LAG-3 Assay Portfolio Service
Lymphocyte activation gene-3 (LAG-3) is a new generation of immunotherapy targets and plays a balanced regulatory role in the human immune system. With advanced and high-end technologies, rich experienced scientists, Creative Biolabs is an excellent service provider in the field of tumor marker assay. After many years of research to fully comprehend tumor markers, we launched our LAG-3 assay portfolio service, which can be useful for targeted cancer therapy and diagnosis.
Background What We Can Offer Publication Why Choose Us FAQs Customer Review Related Services Contact Us
The Scientific Rationale: Understanding the Role of LAG-3
LAG-3 (CD223) is a 70 kDa protein belonging to the immunoglobulin superfamily. Its expression is detected on activated CD4+ and CD8+ T cells, regulatory T cells (Tregs), and other immune cells. It is involved in regulating the proliferation and activation of T lymphocytes, and its overexpression is associated with autoimmune diseases, tumors, and chronic toxic infectious diseases.
LAG-3 has several ligands, including Galectin-3, LSECtin, α-synuclein, and Fibrinogen-like protein 1 (FGL1). The binding of these ligands to LAG-3 can inhibit T-cell function and proliferation. For example, FGL1 binding to the LAG-3 receptor on the surface of T cells inhibits T-cell proliferation and affects immune activity. Blocking the interaction between FGL1 and LAG-3 can enhance the antitumor effect of T lymphocytes, which is significant in tumor immunotherapy.
Creative Biolabs' Comprehensive LAG-3 Assay Portfolio Service
At Creative Biolabs, we have leveraged two decades of expertise to develop an industry-leading LAG-3 assay portfolio service. Our meticulously designed assays provide the critical data required to accelerate the discovery and development of next-generation LAG-3-targeting therapeutics. Our portfolio includes, but is not limited to, the following key assays:
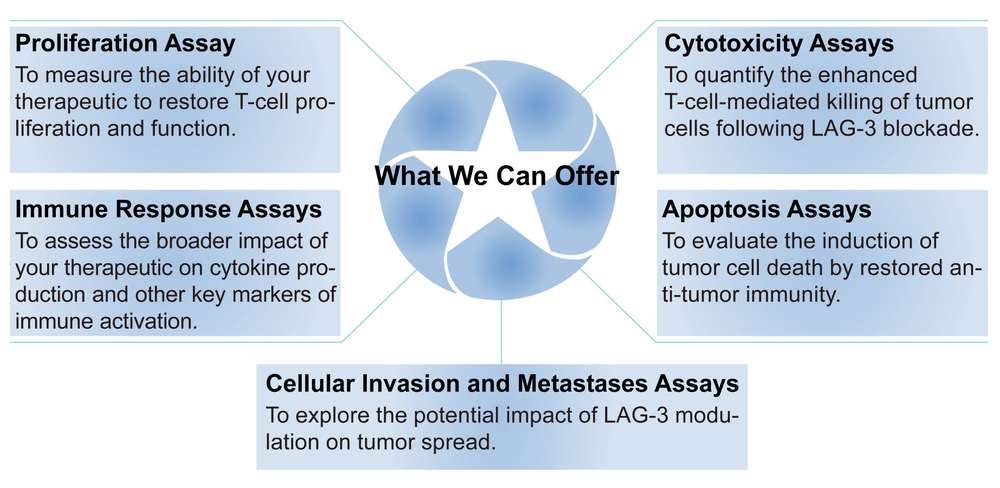
To learn more about how our LAG-3 assay portfolio can accelerate your therapeutic development, please contact us today for a confidential consultation.
Publication
This review details LAG-3, an inhibitory immune checkpoint. It suppresses T-cell function within the tumor microenvironment and synergizes with PD-1 to promote immune escape. The article highlights how dual LAG-3/PD-1 blockade can overcome treatment resistance in melanoma. It also covers LAG-3's role in autoimmunity, novel ligands like Galectin-3, and promising clinical trials of LAG-3-targeting therapies, underscoring its potential as a cancer immunotherapy target.
