Related Services Contact Us
The Criticality of the DLD-1 Model in Colorectal Cancer Research
The DLD-1 human colorectal adenocarcinoma cell line is a cornerstone model for predictive drug efficacy screening in CRC. Established in 1977, the line exhibits high clinical relevance, maintaining critical characteristics of primary human CRC. Its pathway fidelity allows for detailed investigation into complex signaling, and its established responsiveness to conventional therapies provides a reliable baseline for comparing novel agents. Leveraging the DLD-1 model—proven in high-throughput screening for targets like KLF5 inhibitors—ensures generated efficacy data has high translational potential.
Precision in Proliferation Assessment: Our Service Overview
High-Confidence Drug Screening for Potency Triage
We deliver validated IC50 values with narrow confidence intervals to rapidly and reliably select the most potent anti-cancer agents, supporting critical go/no-go decisions.
Mechanistic Pathway Deconvolution and Cell Fate Analysis
Our multi-pronged approach (colorimetric, HCI) definitively distinguishes between cytostatic effects, direct cytotoxicity, and non-proliferative cell fate (e.g., senescence), crucial for validating targeted therapies.
Advanced Modeling of Combination Efficacy
We rigorously evaluate the synergistic, additive, or antagonistic effects of drug combinations on proliferation and IC50 values, including the confirmation of autophagy or senescence induction.
Biomarker Identification and Resistance Profiling
We support parallel screening against wild-type and engineered resistant DLD-1 variants to identify potential biomarkers of sensitivity or intrinsic resistance, informing clinical trial design.
To discuss your compound library and determine the optimal screening density, please contact our technical team.
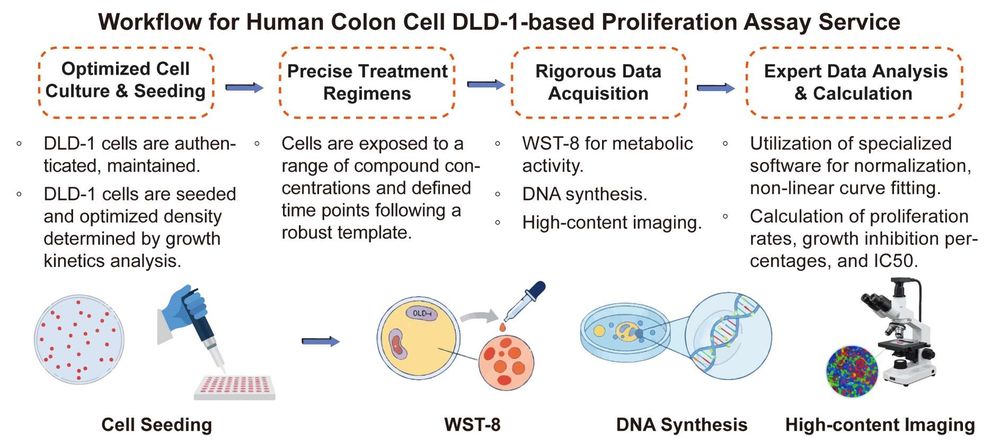
Workflow: A Meticulously Managed, Step-by-Step Efficacy Assessment
Our comprehensive assay procedure is meticulously managed by our Ph.D.-level scientists, ensuring consistency and maximizing data granularity, and is suitable for visualization as a clear flowchart.
Why Choose Us
Choosing Creative Biolabs for your DLD-1 assays means partnering with a team that delivers scientific excellence and translational insight. Our 20-year advantage in oncology research ensures deep domain knowledge and optimized, clinically relevant experimental designs. We offer customized assay design uniquely tailored to your compound's specific chemical and mechanistic properties, guaranteeing relevant results. Our reproducibility guarantee via quality management system ensures all data is highly traceable and reproducible, providing the high-confidence data required for regulatory submissions. Finally, we provide expert scientific interpretation, transforming raw data into actionable intelligence with clear reports and senior commentary to guide your next research steps.
Experience the Creative Biolabs Advantage - Get a Quote Today
FAQs
Q: How do your assays distinguish between cell growth inhibition and true cell death?
A: Our service utilizes a multi-modal approach. While colorimetric assays (MTT/WST-8) measure metabolic activity and overall viability (growth inhibition), we complement this with assays like high-content imaging, which directly measure the cell cycle, apoptosis, or senescence.
Q: Can I test compounds that are highly insoluble or require a high concentration of DMSO?
A: We offer preliminary solubility testing and toxicity profiling of the vehicle (e.g., DMSO) on DLD-1 cells. We will establish the maximum non-toxic concentration of your solvent to ensure that any observed inhibitory effects are solely due to your test compound, not vehicle toxicity.
Q: The DLD-1 cell line is often used for resistance studies; can your assay support this?
A: Absolutely. The DLD-1 model is highly versatile for resistance analysis. We can perform parallel IC50 screens against the wild-type DLD-1 and any of our engineered or custom-requested resistant sub-lines, providing critical comparative data for biomarker discovery.
Customer Review
-
Clear Mechanism of Action
Using Creative Biolabs' service in our research has significantly facilitated the distinction between a cytostatic effect and true apoptosis, which was critical for confirming our compound's MOA targeting the PI3K/AKT pathway. - S*n W***iams
-
Resistance Analysis Utility
Using Creative Biolabs' service in our research has significantly improved our resistance analysis by quickly comparing compound potency against wild-type and a p53-mutant DLD-1 variant, giving us an early read on potential patient selection biomarkers. - A**a P*l
Related Services
To complement your DLD-1 proliferation assay and fully advance your therapeutic agent, Creative Biolabs also recommends the following complementary services:
Function Assays for Immune Checkpoint
Immunotherapy targeting immune checkpoints is promising for cancer. Creative Biolabs offers a novel one-stop service with advanced platforms for immune checkpoint functional assays to identify target modulators.
Learn More →
Immunotoxicology
Creative Biolabs offers immunotoxicological studies and a wide breadth of assays to monitor safety risks and define immune endpoints in cancer immunotherapy preclinical testing.
Learn More →
Contact Us
Creative Biolabs is ready to partner with you to accelerate your oncology drug development. For detailed information on customizing this essential service to your specific therapeutic agent and research goals, please reach out to our team of experts today.



