Immunophenotyping based Autoimmune Disorder Recognition Services
Background Service Workflow Advantages Published Data FAQs Contact Us
Long diagnosis times, misdiagnosis, a lack of specific biomarkers, and challenges in monitoring disease progression are common issues in the biopharmaceutical field. Creative Biolabs offers immunophenotyping based autoimmune disorder recognition services to accelerate accurate diagnosis, enable personalized treatment strategies, and facilitate effective disease monitoring through advanced flow cytometry, mass cytometry, and comprehensive immune cell profiling.
The Imperative for Precision in Autoimmune Diagnostics
Autoimmune diseases are complex and highly variable, often presenting diagnostic challenges that lead to delayed or inaccurate diagnoses. Early and precise identification of these conditions is crucial for initiating timely and effective treatments, thereby preventing irreversible tissue damage and improving patient outcomes. Immunophenotyping offers a powerful solution by enabling the detailed characterization of immune cell subsets and their activation states, which is essential for understanding disease pathogenesis and discovering specific biomarkers. High-dimensional immunophenotyping, utilizing advanced techniques like spectral flow and mass cytometry, provides unparalleled resolution of complex immune landscapes in autoimmune diseases, aiding in differential diagnosis, predicting therapeutic responses, and monitoring disease activity.
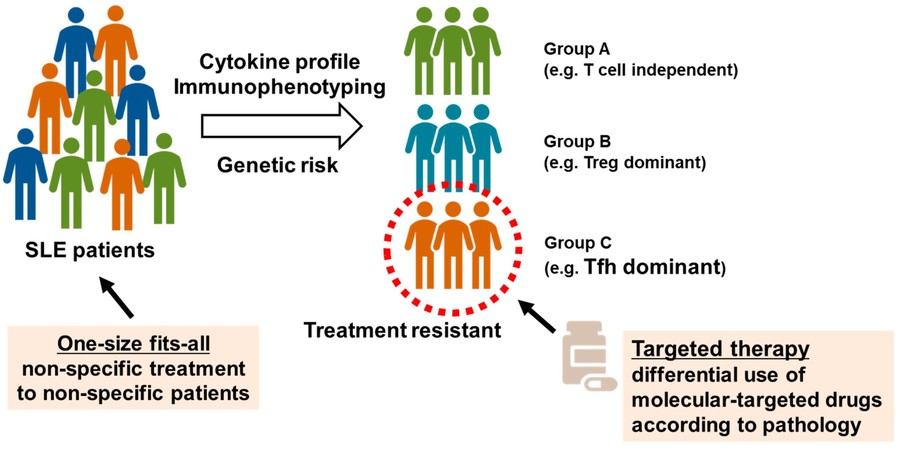 Fig.1 Determination of SLE subgroups using peripheral blood immunophenotyping.1,3
Fig.1 Determination of SLE subgroups using peripheral blood immunophenotyping.1,3
Immunophenotyping based Autoimmune Disorder Recognition Services at Creative Biolabs
Creative Biolabs' Immunophenotyping based Autoimmune Disorder Recognition Services provide comprehensive solutions for precise immune cell profiling, robust biomarker identification, accurate disease stratification, and reliable prediction of therapy responses. Our services are designed to help you navigate the complexity of autoimmune disease diagnosis and research, providing useful insights that propel your projects ahead. Our service can be applied to recognize the following autoimmune disorders, which include:
Workflow
This initial stage necessitates the meticulous isolation of target immune cells from client-provided samples. Rigorous viability assessments and comprehensive quality control checks are subsequently performed to ensure sample integrity and suitability for downstream analytical procedures, thereby minimizing experimental variability.
Our expert team collaborates with clients to design bespoke antibody panels. This process involves the judicious selection of optimal fluorochrome or metal-conjugated antibodies targeting specific immune cell markers. The resultant panels undergo rigorous optimization to ensure minimal spectral overlap and maximal resolution of desired immune cell subsets.
Prepared samples are subjected to analysis utilizing state-of-the-art high-dimensional cytometers. This methodology facilitates the simultaneous detection of numerous markers per cell, generating rich, multi-parametric data indicative of immune cell phenotypes and functions.
Raw data undergoes extensive pre-processing, which includes compensation for spectral overlap (in flow cytometry) or debarcoding (in mass cytometry). Advanced normalization techniques are then applied to mitigate batch effects and ensure data consistency across disparate experiments, thereby preparing the dataset for robust analytical evaluation.
Leveraging sophisticated bioinformatics pipelines, unsupervised clustering algorithms are employed to identify both novel and established immune cell populations. Furthermore, supervised machine learning algorithms and comprehensive statistical analyses are applied to ascertain cell frequencies, activation states, and functional characteristics, thereby revealing intricate immunological landscapes.
Our team of experienced immunologists and bioinformaticians provides expert interpretation of the analyzed data. Findings are correlated with clinical information, and comprehensive reports are generated, encompassing detailed flow cytometry plots, heatmaps, clustering analyses, and the delivery of actionable insights pertinent to the project's objectives.
Our Service Highlights
-
High-Resolution Immune Profiling: Enables early and accurate diagnosis of complex autoimmune conditions.
-
Robust Biomarker Discovery: Facilitates the identification of novel biomarkers, providing personalized treatment insights.
-
Accelerated Research Timelines: Streamlines the diagnostic and research processes, saving valuable time.
-
Comprehensive Data Analysis: Delivers precise, actionable results through expert interpretation of complex data.
