Immunophenotyping based Systemic Sclerosis Recognition Service
Background Service Workflow Advantages FAQs Related Sections Contact Us
Are you currently facing challenges in accurately diagnosing or monitoring systemic sclerosis (SSc) progression, or seeking precise stratification for clinical trials? Creative Biolabs' immunophenotyping based systemic sclerosis recognition service helps you accelerate research and clinical development by providing high-quality, actionable insights into SSc pathology through advanced immunophenotyping techniques.
The Critical Need for SSc Immunophenotyping
Systemic sclerosis (SSc) is a complex autoimmune disease characterized by widespread fibrosis and vascular damage, presenting significant diagnostic and prognostic challenges. Current diagnostic methods often lack the specificity needed for early detection and precise disease stratification. Immunophenotyping, by characterizing immune cell subsets, offers a crucial avenue for identifying novel biomarkers and understanding disease pathogenesis. The development of advanced recognition services based on immunophenotyping is essential to improve early diagnosis, monitor disease activity, predict complications like interstitial lung disease (ILD) and vascular issues, and ultimately guide more effective therapeutic strategies.
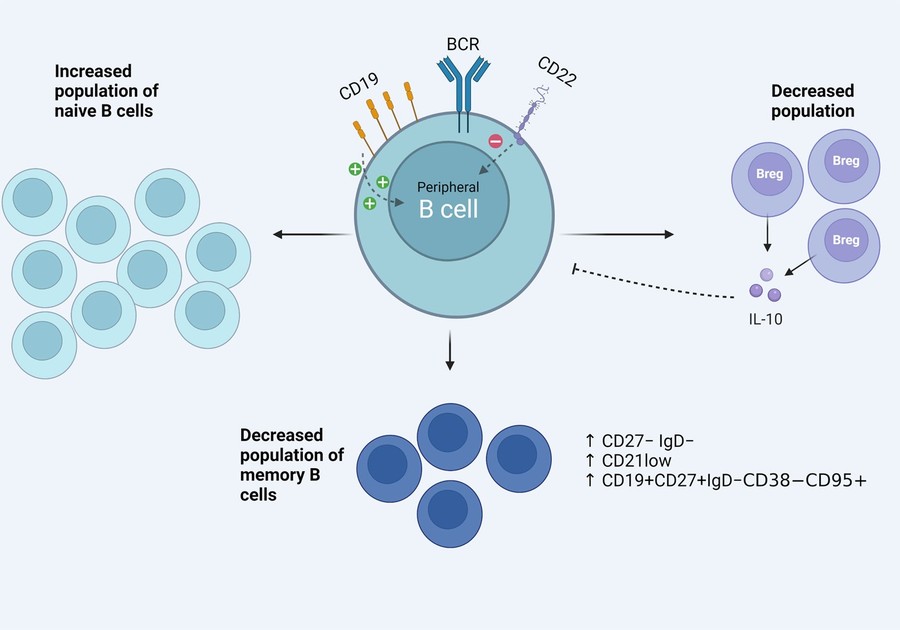 Fig.1 Activated B cell phenotype from SSc patients.1
Fig.1 Activated B cell phenotype from SSc patients.1
Immunophenotyping based Systemic Sclerosis Recognition Service at Creative Biolabs
At Creative Biolabs, our immunophenotyping based systemic sclerosis recognition service offers comprehensive immune profiling by meticulously processing your biological samples (typically PBMCs) using optimized multi-parameter flow cytometry and advanced bioinformatics. This process, typically completed within 4 to 10 weeks, delivers detailed data reports, statistical analyses highlighting potential biomarkers, and expert scientific interpretations. Creative Biolabs ensures the highest data quality, accuracy, and reproducibility through stringent quality control measures at every stage, providing you with confidence in your research findings for SSc diagnosis, prognosis, and therapeutic response.
Required Starting Materials
-
Peripheral Blood Mononuclear Cells (PBMCs): Freshly isolated or cryopreserved PBMCs from SSc patients or relevant control groups.
-
Clinical Data & Patient Cohort Information: Detailed clinical histories, disease activity scores, treatment regimens, and any relevant imaging or biopsy data for comprehensive correlation.
-
Specific Research Objectives: Clear definition of desired outcomes, such as biomarker identification, patient stratification, or therapeutic response monitoring.
