T Cell Proliferation Assay Service
Creative Biolabs specializes in delivering a high-resolution, cell-subset-specific analysis: our optimized dye-based flow cytometry protocols reliably detect proliferation in both CD4+ and CD8+ T cell subsets, even when working with low-frequency, precious clinical samples. This level of detail is vital for understanding drug efficacy, especially in heterogeneous patient populations, and ensures your data is reproducible, robust, and regulatory-ready.
Background What We Can Offer Workflow Publication Why Choose Us FAQs Customer Review Related Services Contact Us
The Crucial Metrics in Immunological Advancement
T lymphocytes stand at the epicenter of adaptive immunity. Their precise activation and subsequent clonal expansion—or proliferation—are the fundamental biological events that define successful immune responses against pathogens, and more critically, against malignancies. For researchers and developers in the fields of immuno-oncology (IO) and autoimmune disease, accurately quantifying T cell proliferation is not just a standard bioassay; it is the cornerstone for validating therapeutic candidates, optimizing dosage, and dissecting complex immunomodulatory mechanisms. The need for standardized, highly reproducible functional assays has never been greater, particularly when analyzing rare, antigen-specific cell populations from precious samples.
Precision Methodology: Combining Sensitivity and Specificity
The quality of proliferation data hinges entirely on the methodology employed. To ensure maximum utility and relevance across diverse experimental contexts, Creative Biolabs offers and expertly executes two gold-standard approaches, emphasizing non-radioactive, high-resolution techniques:
|
Assay Type
|
Key Metric Measured
|
Resolution
|
Best Use Case
|
|
Flow Cytometry
|
Cell division number.
|
-
Single-cell
-
High specificity
|
-
Mechanistic studies;
-
Antigen-specific responses;
-
Treg suppression.
|
|
Luminescence
|
Metabolic activity or DNA synthesis.
|
|
-
Drug screening, potency;
-
Efficacy of the bulk population.
|
Contact our dedicated service specialists today to discuss your specific experimental design and receive a tailored quote.
Workflow: A Standardized Path to High-Quality Proliferation Data
We execute a rigorous, multi-stage workflow designed for maximum data quality and consistency, suitable for visualization as a clear project flowchart.
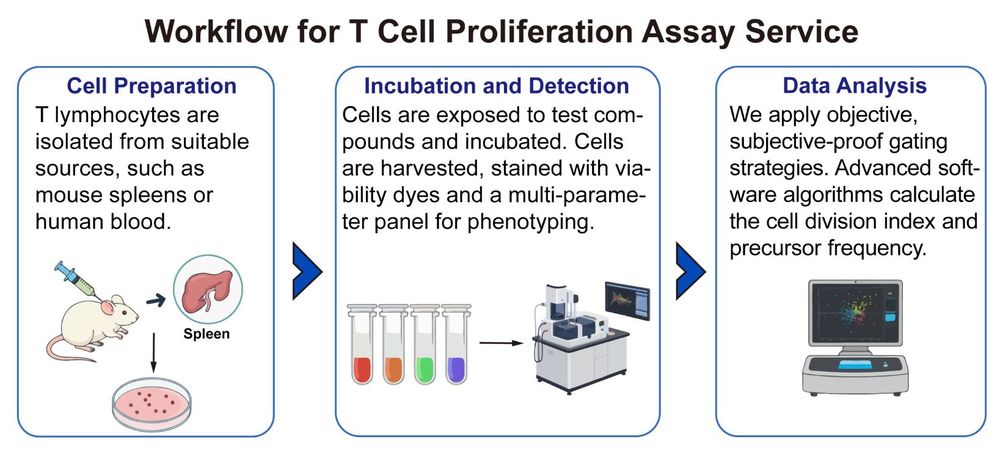
Publication
This study directly compared two methods for quantifying human T cell proliferation: the dilution-based probe and the bioorthogonal 5-ethynyl-2'-deoxyuridine (EdU) assay. The EdU method incorporates into DNA and uses a less harsh "click" reaction for detection. Side-by-side analysis showed the EdU assay was superior: it caused significantly less cytotoxicity in non-stimulated cells, offered higher sensitivity for detecting proliferating cells, and better discerned functional activation markers. The findings establish EdU as the less toxic and more sensitive method.