-
Innovation-Driven Outcomes: Provides the critical insights necessary to drive groundbreaking advancements in immunology.
Published Data
This study utilized individual cell mass cytometric immunophenotyping to analyze peripheral blood mononuclear cells (PBMCs) from 52 participants, comprising 13 healthy controls and 13 therapy-naive patients each with rheumatoid arthritis (RA), progressive systemic sclerosis (SSc), and systemic lupus erythematosus (SLE). A total of 15,387,165 CD45+ live individual cells were analyzed using a 34-marker antibody panel. From this comprehensive immunophenotyping, 17 main cell types were identified, with seven of these exhibiting statistically significant differences in frequency across the investigated healthy and autoimmune disease groups.
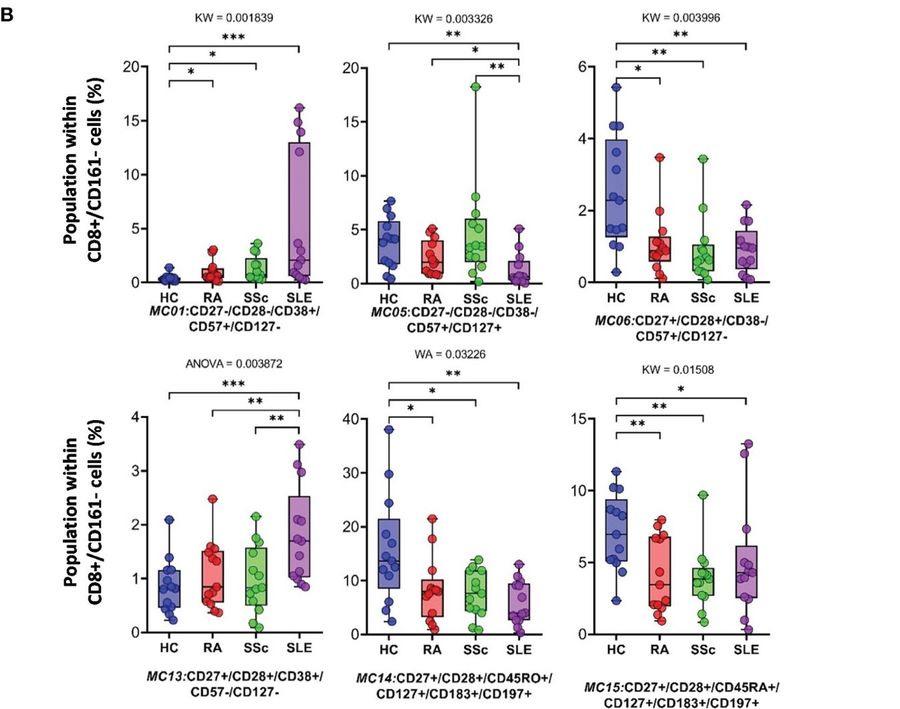 Fig.2 The CD8+/CD161-cytotoxic T cell subpopulations.2,4
Fig.2 The CD8+/CD161-cytotoxic T cell subpopulations.2,4
FAQs
Q1: What types of samples can be analyzed using your Immunophenotyping services?
A1: We can analyze a wide range of biological samples, including peripheral blood mononuclear cells (PBMCs), whole blood, bone marrow, and various tissue biopsies. Our team will guide you on the optimal sample collection and preparation for your specific project needs.
Q2: Can your services help in identifying novel therapeutic targets for autoimmune diseases?
A2: By providing a deep, high-resolution understanding of immune cell subsets and their functional states in autoimmune conditions, our services are instrumental in uncovering novel disease-associated cell populations and molecular pathways, which can serve as promising therapeutic targets.
Q3: What does the client need to provide to initiate the project?
A3: To initiate a project, clients typically need to provide relevant biological samples (e.g., PBMCs, whole blood, tissue biopsies), patient clinical history, preliminary diagnostic information, and specific research objectives or hypotheses for the study. Our team will guide you through the precise requirements based on your project's scope.
Contact Us
Creative Biolabs stands at the forefront of immunophenotyping technology, offering unparalleled expertise and cutting-edge solutions for autoimmune disorder recognition. With years of experience, our team leverages advanced spectral flow cytometry and mass cytometry platforms, ensuring the highest resolution and depth of immune profiling. If you want to know more for this service, please feel free to get in touch with us.
References
-
Nakayamada, Shingo, and Yoshiya Tanaka. "Immune Phenotype as a Biomarker for Systemic Lupus Erythematosus." Biomolecules vol. 13,6 960. 8 Jun. 2023, doi:10.3390/biom13060960.
-
Balog, József Á et al. "Comparative single-cell multiplex immunophenotyping of therapy-naive patients with rheumatoid arthritis, systemic sclerosis, and systemic lupus erythematosus shed light on disease-specific composition of the peripheral immune system." Frontiers in immunology vol. 15 1376933. 25 Apr. 2024, doi:10.3389/fimmu.2024.1376933.
-
Distributed under Open Access License CC BY 4.0, without modification.
-
Distributed under Open Access License CC BY 4.0. The original image was modified by extracting and using part B, and the title was changed to "The CD8+/CD161-cytotoxic T cell subpopulations".


 Fig.1 Determination of SLE subgroups using peripheral blood immunophenotyping.1,3
Fig.1 Determination of SLE subgroups using peripheral blood immunophenotyping.1,3
 Fig.2 The CD8+/CD161-cytotoxic T cell subpopulations.2,4
Fig.2 The CD8+/CD161-cytotoxic T cell subpopulations.2,4