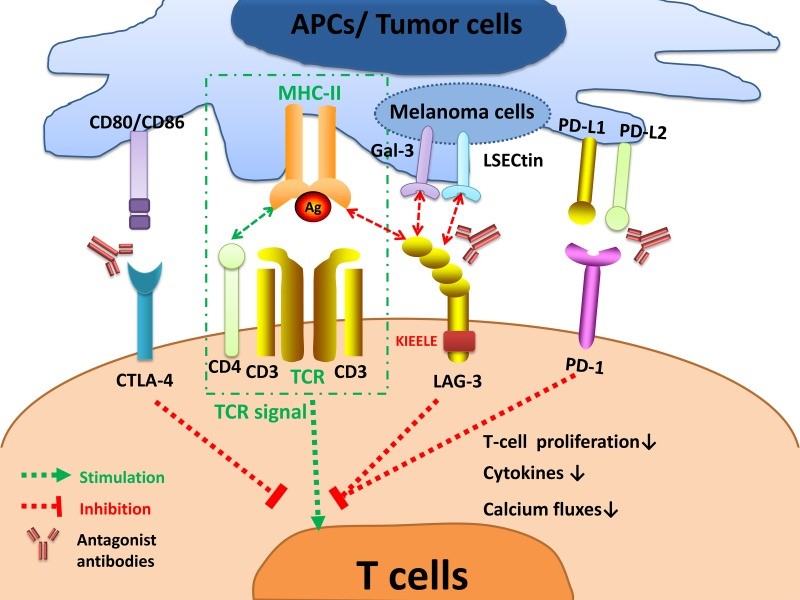 Fig.1 LAG-3 signaling and the interplay with other immune checkpoints. 1
Fig.1 LAG-3 signaling and the interplay with other immune checkpoints. 1
Why Choose Us?
Choosing Creative Biolabs means partnering with a leader in immuno-oncology. Our services are backed by two decades of experience, cutting-edge technology, and a steadfast commitment to scientific excellence. We offer a unique combination of precision, reproducibility, and flexibility to meet the most demanding research needs. Our team of seasoned specialists works with you to design a strategic assay plan, interpret complex results, and chart a clear path forward for your therapeutic program, providing a true partnership and bespoke solutions tailored to your specific research and development needs.
Discover how we can help your project succeed. Request a consultation today to begin the conversation about your specific goals.
FAQs
Q1: What is the primary advantage of using a LAG-3-specific assay over a more general T-cell proliferation assay?
A1: While general proliferation assays can measure T-cell response, our LAG-3-specific assays are meticulously designed to evaluate the precise impact of your therapeutic on the LAG-3 signaling pathway.
Q2: Can your assays be adapted for different therapeutic modalities, such as small molecules or CAR T cells
A2: Yes, our assay portfolio is highly versatile. We have extensive experience in adapting our protocols to evaluate a wide range of therapeutic modalities, including monoclonal antibodies, small molecule inhibitors, and advanced cell therapies like CAR T cells.
Q3: How does the LAG-3 assay portfolio compare to similar services
A3: Our key differentiators are our two decades of experience, our deep scientific expertise, and our comprehensive, all-in-one approach. We don't just provide data; we provide a full partnership, from initial consultation and project design to final data interpretation and expert recommendations.
Customer Review
-
Enhanced Proliferation Assays
The precision and reproducibility of the proliferation assays using Creative Biolabs' LAG-3 assay portfolio service in our research have significantly improved our ability to screen lead compounds. - J*hn A
-
Streamlined In Vitro Validation
The robust data from the cytotoxicity assays using Creative Biolabs' LAG-3 assay portfolio service in our research has significantly streamlined our in vitro validation process and provided a tangible advantage over competitors. - R*bert C
Related Services
Creative Biolabs offers a suite of complementary services to support all stages of your immuno-oncology research. You may also be interested in our:
Dendritic Cell Engineering Service
Creative Biolabs provides dendritic cells (DC) engineering services to optimize DC for therapeutic purposes. Our services facilitate the genetic manipulation of DCs to enhance their function in stimulating the immune system to fight against cancers, infections, and other diseases.
Learn More →
T Cell Engineering Service
Creative Biolabs provides expert T-cell engineering services to advance the field of immunotherapy. Our services, which include CAR T-cell therapy, T-cell receptor (TCR) therapy, and T-cell gene editing, are designed to help clients develop advanced therapies.
Learn More →
Contact Us
Creative Biolabs is your strategic partner in advancing the next generation of cancer immunotherapies. Our comprehensive LAG-3 assay portfolio service provides the critical data and expert support you need to accelerate your drug discovery and development.
Contact Our Team for More Information and to Discuss Your Project.
Reference
-
Long, Long, et al. "The promising immune checkpoint LAG-3: from tumor microenvironment to cancer immunotherapy." Genes & cancer 9.5-6 (2018): 176. Distributed under Open Access license CC BY 3.0, without modification. https://doi.org/10.18632/genesandcancer.180



 Fig.1 LAG-3 signaling and the interplay with other immune checkpoints. 1
Fig.1 LAG-3 signaling and the interplay with other immune checkpoints. 1