Workflow
Step 1: Sample Reception & Quality Control
Upon receiving your samples, our expert team performs rigorous quality checks to ensure sample integrity and viability, crucial for accurate downstream analysis.
Step 2: Immunophenotyping Panel Design & Optimization
Based on your project goals and the latest scientific literature, we design and optimize custom or pre-validated multi-parameter flow cytometry. This involves selecting specific antibodies against relevant immune cell markers (e.g., CD19, CD21, CD27, IgD, CD38, CD95, HLA-DR, tBET, CD86, etc.).
Step 3: Data Acquisition
Samples are processed using state-of-the-art flow cytometers, ensuring high-resolution data collection for a broad range of immune cell populations and their activation states.
Step 4: Advanced Data Analysis & Bioinformatics
aw data undergoes sophisticated bioinformatics analysis, including dimensionality reduction techniques and clustering algorithms, to identify distinct immune cell subsets and their frequencies. We also perform statistical comparisons between different patient groups.
Step 5: Biological Interpretation & Reporting
Our experienced scientists interpret the complex immunophenotyping data in the context of SSc pathogenesis and your specific research questions, identifying key immune signatures and potential biomarkers.
Our Service Highlights
-
Precision & Depth: High-resolution analysis of diverse immune cell subsets, revealing subtle but critical phenotypic changes.
-
Biomarker Discovery: Identification of novel diagnostic, prognostic, and predictive biomarkers for SSc and its complications.
-
Disease Stratification: Enhanced ability to stratify SSc patients into distinct subgroups for targeted therapies and clinical trials.
-
Mechanism Elucidation: Deeper understanding of immune cell involvement in SSc pathogenesis, guiding therapeutic development.
-
Accelerated Research: Streamlined workflow and expert analysis reduce your internal workload and accelerate project timelines.
FAQs
Q1: What types of samples can I submit for this service?
A1: We primarily work with peripheral blood mononuclear cells (PBMCs), which can be submitted fresh or cryopreserved. If you have other sample types you'd like to discuss, please don't hesitate to contact our team to explore feasibility for your specific project.
Q2: How does this service help in identifying new therapeutic targets for SSc?
A2: By providing a detailed characterization of immune cell subsets and their activation states in SSc patients, our service helps pinpoint specific cellular pathways and phenotypic markers that are dysregulated in the disease. These identified dysregulations can serve as promising targets for novel therapeutic interventions.
Q3: Can this service differentiate between different SSc subtypes or disease stages?
A3: Absolutely. Our advanced immunophenotyping panels and bioinformatics analysis are specifically designed to identify distinct immune signatures that correlate with various SSc subtypes (e.g., diffuse vs. limited cutaneous SSc) and disease stages, including the presence of complications like interstitial lung disease (ILD) or vascular involvement.
Q4: How does Creative Biolabs ensure the accuracy and reproducibility of the immunophenotyping data?
A4: We maintain the highest standards of quality through stringent quality control measures at every stage, from sample handling and panel optimization to data acquisition and analysis. Our instruments are regularly calibrated, and all protocols are standardized and validated by experienced scientists, ensuring exceptional accuracy, reproducibility, and consistency in your data.
Related Sections
To further enhance your autoimmune disease research and development, Creative Biolabs extends its expertise beyond SSc immunophenotyping with a comprehensive portfolio of related services:
Why Choose Creative Biolabs
Creative Biolabs leads the field in immunophenotyping services, combining unparalleled expertise with cutting-edge technology and a steadfast commitment to scientific excellence. Our dedicated team's deep knowledge of SSc immunology ensures the highest quality results and insightful interpretations for your critical projects. Please feel free to get in touch with us with your meaningful projects.
Reference
-
Melissaropoulos, Konstantinos et al. "Pathogenetic Aspects of Systemic Sclerosis: A View Through the Prism of B Cells." Frontiers in immunology vol. 13 925741. 23 Jun. 2022, doi:10.3389/fimmu.2022.925741. Distributed under Open Access License CC BY 4.0, without modification.
For Research Use Only | Not For Clinical Use


 Fig.1 Activated B cell phenotype from SSc patients.1
Fig.1 Activated B cell phenotype from SSc patients.1