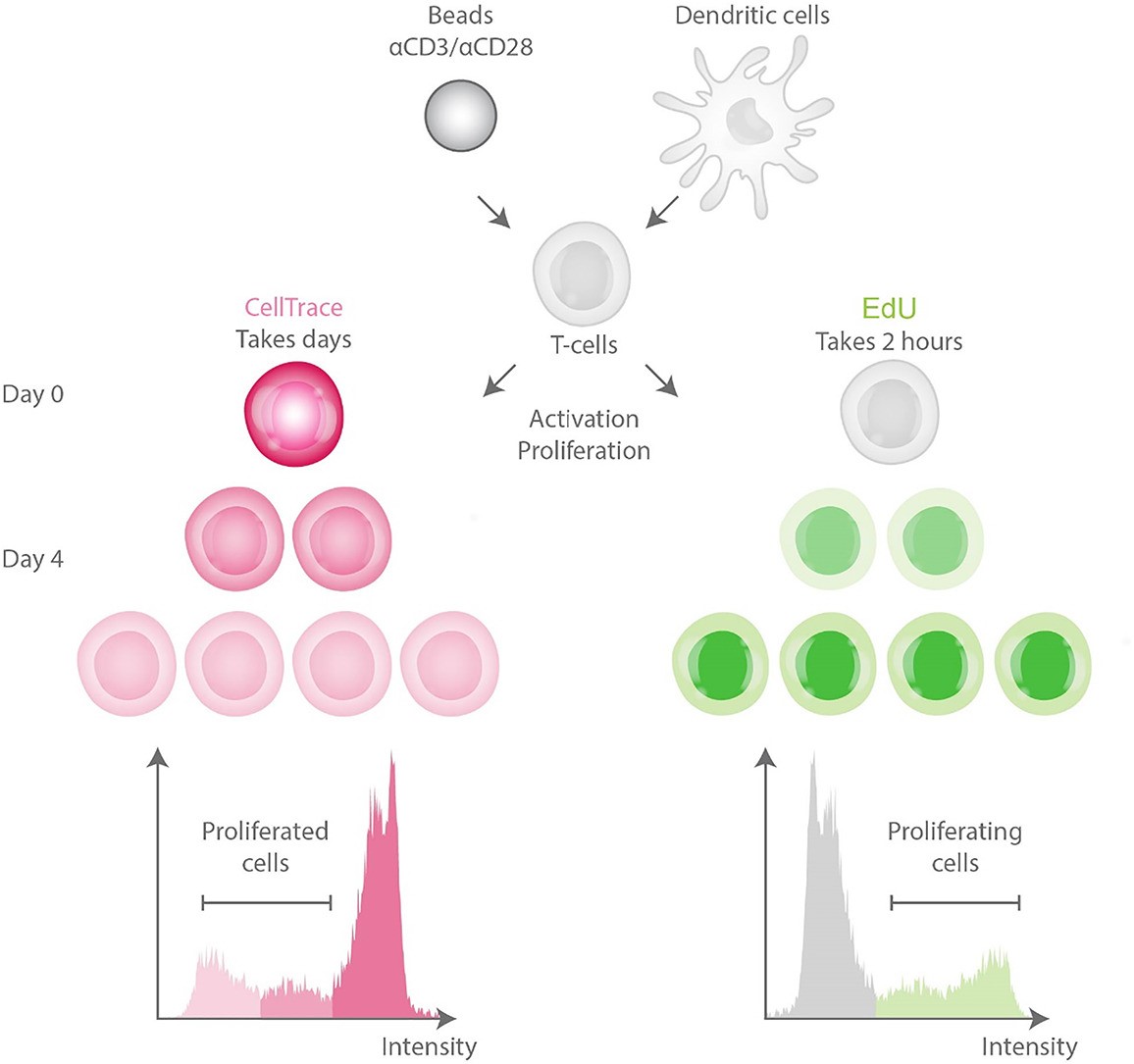 Fig.1 Comparison of T-cell proliferation assays. 1
Fig.1 Comparison of T-cell proliferation assays. 1
Why Choose Us
In a field where reproducibility and accuracy are non-negotiable, Creative Biolabs offers decisive advantages: We provide unrivaled customization and standardization, tailoring protocols to unique stimulation conditions and cell sources while strictly adhering to regulatory-grade practices for translational success. Our expert flow cytometry and data Interpretation team, composed of PhD-level immunologists, applies appropriate mathematical models and standardized, subject-proof gating strategies to overcome inter-operator variability. Furthermore, we maintain robust control systems, incorporating essential positive and negative controls and running assays in triplicate to ensure assay validity, data integrity, and high precision in cell division index calculation.
Partner with Creative Biolabs to unlock the true proliferative potential of your therapeutic candidates.
FAQs
Q1: Why should I switch from the traditional 3H-thymidine assay to a dye-based method?
A1: Dye-based assays eliminate the need for radioisotopes and, more importantly, provide single-cell resolution. Dye dilution allows you to distinguish between dividing and non-dividing cells and precisely quantify the number of cell divisions within specific CD4+ or CD8+ subsets.
Q2: How do you determine the best stimulation method (beads, mitogen, or antigen) for my study?
A2: The choice is based on your specific project goals. For broad, maximum T cell activation (e.g., cell QC), we use mitogens (PHA) or aAPC beads (anti-CD2/CD3/CD28), which provide a standardized, physiological trigger.
Q3: What is the cell division index (CDI), and why is it important for regulatory data?
A3: The CDI is a metric calculated from flow cytometry data that represents the average number of divisions experienced by the entire starting T cell population. It is preferred over simpler metrics because it incorporates both the frequency of responders and the average number of divisions.
Customer Review
-
High-Resolution Subset Analysis
Using Creative Biolabs' service in our research has significantly facilitated the accurate tracking of CD8+ proliferation kinetics. The CFSE data allowed us to see specific division peaks, giving us confidence in our studies. - J. Smi***
-
Regulatory-Grade Data
Using Creative Biolabs' service in our research has significantly improved the reproducibility of our Treg suppression assays for our trial. Their use of pooled unstimulated control means for calculation ensures the standard is consistently met, making the data regulatory-ready. - A. Joh***
Related Services
To achieve a comprehensive functional profile of your immunomodulatory candidate, we recommend pairing the T cell proliferation assay with these complementary services:
In Vitro Treg Suppression Assay
Creative Biolabs provides comprehensive Treg suppression assay services using FACS for high-purity isolation. We quantify Treg function by measuring Teff proliferation inhibition and cytokine secretion, ensuring high-quality, reproducible immunological data.
Learn More →
Regulatory T Cell Therapy Development Services
Creative Biolabs supports Treg therapy development through services like Treg isolation, genetic modification (CAR-Tregs), and functional profiling. We facilitate next-generation approaches to enhance antigen specificity and off-the-shelf potential.
Learn More →
Contact Us
To discuss your specific project needs, receive a detailed protocol design, and get a tailored quote, please reach out to our team of dedicated service specialists. Our expertise ensures your T cell assay data is precise, reliable, and clinically relevant.
Contact Our Team for More Information and to Discuss Your Project
Reference
-
Stempels, F. C., et al. "A sensitive and less cytotoxic assay for identification of proliferating T cells based on bioorthogonally-functionalized uridine analogue." Journal of immunological methods 502 (2022): 113228. Distributed under Open Access license CC BY 4.0, without modification. https://doi.org/10.1016/j.jim.2022.113228



 Fig.1 Comparison of T-cell proliferation assays. 1
Fig.1 Comparison of T-cell proliferation assays. 1
